Revolutionizing Aromatic Primary Amine Production with Advanced OTf Amination Technology
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct aromatic primary amine motifs, which serve as critical building blocks for active pharmaceutical ingredients (APIs) and advanced intermediates. A recent technological breakthrough documented in patent CN113387842A introduces a highly efficient method for synthesizing aromatic primary amines through OTf (triflate) amination. This innovation addresses long-standing challenges in organic synthesis by replacing hazardous traditional reduction pathways with a sophisticated palladium-catalyzed cross-coupling strategy. By utilizing benzophenone imine as a stable ammonia equivalent, this process circumvents the safety risks associated with explosive reducing agents while delivering exceptional purity profiles. For global procurement leaders and R&D directors, this represents a pivotal shift towards safer, more sustainable, and economically viable manufacturing protocols that align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of aromatic primary amine groups has relied heavily on the reduction of nitro compounds or the reaction of aryl halides with ammonia water. These conventional pathways are fraught with significant industrial drawbacks that hinder efficient commercial scale-up of complex polymer additives and pharmaceutical intermediates. The use of flammable and explosive reducing agents creates a hazardous working environment, necessitating expensive safety infrastructure and rigorous risk management protocols that drive up operational costs. Furthermore, these reduction systems often lack chemoselectivity, particularly when dealing with multifunctional molecules like amino acids containing both amino and carboxyl groups. This lack of selectivity leads to numerous side reactions, resulting in low conversion rates and the formation of difficult-to-remove impurity compounds that complicate downstream purification processes and negatively impact overall yield.
The Novel Approach
In stark contrast, the novel OTf amination method described in the patent data offers a transformative solution by leveraging the reactivity of aromatic triflates with benzophenone imine. This approach eliminates the need for dangerous reducing agents entirely, substituting them with a controlled catalytic system that operates under mild and manageable conditions. The process is characterized by its operational simplicity and low energy consumption, making it inherently safer and more environmentally friendly than legacy technologies. By avoiding the harsh conditions typical of nitro reductions, this method preserves the integrity of sensitive functional groups, thereby minimizing side reactions and ensuring a cleaner reaction profile. This strategic shift not only enhances the safety of the manufacturing facility but also drastically simplifies post-reaction treatment, allowing for more straightforward isolation of the target aromatic primary amine compounds with superior quality.
Mechanistic Insights into Pd-Catalyzed Amination and Deprotection
The core of this synthetic innovation lies in a two-step sequence that begins with a palladium-catalyzed cross-coupling reaction. In the first step, an aromatic triflate (Compound A-1) undergoes oxidative addition with a palladium catalyst, such as Pd2(dba)3 combined with ligands like BINAP. This activated complex then reacts with benzophenone imine in the presence of a base like cesium carbonate within an aprotic solvent system. The reaction proceeds through a well-defined catalytic cycle to form the protected intermediate Compound A-2. This mechanistic pathway is highly advantageous because the bulky benzophenone imine group acts as a protecting group that prevents over-alkylation and ensures mono-amination, a common issue when using free ammonia. The subsequent step involves the acidic hydrolysis of the benzhydryl group under mild conditions using solvents like tetrahydrofuran and aqueous acid. This deprotection step is rapid and efficient, cleanly revealing the free aromatic primary amine without degrading the sensitive amino acid backbone.
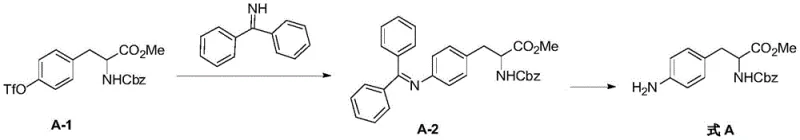
Impurity control is another critical aspect where this mechanism excels, providing significant value for R&D teams focused on purity specifications. Traditional reduction methods often generate complex mixtures of hydroxylamines and azo compounds that are structurally similar to the product, making chromatographic separation challenging and costly. The OTf amination route, however, generates byproducts that are chemically distinct from the target molecule, such as benzophenone and triflic acid salts, which are easily removed during the aqueous workup and filtration stages. The patent data indicates that the final product achieves a purity greater than 99%, demonstrating the high selectivity of the catalytic system. This level of purity is essential for pharmaceutical applications where strict regulatory limits on genotoxic impurities and heavy metals must be met, ensuring that the final API intermediate is safe for subsequent biological testing and clinical use.
How to Synthesize Aromatic Primary Amine Efficiently
To implement this synthesis effectively, manufacturers must adhere to precise reaction parameters regarding temperature, stoichiometry, and solvent selection. The process begins with the preparation of the reaction mixture under an inert atmosphere to prevent catalyst oxidation, followed by careful temperature control during the amination phase. Detailed standardized synthesis steps are crucial for reproducibility and safety, ensuring that the delicate balance between catalyst activity and substrate stability is maintained throughout the batch cycle. Operators should refer to the specific molar ratios and solvent volumes outlined in the technical documentation to optimize yield and minimize waste generation during the scale-up process.
- Perform amination reaction on aromatic OTf and benzophenone imine using Pd catalyst and base in aprotic solvent at 100-130°C.
- Isolate the intermediate compound A-2 through filtration and concentration.
- Conduct benzhydryl removal reaction on compound A-2 in aprotic oxygen-containing solvent under acidic conditions to obtain the target aromatic primary amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this OTf amination technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The elimination of hazardous reducing agents significantly reduces the regulatory burden and insurance costs associated with storing and handling dangerous chemicals, leading to substantial cost savings in facility management. Moreover, the use of cheap and easily available raw materials ensures a stable supply chain that is less susceptible to market volatility compared to specialized reducing agents. The mild operation conditions also mean lower energy consumption for heating and cooling, contributing to a reduced carbon footprint and aligning with corporate sustainability goals. This process optimization allows for a more predictable production schedule, reducing lead time for high-purity intermediates and ensuring consistent availability for downstream customers.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven by the simplification of the synthetic route and the avoidance of expensive safety measures required for explosive reagents. By removing the need for complex hazard mitigation systems, manufacturers can allocate resources more efficiently, resulting in a lower cost of goods sold. Additionally, the high yield and purity reduce the amount of raw material wasted on failed batches or extensive purification efforts, further enhancing the overall cost-effectiveness of the production line. The ability to use standard laboratory equipment without specialized high-pressure or cryogenic setups also lowers capital expenditure requirements for new production lines.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on commodity chemicals such as aromatic triflates and benzophenone imine, which are widely available from multiple global suppliers. This diversification of raw material sources mitigates the risk of supply disruptions that can occur with niche reagents used in traditional methods. The robustness of the reaction conditions means that production is less likely to be halted due to minor fluctuations in environmental controls, ensuring a steady flow of materials to meet customer demand. This reliability is critical for maintaining long-term contracts with pharmaceutical clients who require guaranteed delivery schedules for their own manufacturing timelines.
- Scalability and Environmental Compliance: From an environmental perspective, this method supports green chemistry initiatives by minimizing waste generation and avoiding the release of toxic byproducts. The simple post-treatment process involving filtration and extraction reduces the volume of solvent waste, lowering disposal costs and environmental impact. The process is inherently scalable, allowing for seamless transition from kilogram-scale development to multi-ton commercial production without significant re-engineering of the process flow. This scalability ensures that the technology can grow with market demand, providing a future-proof solution for the manufacturing of complex amino acid derivatives and other high-value intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the patent specifications and are intended to provide clarity on the operational benefits and safety profile of the technology. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the OTf amination method safer than traditional nitro reduction?
A: Traditional nitro reduction often requires flammable and explosive reducing agents, posing significant safety risks during industrial scale-up. The OTf amination method utilizes stable benzophenone imine and palladium catalysis under mild conditions, eliminating the need for hazardous reducing systems and greatly enhancing operational safety.
Q: How does this method improve purification and yield?
A: Conventional methods often suffer from side reactions due to incompatible functional groups like amino or carboxyl groups, leading to difficult purification. This novel route avoids such interference, resulting in simpler post-reaction treatment, easier purification via standard column chromatography, and consistently high yields exceeding 99% purity in validated examples.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is specifically designed for industrial popularization. It features cheap and easily available raw materials, low energy consumption due to moderate temperature requirements, and a short synthetic route that minimizes waste generation, making it highly favorable for green and environment-friendly large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Primary Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global fine chemical market. Our team of expert chemists has thoroughly evaluated the OTf amination pathway described in CN113387842A and confirmed its potential for robust industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this newer, safer technology is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aromatic primary amine we produce meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this innovative process. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. By partnering with us, you gain access to a reliable supply of high-quality intermediates produced via cutting-edge chemistry that prioritizes safety, sustainability, and economic efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
