Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
Advanced Metal-Free Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those functionalized with trifluoromethyl groups which enhance metabolic stability and lipophilicity. A significant breakthrough in this domain is detailed in patent CN110467579B, which discloses a novel preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a more sustainable, iodine-promoted protocol. The strategic importance of the 1,2,4-triazole scaffold cannot be overstated, as it serves as a critical bioisostere in numerous active pharmaceutical ingredients (APIs) and functional materials. As illustrated in the structural diversity of known bioactive molecules, the ability to efficiently install a trifluoromethyl group at the 5-position opens vast avenues for drug discovery and material science applications.
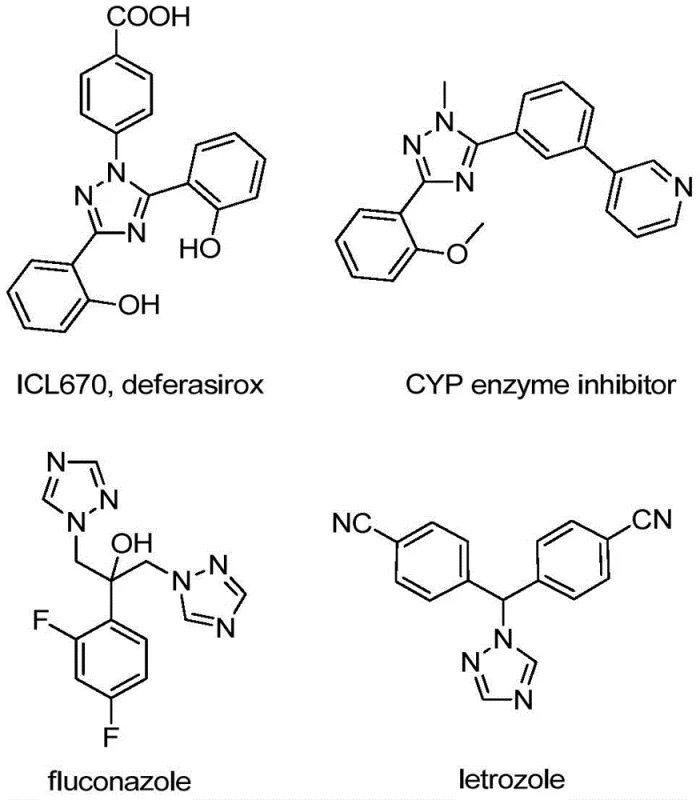
This patent specifically addresses the long-standing challenges associated with trifluoromethylation by utilizing readily available hydrazones and trifluoroethylimidoyl chlorides. For R&D directors and procurement managers alike, this development signals a move towards simpler supply chains and reduced dependency on scarce noble metals. The method's operational simplicity, avoiding stringent anhydrous or oxygen-free environments, further underscores its potential for immediate adoption in GMP manufacturing facilities aiming for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen heterocycles has been fraught with synthetic inefficiencies and economic burdens. Conventional literature reports primarily describe two dominant strategies, both of which present significant drawbacks for large-scale operations. The first approach involves the direct trifluoromethylation of pre-synthesized nitrogen heterocycles. This pathway typically necessitates the use of specialized, often hazardous, and prohibitively expensive trifluoromethylating reagents. These reagents not only drive up the raw material costs but also introduce complex safety handling protocols due to their potential instability. Furthermore, the second mainstream method relies on the reaction of trifluoromethyl-bearing synthons, such as trifluorodiazoethane, with coupling substrates. While effective in academic settings, trifluorodiazoethane is notoriously difficult to handle on an industrial scale due to its explosive nature and the requirement for specialized equipment. Additionally, many existing protocols rely heavily on transition metal catalysts, which pose severe challenges regarding residual metal limits in final drug substances, necessitating expensive purification steps to meet regulatory standards.
The Novel Approach
In stark contrast to these cumbersome traditional routes, the methodology disclosed in patent CN110467579B offers a streamlined, metal-free alternative that fundamentally simplifies the synthetic landscape. By employing trifluoroethylimidoyl chloride and hydrazones as the primary building blocks, this novel approach bypasses the need for dangerous diazo compounds or exotic trifluoromethyl sources. The core innovation lies in the use of elemental iodine as a promoter in conjunction with a mild base, sodium acetate. This combination facilitates a cascade reaction that efficiently constructs the triazole ring with high regioselectivity. The reaction conditions are remarkably benign, proceeding effectively in common organic solvents like 1,2-dichloroethane (DCE) at moderate temperatures ranging from 80°C to 100°C. Crucially, the process does not demand rigorous exclusion of moisture or oxygen, significantly lowering the barrier to entry for manufacturing facilities. This robustness translates directly into operational expenditure savings and enhanced process safety, making it an ideal candidate for the cost reduction in API manufacturing.
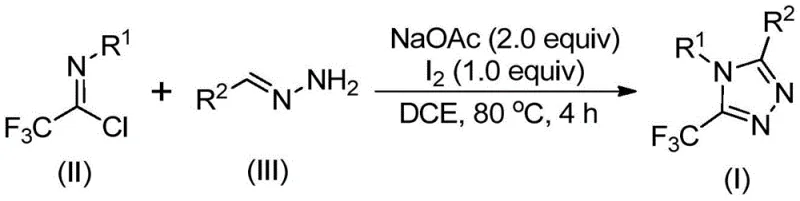
Mechanistic Insights into Iodine-Promoted Cyclization
To fully appreciate the technical elegance of this synthesis, one must delve into the proposed mechanistic pathway which ensures high conversion and purity. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the trifluoroethylimidoyl chloride and the hydrazone substrate. This initial condensation yields a trifluoroacetamidine intermediate, setting the stage for the subsequent ring closure. Following this, an isomerization event occurs, positioning the molecule for the critical oxidative step. The addition of elemental iodine acts as a mild oxidant, promoting an oxidative iodination that generates a reactive iodine-containing intermediate. This species is pivotal, as it activates the system for the final intramolecular electrophilic substitution. The cyclization is completed through an aromatization process, driven by the thermodynamic stability of the resulting 1,2,4-triazole ring, ultimately delivering the 5-trifluoromethyl substituted product with excellent fidelity.
The beauty of this mechanism lies in its tolerance for diverse functional groups, a key requirement for a reliable pharmaceutical intermediate supplier. The mild nature of the iodine promoter and sodium acetate base ensures that sensitive moieties on the aromatic rings of the starting materials remain intact. As demonstrated by the variety of substituents tolerated in the patent examples, including halogens, alkyl groups, and heterocycles, the electronic properties of the substrates do not severely impede the reaction progress. This broad substrate scope allows medicinal chemists to rapidly generate libraries of analogues for structure-activity relationship (SAR) studies without redesigning the core synthetic route. The avoidance of strong acids or bases further minimizes side reactions such as hydrolysis or polymerization, thereby simplifying the impurity profile and reducing the burden on downstream purification processes.
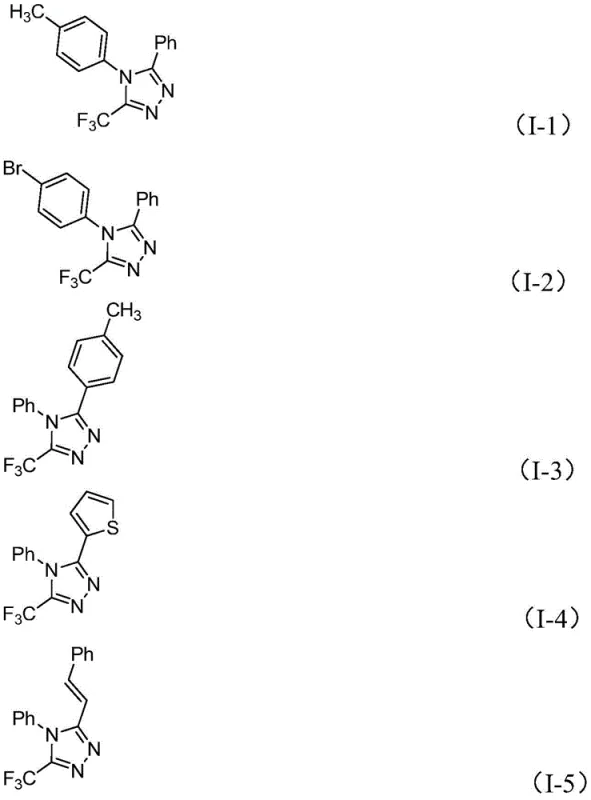
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and thermal profiles to maximize yield and minimize waste. The patent outlines a straightforward protocol where the molar ratio of trifluoroethylimidoyl chloride to hydrazone is optimized, typically favoring an excess of the hydrazone to drive the equilibrium forward. The detailed standardized synthesis steps below provide a clear roadmap for technical teams to replicate these results, ensuring consistency across batches. By following these guidelines, manufacturers can achieve the high purity specifications required for clinical grade materials while maintaining operational efficiency.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane (DCE).
- Heat the reaction mixture to 80-100°C and stir for 2 to 4 hours to facilitate the initial condensation.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to promote oxidative cyclization, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this iodine-promoted methodology offers tangible strategic benefits beyond mere chemical curiosity. The primary advantage stems from the drastic simplification of the raw material portfolio. By eliminating the need for precious metal catalysts and unstable diazo reagents, the supply chain becomes significantly more resilient and less prone to market volatility. The reliance on commodity chemicals like sodium acetate and elemental iodine, alongside easily sourced hydrazones, ensures a steady flow of inputs. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients. Furthermore, the simplified work-up procedure, which often involves basic filtration and standard chromatography, reduces the consumption of silica gel and solvents, contributing to a leaner and more cost-effective manufacturing operation.
- Cost Reduction in Manufacturing: The economic implications of adopting this metal-free route are profound. Traditional methods often incur hidden costs associated with the procurement, storage, and disposal of heavy metal catalysts. By completely removing transition metals from the process, manufacturers save substantially on catalyst acquisition costs and, more importantly, on the expensive scavenging resins and activated carbon treatments required to reduce metal residues to ppm levels. Additionally, the reaction's ability to proceed without strict anhydrous conditions reduces the energy load associated with solvent drying and inert gas purging. These cumulative efficiencies lead to a significantly lower cost of goods sold (COGS), allowing for more competitive pricing in the global marketplace without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the geopolitical and logistical complexities surrounding rare earth metals and specialized reagents. This novel synthesis mitigates such risks by utilizing universally available starting materials. Trifluoroethylimidoyl chloride and hydrazones are produced by multiple vendors worldwide, preventing single-source bottlenecks. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in environmental controls, ensuring consistent batch-to-batch quality. This reliability is paramount for long-term contracts where delayed shipments can result in severe penalties. Consequently, partnering with a supplier utilizing this technology provides a buffer against supply shocks, ensuring that critical API intermediates are available exactly when needed.
- Scalability and Environmental Compliance: As the industry moves towards greener chemistry, the environmental footprint of a synthesis route is a key decision factor. This method aligns perfectly with green chemistry principles by avoiding toxic heavy metals and minimizing hazardous waste generation. The use of elemental iodine, which can potentially be recovered and recycled, further enhances the sustainability profile. From a scalability perspective, the exothermic profile of the reaction is manageable, and the lack of gas evolution (unlike diazo decomposition) makes it safer to scale from gram to kilogram and eventually to tonnage production. The simplified post-treatment reduces the volume of organic waste solvents requiring incineration, helping facilities meet increasingly stringent environmental regulations and reducing disposal costs.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of a new synthetic route is essential for making informed sourcing decisions. The following questions address common inquiries regarding the practical application and compatibility of this 5-trifluoromethyl-1,2,4-triazole synthesis. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: Does this synthesis require expensive heavy metal catalysts?
A: No, the patented method utilizes inexpensive elemental iodine as a promoter, completely eliminating the need for toxic and costly transition metal catalysts like palladium or copper.
Q: What are the optimal reaction conditions for this triazole synthesis?
A: The reaction proceeds efficiently in aprotic solvents like dichloroethane (DCE) at temperatures between 80°C and 100°C, without the need for strict anhydrous or anaerobic conditions.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process uses cheap, commercially available starting materials and simple post-treatment steps like filtration and column chromatography, making it highly amenable to kilogram-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless. We understand the critical nature of stringent purity specifications in the pharmaceutical sector and have invested heavily in rigorous QC labs equipped with state-of-the-art analytical instrumentation. This infrastructure allows us to guarantee the identity and purity of every batch of 5-trifluoromethyl-1,2,4-triazole derivatives we produce, giving our partners complete confidence in their supply chain.
We invite forward-thinking R&D and procurement leaders to collaborate with us to leverage this advanced synthesis technology for their next-generation drug candidates. By optimizing this route for your specific needs, we can deliver a Customized Cost-Saving Analysis that highlights the specific economic benefits for your project. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Together, we can accelerate the development of life-saving medicines through superior chemical innovation and reliable supply.
