Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles: A Scalable Route for Pharmaceutical Intermediates
Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles: A Scalable Route for Pharmaceutical Intermediates
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign synthetic routes. A significant advancement in this domain is detailed in patent CN110467579B, which discloses a robust preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds. These heterocyclic scaffolds are ubiquitous in medicinal chemistry, serving as critical cores in a wide array of bioactive molecules ranging from antifungal agents to kinase inhibitors. The strategic incorporation of a trifluoromethyl group into these structures often enhances metabolic stability, lipophilicity, and bioavailability, making them highly prized assets in drug discovery pipelines. This technical insight report analyzes the novel iodine-promoted cyclization strategy, offering R&D directors and procurement leaders a clear view of its potential to streamline supply chains for high-purity pharmaceutical intermediates.
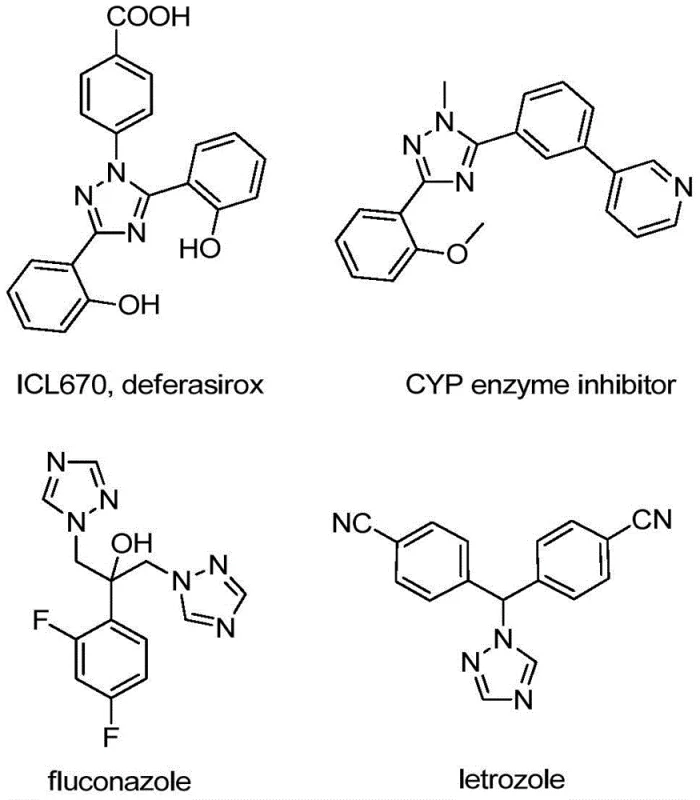
The significance of the 1,2,4-triazole motif cannot be overstated, as evidenced by its presence in blockbuster drugs like fluconazole and letrozole, depicted in the structural overview. However, accessing these fluorinated heterocycles has historically presented synthetic challenges. Conventional methods often rely on harsh conditions or expensive reagents that complicate scale-up. The methodology outlined in the patent addresses these pain points directly by utilizing readily available hydrazones and trifluoroacetimidoyl chlorides. By shifting the paradigm towards a non-metallic, iodine-promoted system, this approach not only broadens the substrate scope but also aligns with modern green chemistry principles, offering a compelling value proposition for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethyl-substituted nitrogen-containing heterocycles has been fraught with inefficiencies that hinder commercial viability. Traditional literature reports primarily describe two pathways: the direct trifluoromethylation of pre-synthesized heterocycles or the cycloaddition of trifluoromethyl synthons. The former often necessitates the use of specialized and costly trifluoromethylating reagents, which can be economically prohibitive for large-scale production. Furthermore, these reactions frequently require stringent anhydrous and anaerobic conditions to prevent reagent decomposition, adding significant operational complexity and infrastructure costs to the manufacturing process. The alternative pathway, utilizing synthons like trifluorodiazoethane, introduces severe safety hazards due to the explosive nature of diazo compounds, creating substantial risk management burdens for production facilities. Additionally, many existing protocols rely on transition metal catalysts, which introduce the risk of heavy metal contamination in the final API, necessitating expensive and time-consuming purification steps to meet regulatory limits.
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent leverages a simple yet highly effective iodine-promoted cyclization between hydrazones and trifluoroacetimidoyl chlorides. This strategy fundamentally alters the economic and operational landscape of triazole synthesis. By employing elemental iodine as a promoter rather than a stoichiometric oxidant or a toxic heavy metal catalyst, the process achieves high efficiency while maintaining a benign safety profile. The reaction proceeds smoothly in common aprotic solvents like dichloroethane at moderate temperatures, typically around 80°C, eliminating the need for cryogenic conditions or specialized pressure vessels. This operational simplicity translates directly into reduced capital expenditure and lower energy consumption. Moreover, the tolerance for various functional groups on both the hydrazone and the imidoyl chloride allows for the rapid generation of diverse chemical libraries, enabling medicinal chemists to explore structure-activity relationships with greater speed and flexibility.
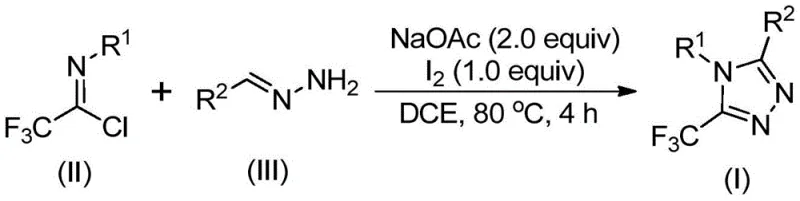
Mechanistic Insights into Iodine-Promoted Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D teams aiming to optimize the process for specific substrates. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone and the trifluoroacetimidoyl chloride, yielding a trifluoroacetamidine intermediate. This step is facilitated by sodium acetate, which acts as a mild base to scavenge the generated hydrochloric acid without inducing unwanted side reactions. Following this condensation, the system undergoes an isomerization that positions the nucleophilic nitrogen for the subsequent cyclization event. The introduction of elemental iodine then triggers a base-promoted oxidative iodination, generating a reactive iodine-containing intermediate. This species is pivotal, as it activates the system for the final intramolecular electrophilic substitution. The cyclization is followed by spontaneous aromatization, driven by the thermodynamic stability of the resulting 1,2,4-triazole ring, to furnish the final 5-trifluoromethyl substituted product. This elegant cascade avoids the formation of stable off-cycle intermediates, ensuring high atom economy and minimizing waste generation.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. The absence of transition metals eliminates the formation of metal-complexed byproducts that are notoriously difficult to remove. Furthermore, the mild reaction conditions (80°C) suppress thermal degradation pathways that often plague high-temperature heterocycle syntheses. The use of sodium acetate as a buffer helps maintain a consistent pH environment, preventing the hydrolysis of the sensitive imidoyl chloride starting material. For procurement managers, this implies a cleaner crude reaction profile, which significantly reduces the burden on downstream purification units. The ability to isolate high-purity products via standard silica gel chromatography or even simple crystallization in some cases demonstrates the robustness of the chemical design, ensuring that the final material meets the stringent purity specifications required for GMP manufacturing of active pharmaceutical ingredients.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Efficiently
The practical execution of this synthesis is designed for ease of operation, making it accessible for both laboratory-scale optimization and pilot plant campaigns. The protocol involves a straightforward one-pot procedure where the key reagents are combined in a specific sequence to maximize yield and minimize side reactions. The initial mixing of sodium acetate, trifluoroacetimidoyl chloride, and the hydrazone derivative in a solvent like dichloroethane sets the stage for the condensation phase. Heating this mixture allows the reaction to reach completion before the oxidative cyclization step is initiated. The delayed addition of iodine is a critical process parameter that ensures the condensation occurs prior to oxidation, preventing premature consumption of the reagents. Detailed standardized operating procedures for this synthesis, including precise stoichiometry and work-up protocols, are essential for reproducibility.
- Mix sodium acetate, trifluoroacetimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane.
- Heat the reaction mixture to 80°C and stir for 2 to 4 hours to facilitate initial condensation.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to complete the oxidative cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement managers, the adoption of this synthetic route represents a strategic opportunity to enhance supply security and reduce total landed costs. The shift away from exotic reagents and hazardous conditions mitigates many of the risks associated with sourcing complex intermediates. By relying on commodity chemicals like hydrazones and imidoyl chlorides, manufacturers can diversify their supplier base and avoid single-source bottlenecks. The operational simplicity of the process also means that production lead times can be significantly shortened, as there is no need for extensive equipment preparation or specialized safety protocols associated with pyrophoric or explosive reagents. This agility allows for more responsive inventory management and faster time-to-market for new drug candidates entering clinical trials.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts such as palladium or copper results in substantial direct cost savings on raw materials. More importantly, it removes the necessity for costly metal scavenging resins or complex chromatographic purification steps required to meet residual metal limits in pharmaceuticals. The use of inexpensive elemental iodine and sodium acetate further drives down the bill of materials. Additionally, the ability to run the reaction without strict anhydrous or anaerobic conditions reduces utility costs related to nitrogen purging and solvent drying, contributing to a leaner and more cost-efficient manufacturing process overall.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically the hydrazones and trifluoroacetimidoyl chlorides, are derived from widely available aldehydes and amines. This reliance on bulk chemical feedstocks ensures a stable and continuous supply chain, insulating production schedules from the volatility often seen with specialized fine chemical reagents. The robustness of the reaction conditions means that production is less susceptible to delays caused by equipment failures or environmental constraints. Consequently, partners can rely on consistent delivery schedules and improved capacity planning, which is critical for maintaining uninterrupted API production lines.
- Scalability and Environmental Compliance: The process has been demonstrated to be easily scalable from gram to multi-kilogram quantities without loss of efficiency, indicating strong potential for ton-scale commercial production. The avoidance of toxic heavy metals simplifies waste stream management and reduces the environmental footprint of the manufacturing site. Effluent treatment becomes more straightforward as the waste does not contain regulated heavy metal contaminants, lowering disposal costs and ensuring compliance with increasingly stringent environmental regulations. This sustainability profile aligns well with the corporate social responsibility goals of major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 5-trifluoromethyl-1,2,4-triazole synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing frameworks.
Q: Does this synthesis require expensive heavy metal catalysts?
A: No, the method described in patent CN110467579B utilizes elemental iodine as a promoter instead of toxic or expensive transition metals like palladium or copper, significantly simplifying downstream purification.
Q: What are the typical reaction conditions for this triazole formation?
A: The reaction proceeds efficiently in aprotic solvents like dichloroethane at moderate temperatures around 80°C, without the need for strict anhydrous or anaerobic conditions.
Q: Is this method suitable for large-scale manufacturing?
A: Yes, the process uses cheap, commercially available starting materials and avoids sensitive conditions, making it highly amenable to commercial scale-up from gram to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of global drug development programs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering materials that adhere to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Whether you require custom synthesis of novel triazole derivatives or established scale-up of known intermediates, our infrastructure is designed to support your journey from bench to bedside with unwavering quality assurance.
We invite you to engage with our technical procurement team to discuss how this advanced iodine-promoted synthesis can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages this route offers compared to your current supply chain. We encourage potential partners to contact us to obtain specific COA data for our catalog compounds and to request comprehensive route feasibility assessments tailored to your unique molecular targets. Let us collaborate to drive innovation and efficiency in your pharmaceutical manufacturing operations.
