Scalable Metal-Free Synthesis of Alpha-Sulfonyl Pyrrole Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing highly functionalized heterocycles, particularly pyrrole derivatives, which serve as critical scaffolds in bioactive molecules and organic materials. Patent CN102911104A introduces a groundbreaking approach for preparing pyrrole derivatives bearing a sulfonyl group on the alpha-substituent, utilizing a novel sulfonyl migration strategy from 3-aza-1,5-enyne precursors. This technology represents a significant leap forward in heterocyclic chemistry, offering a metal-free pathway that circumvents the limitations of traditional transition-metal catalyzed cyclizations. By leveraging a thermal rearrangement mechanism in polar aprotic solvents, this method achieves high atom economy and produces densely functionalized products suitable for downstream derivatization. For global procurement teams and R&D directors, this patent outlines a scalable route to complex intermediates that aligns with modern green chemistry principles while maintaining rigorous structural control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of substituted pyrroles has heavily relied on transition-metal catalysis, such as palladium or copper-mediated coupling reactions, which introduce significant complexities into the manufacturing supply chain. These conventional methods often necessitate stringent removal of trace heavy metals to meet pharmaceutical regulatory standards, adding costly purification steps like scavenging or extensive chromatography. Furthermore, many classical pyrrole syntheses suffer from poor regioselectivity or require harsh reaction conditions that limit the tolerance of sensitive functional groups, thereby restricting the chemical diversity accessible to medicinal chemists. The reliance on expensive catalysts and the generation of stoichiometric metal waste also pose environmental challenges, increasing the overall carbon footprint and disposal costs associated with large-scale production. Consequently, there is a persistent demand for alternative strategies that can deliver high-purity heterocycles without the baggage of metal contamination.
The Novel Approach
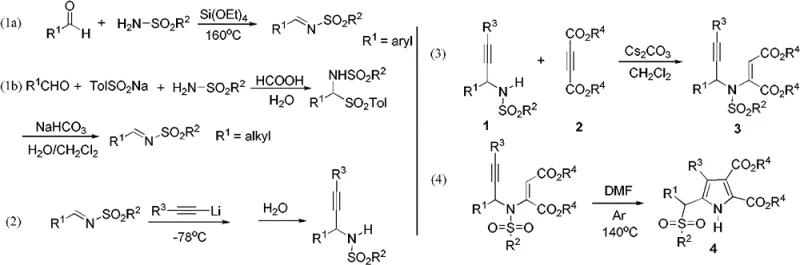
The methodology described in the patent data offers a transformative solution by employing a catalyst-free thermal rearrangement of 3-aza-1,5-enynes to generate the target pyrrole skeleton. This innovative route utilizes readily available starting materials, such as N-sulfonyl-allylamines and activated alkynes, which are combined under mild basic conditions to form the key enyne intermediate. As illustrated in the reaction scheme, the subsequent heating of this intermediate in DMF triggers a spontaneous sulfonyl migration, effectively constructing the pyrrole ring while installing the sulfonyl moiety at the alpha-position in a single operational step. This approach not only simplifies the synthetic sequence but also ensures that the nitrogen atom of the pyrrole ring remains unsubstituted (N-H), providing a versatile handle for further chemical modification. The elimination of metal catalysts streamlines the workup procedure, making this process exceptionally attractive for cost-sensitive commercial manufacturing.
Mechanistic Insights into Thermal Sulfonyl Migration
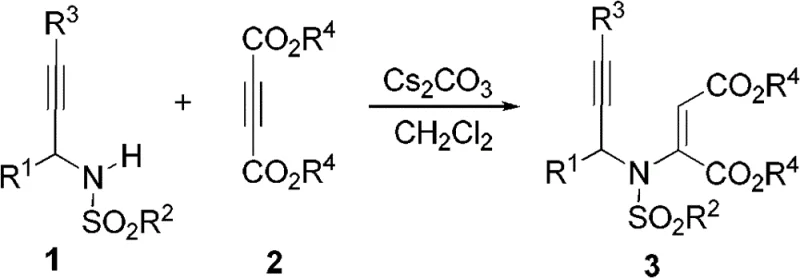
The core of this technological advancement lies in the unique intramolecular rearrangement where the sulfonyl group migrates from the nitrogen atom to the carbon backbone during cyclization. Mechanistically, the reaction proceeds through a concerted or stepwise pathway where the N-S bond is cleaved and a new C-S bond is formed simultaneously with the closure of the five-membered pyrrole ring. This sulfonyl migration is driven by thermal energy in a polar solvent environment, which stabilizes the transition state and facilitates the reorganization of electron density across the conjugated enyne system. The absence of external catalysts suggests that the reaction is governed by the inherent electronic properties of the substrate, specifically the electron-withdrawing nature of the ester groups and the sulfonyl moiety, which activate the system for nucleophilic attack and subsequent rearrangement. Understanding this mechanism is crucial for process optimization, as it allows chemists to fine-tune reaction parameters such as temperature and solvent polarity to maximize conversion rates.
From an impurity control perspective, this metal-free mechanism offers distinct advantages by eliminating the formation of metal-complexed side products that are notoriously difficult to separate. The high selectivity of the thermal migration ensures that the primary byproduct profile is limited to unreacted starting materials or simple hydrolysis products, which can be easily removed via standard crystallization or chromatography techniques. The preservation of the N-H bond in the final product is particularly valuable, as it prevents the formation of N-alkylated impurities that often plague other pyrrole synthesis methods. This clean reaction profile translates directly to higher isolated yields and reduced solvent consumption during purification, addressing key pain points for supply chain managers focused on efficiency. The ability to produce highly functionalized pyrroles with predictable substitution patterns makes this chemistry a powerful tool for building complex molecular libraries.
How to Synthesize Alpha-Sulfonyl Pyrrole Derivatives Efficiently
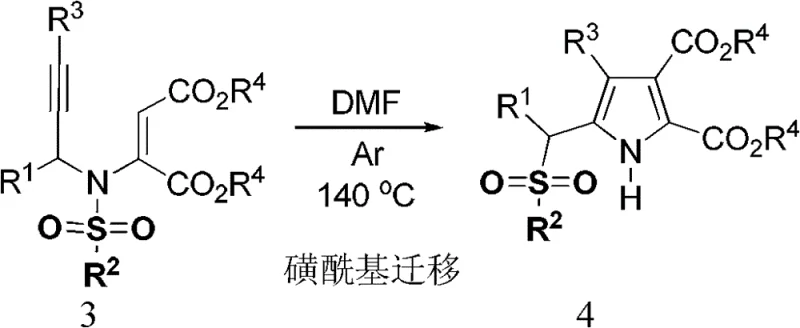
The practical implementation of this synthesis involves a straightforward two-stage protocol that begins with the preparation of the 3-aza-1,5-enyne precursor followed by the thermal cyclization step. The initial condensation of N-sulfonyl-allylamine with dialkyl acetylenedicarboxylate is performed in dichloromethane using cesium carbonate as a base, yielding the enyne intermediate in moderate to good yields after chromatographic purification. This intermediate is then subjected to heating in N,N-dimethylformamide (DMF) under an inert argon atmosphere, where the temperature is maintained between 100°C and 145°C for a duration of 2 to 12 hours. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- Synthesize the 3-aza-1,5-enyne precursor by reacting N-sulfonyl-allylamine with dialkyl acetylenedicarboxylate using Cs2CO3 in dichloromethane.
- Purify the intermediate enyne via silica gel column chromatography to ensure high purity before the rearrangement step.
- Heat the purified 3-aza-1,5-enyne in DMF at 100-145°C under argon protection for 2-12 hours to effect sulfonyl migration and cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free pyrrole synthesis route offers substantial strategic benefits regarding cost stability and operational reliability. By removing the dependency on precious metal catalysts, manufacturers can insulate their production costs from the volatile pricing of commodities like palladium or rhodium, leading to more predictable budgeting and long-term supply contracts. The simplified purification process, which avoids the need for specialized metal scavengers, reduces both the direct material costs and the time required for quality control testing, thereby accelerating the overall time-to-market for new drug candidates. Furthermore, the use of common industrial solvents like DMF and dichloromethane ensures that the process can be easily integrated into existing manufacturing infrastructure without requiring significant capital investment in new equipment.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost driver from the bill of materials, as these reagents are often expensive and required in specific loadings to maintain activity. Additionally, the high atom economy of the sulfonyl migration reaction means that a greater proportion of the raw material mass is converted into the final product, minimizing waste disposal fees and maximizing raw material utilization efficiency. The streamlined workup procedure, which relies on standard solvent evaporation and silica gel chromatography, further reduces labor and utility costs associated with complex aqueous extractions or distillation processes required for metal removal.
- Enhanced Supply Chain Reliability: Sourcing high-purity transition metal catalysts can sometimes be subject to geopolitical constraints or supply shortages, whereas the reagents used in this protocol, such as cesium carbonate and DMF, are commodity chemicals with robust global supply chains. The robustness of the thermal reaction conditions also implies a lower risk of batch failure due to catalyst deactivation or sensitivity to trace impurities in the feedstock, ensuring consistent production output. This reliability is critical for maintaining continuous supply to downstream pharmaceutical customers who require strict adherence to delivery schedules and quality specifications.
- Scalability and Environmental Compliance: The metal-free nature of this process significantly simplifies environmental compliance, as there is no need to treat wastewater for heavy metal content, which is a stringent regulatory requirement in many jurisdictions. The reaction generates minimal hazardous waste, aligning with green chemistry initiatives and reducing the environmental footprint of the manufacturing site. Scaling this process from laboratory to commercial tonnage is facilitated by the simplicity of the thermal operation, which does not require specialized pressure vessels or intricate dosing systems, making it an ideal candidate for large-scale production of pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfonyl migration technology for pyrrole synthesis. These answers are derived directly from the experimental data and mechanistic understanding provided in the patent literature, offering clarity on process capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this chemistry into their development pipelines.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented process operates under metal-free conditions, utilizing thermal energy in DMF to drive the sulfonyl migration, which significantly reduces heavy metal contamination risks.
Q: What is the atom economy of this pyrrole synthesis route?
A: The reaction exhibits high atom economy as all atoms from the 3-aza-1,5-enyne reactant are incorporated into the final pyrrole derivative structure without generating significant byproduct waste.
Q: Can the nitrogen atom on the pyrrole ring be further functionalized?
A: Yes, the resulting pyrrole derivative retains a hydrogen atom on the nitrogen, allowing for subsequent N-alkylation or other functionalization reactions to diversify the chemical library.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this metal-free sulfonyl migration route for producing high-value pyrrole intermediates essential for modern drug discovery. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless and efficient. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, verifying that every batch of pyrrole derivative meets the exacting standards required for clinical and commercial applications. We are committed to leveraging this innovative chemistry to deliver cost-effective solutions that enhance your R&D productivity.
We invite you to collaborate with us to optimize your supply chain for these critical heterocyclic building blocks. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free process can reduce your overall cost of goods. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules, and let us help you secure a reliable supply of high-quality pyrrole derivatives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
