Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
Scalable Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for nitrogen-containing heterocycles, particularly 1,2,4-triazoles, which serve as critical scaffolds in numerous bioactive molecules. Patent CN113105402B discloses a groundbreaking preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds that addresses long-standing challenges in heterocyclic synthesis. This technology is particularly relevant for the production of high-value active pharmaceutical ingredients (APIs) such as Maraviroc, Sitagliptin, and Deferasirox, all of which rely on the unique physicochemical properties imparted by the triazole ring system. The introduction of a trifluoromethyl group into these heterocyclic frameworks significantly enhances metabolic stability, lipophilicity, and bioavailability, making this synthetic methodology a vital asset for modern drug discovery and development pipelines.
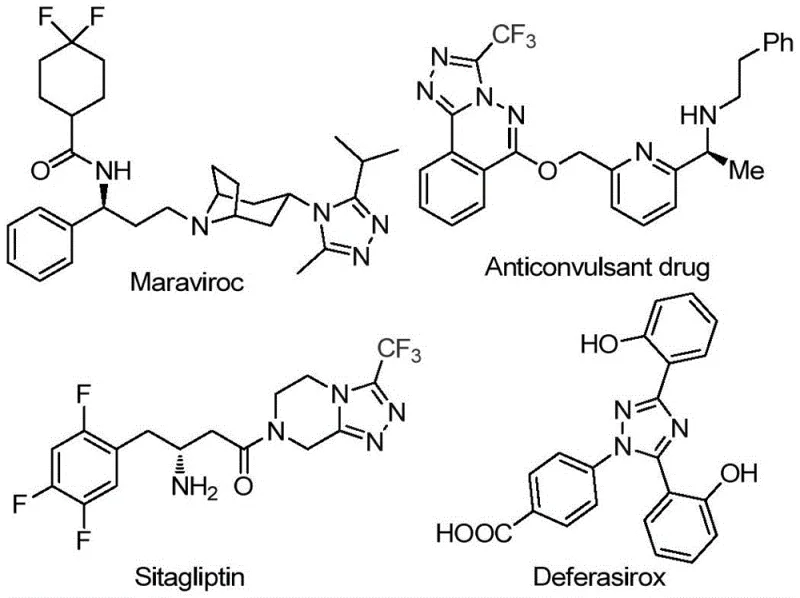
For R&D directors and process chemists, the significance of this patent lies in its ability to construct complex molecular architectures with high precision and minimal environmental impact. The disclosed method utilizes a non-metal promoted strategy, specifically leveraging elemental iodine and dimethyl sulfoxide (DMSO) to drive the reaction forward. This approach circumvents the stringent requirements often associated with transition metal catalysis, such as the need for rigorous anhydrous and oxygen-free environments. By simplifying the operational parameters, this technology lowers the barrier to entry for synthesizing these valuable intermediates, thereby accelerating the timeline from laboratory bench to pilot plant. The versatility of the reaction allows for the incorporation of diverse substituents at the 3, 4, and 5 positions, providing medicinal chemists with a powerful tool for structure-activity relationship (SAR) studies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polysubstituted 1,2,4-triazoles, especially those bearing both trifluoromethyl and acyl groups, has been fraught with significant technical hurdles. Traditional methodologies often rely on the use of expensive and toxic heavy metal catalysts, which not only inflate the raw material costs but also introduce complex downstream purification challenges to meet strict residual metal specifications required by regulatory bodies. Furthermore, many existing protocols demand harsh reaction conditions, including high temperatures, strong acids or bases, and strictly inert atmospheres, which complicate process safety and increase energy consumption. The limited substrate scope of older methods frequently restricts the types of functional groups that can be tolerated, forcing chemists to employ lengthy protection and deprotection strategies that reduce overall yield and efficiency. These factors collectively contribute to higher manufacturing costs and longer lead times, creating bottlenecks in the supply chain for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in patent CN113105402B offers a streamlined and economically viable alternative that leverages a tandem iodination and Kornblum oxidation sequence. This method utilizes cheap and readily available aryl ethyl ketones and trifluoroethylimide hydrazides as starting materials, reacting them in the presence of elemental iodine and a base system comprising sodium dihydrogen phosphate and pyridine. The reaction proceeds efficiently in DMSO, which acts as both the solvent and an oxidant, eliminating the need for external oxidizing agents. As demonstrated by the successful synthesis of various derivatives shown in the patent examples, this protocol exhibits excellent functional group tolerance, accommodating electron-donating and electron-withdrawing groups alike. The simplicity of the workup procedure, involving basic filtration and standard column chromatography, further underscores the practicality of this method for both small-scale research and larger-scale production.
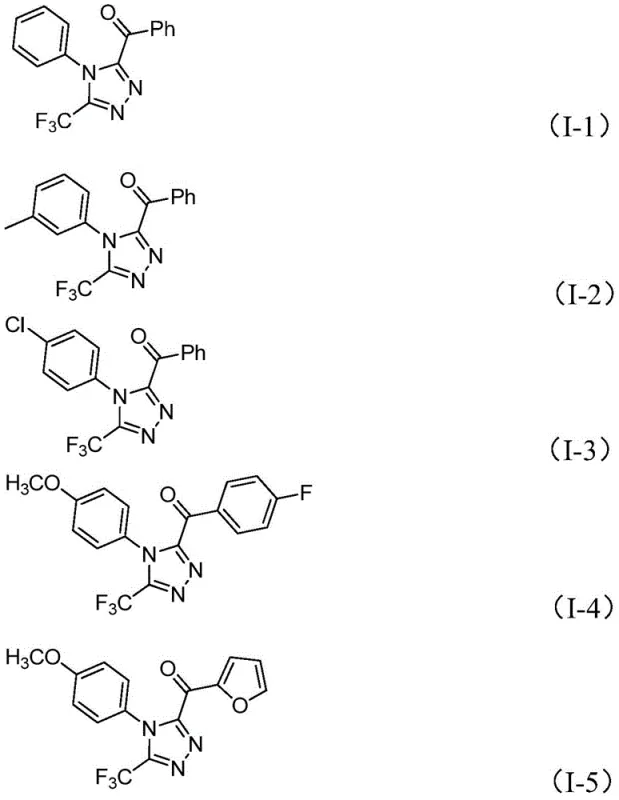
Mechanistic Insights into Iodine-Promoted Cyclization
The mechanistic pathway of this transformation is a sophisticated interplay of oxidation and condensation reactions that ultimately leads to the formation of the triazole ring. The process likely initiates with the iodination of the aryl ethyl ketone, followed by a Kornblum oxidation mediated by DMSO to generate an aryl diketone intermediate in situ. This highly reactive diketone species then undergoes a dehydration condensation with the trifluoroethylimide hydrazide to form a hydrazone intermediate. Subsequently, under the promotion of iodine and the basic environment provided by pyridine and phosphate salts, an intramolecular cyclization occurs. This final cyclization step closes the five-membered ring, yielding the desired 3,4,5-trisubstituted 1,2,4-triazole compound. Understanding this mechanism is crucial for process optimization, as it highlights the dual role of iodine as both an activator and a promoter, and the essential function of DMSO beyond merely being a solvent.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed routes. The absence of transition metals eliminates the risk of metal-induced side reactions or the formation of difficult-to-remove metal-organic complexes. The primary byproducts are likely inorganic salts and reduced iodine species, which are generally easier to separate from the organic product through aqueous workups or crystallization. The use of sodium dihydrogen phosphate helps buffer the reaction medium, preventing excessive acidity that could lead to the decomposition of sensitive functional groups or the hydrolysis of the triazole ring. This controlled environment ensures a cleaner reaction profile, resulting in higher crude purity and reducing the burden on downstream purification processes. For quality assurance teams, this translates to a more predictable impurity profile and greater confidence in meeting stringent pharmacopeial standards for final drug substances.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazole Efficiently
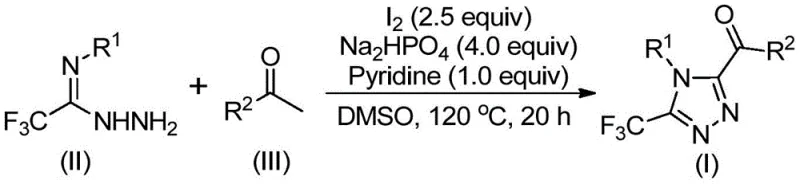
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize yield and minimize side reactions. The patent outlines a two-stage heating protocol where the initial oxidation of the ketone is performed at a moderate temperature before the addition of the hydrazide and subsequent cyclization at a higher temperature. This staged approach ensures that the reactive diketone intermediate is generated efficiently before being consumed by the nucleophilic attack of the hydrazide. Operators should ensure that the molar ratios of iodine, base, and substrates are maintained within the specified ranges to drive the reaction to completion. Detailed standardized synthesis steps see the guide below.
- Combine aryl ethyl ketone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the solution to 110-130°C for 12-20 hours to complete the cyclization, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iodine-promoted synthesis method presents a compelling value proposition centered on cost efficiency and supply reliability. The shift away from precious metal catalysts to abundant elemental iodine represents a significant reduction in raw material volatility and cost exposure. Since iodine and the requisite aryl ketones are commodity chemicals with stable global supply chains, manufacturers can secure long-term contracts with favorable pricing, insulating their operations from the market fluctuations often seen with specialized catalytic reagents. Furthermore, the elimination of heavy metals simplifies the waste treatment process, potentially lowering disposal costs and reducing the environmental footprint of the manufacturing facility. This aligns perfectly with the growing industry emphasis on green chemistry and sustainable manufacturing practices.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the substitution of expensive catalysts with inexpensive iodine and the use of DMSO, a low-cost polar aprotic solvent. By removing the need for rigorous anhydrous and oxygen-free conditions, the method reduces capital expenditure on specialized equipment such as gloveboxes or high-purity nitrogen lines, allowing production to occur in standard glass-lined reactors. The simplified post-treatment process, which avoids complex metal scavenging steps, further reduces labor and material costs associated with purification. These cumulative savings contribute to a lower cost of goods sold (COGS), enhancing the competitiveness of the final pharmaceutical intermediate in the global market.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials significantly mitigates supply chain risks. Aryl ethyl ketones and trifluoroethylimide hydrazides are widely produced by multiple chemical suppliers, ensuring that sourcing bottlenecks are unlikely to disrupt production schedules. The robustness of the reaction conditions means that the process is less susceptible to variations in raw material quality or minor deviations in operating parameters, leading to consistent batch-to-batch reproducibility. This reliability is critical for maintaining continuous supply to downstream API manufacturers, preventing costly delays in drug development or commercial launch timelines. Additionally, the scalability of the method ensures that supply can be rapidly ramped up to meet increasing demand without the need for extensive process re-engineering.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers substantial advantages that facilitate regulatory compliance and community acceptance. The absence of toxic heavy metals reduces the hazard profile of the process, lowering the risk of worker exposure and environmental contamination. The use of DMSO, while requiring proper handling, is well-understood in industrial settings and can be effectively managed with standard ventilation and containment systems. The generation of less hazardous waste streams simplifies the permitting process for new manufacturing lines and reduces the liability associated with waste disposal. As regulatory agencies worldwide tighten restrictions on pharmaceutical manufacturing emissions, adopting such cleaner technologies positions companies as responsible corporate citizens and future-proofs their operations against evolving environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazole synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this method into their existing manufacturing portfolios. It clarifies the operational boundaries and potential benefits, ensuring that expectations are aligned with the proven capabilities of the technology.
Q: What are the key advantages of this iodine-promoted synthesis method?
A: This method eliminates the need for expensive and toxic heavy metal catalysts, operates under air and moisture tolerant conditions, and utilizes cheap, commercially available starting materials like aryl ketones.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly states that the reaction can be easily expanded to the gram level and beyond, making it suitable for commercial scale-up due to its simple operation and robust conditions.
Q: What is the substrate scope for R1 and R2 groups?
A: The method demonstrates broad functional group tolerance, accommodating substituted or unsubstituted aryl and heteroaryl groups, including those with methyl, methoxy, halogen, or trifluoromethyl substituents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocyclic synthesis in the development of next-generation therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into viable industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our dedication to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking to optimize their supply chains and accelerate their drug development programs.
We invite you to explore how our capabilities align with your project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis methods can enhance your project's success. Let us collaborate to bring your innovative molecules to market faster and more efficiently.
