Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Pharmaceutical Applications
Advanced Metal-Free Synthesis of 3,4,5-Trisubstituted 1,2,4-Triazoles for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective methodologies for constructing nitrogen-rich heterocyclic scaffolds, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in Chinese Patent CN113105402B, which discloses a novel preparation method for 3,4,5-trisubstituted 1,2,4-triazole compounds. These structural motifs are not merely academic curiosities but are foundational components in high-value active pharmaceutical ingredients (APIs). As illustrated in the reference data, prominent drugs such as Maraviroc, Sitagliptin, and Deferasirox rely on similar triazole cores to exert their therapeutic effects. The introduction of a trifluoromethyl group into these heterocycles further amplifies their pharmacological profile by improving lipophilicity and electronegativity. This patent presents a transformative approach that bypasses the traditional reliance on precious metal catalysts, offering a streamlined pathway for the commercial scale-up of complex pharmaceutical intermediates.
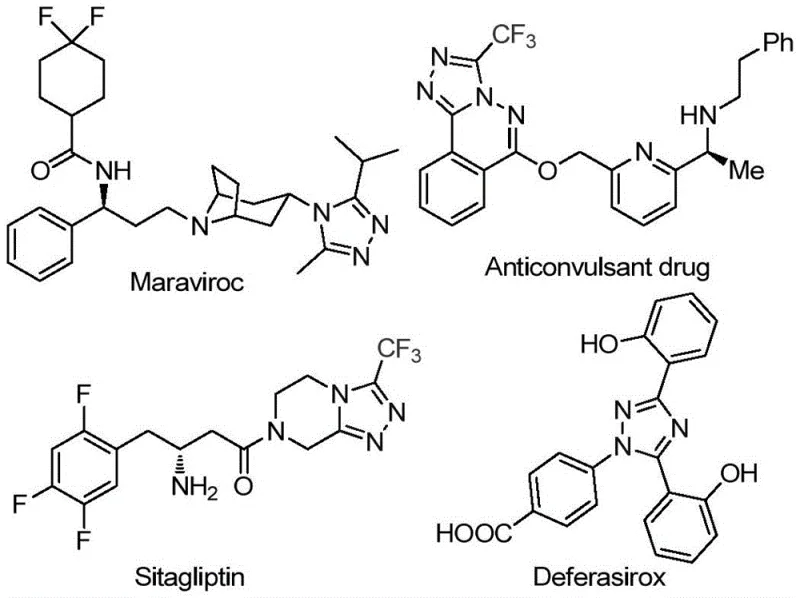
The strategic importance of this technology cannot be overstated for R&D directors and procurement specialists alike. By leveraging a metal-free iodine-promoted system, manufacturers can significantly reduce the impurity profile associated with transition metal leaching, a critical parameter for regulatory compliance in drug substance manufacturing. Furthermore, the operational simplicity of the process—requiring neither rigorous anhydrous conditions nor inert atmospheres—drastically lowers the barrier to entry for production facilities. This report analyzes the technical nuances of patent CN113105402B, evaluating its potential to redefine supply chain strategies for high-purity pharmaceutical intermediates and deliver substantial value through process intensification and cost optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of polysubstituted 1,2,4-triazole rings, especially those bearing both acyl and trifluoromethyl groups, has been fraught with synthetic challenges. Traditional methodologies often depend heavily on transition metal catalysis, utilizing expensive reagents such as palladium, copper, or rhodium complexes. These metals not only inflate the raw material costs but also introduce significant downstream processing burdens. For a reliable pharmaceutical intermediate supplier, the removal of trace heavy metals to meet stringent ICH Q3D guidelines requires additional purification steps, such as scavenging resins or repeated recrystallizations, which inevitably erode overall yield and extend production timelines. Moreover, many classical routes demand strictly anhydrous and oxygen-free environments, necessitating the use of specialized Schlenk lines or gloveboxes, which are difficult to justify economically on a multi-ton scale. The sensitivity of reagents and the complexity of workup procedures in these conventional methods often result in inconsistent batch-to-batch quality and limited substrate tolerance.
The Novel Approach
In stark contrast, the methodology outlined in CN113105402B represents a paradigm shift towards green and sustainable chemistry. By employing elemental iodine as a non-metal promoter in conjunction with dimethyl sulfoxide (DMSO), the inventors have developed a system that is both operationally simple and chemically efficient. This novel approach eliminates the need for toxic heavy metal catalysts entirely, thereby simplifying the purification workflow and reducing the environmental footprint of the manufacturing process. The reaction proceeds smoothly under aerobic conditions without the need for rigorous drying of solvents or reagents, making it exceptionally robust for industrial application. The use of cheap and readily available starting materials, such as aryl ethyl ketones and trifluoroethylimide hydrazides, ensures that the cost reduction in pharmaceutical intermediate manufacturing is realized at the very beginning of the supply chain. This accessibility of raw materials, combined with the mild reaction conditions, positions this technology as a superior alternative for the large-scale production of bioactive triazole derivatives.
Mechanistic Insights into Iodine-Promoted Cyclization
To fully appreciate the utility of this synthesis for R&D teams, one must delve into the mechanistic underpinnings proposed in the patent. The transformation likely initiates with an iodine and DMSO-mediated oxidation of the aryl ethyl ketone. In this step, the methyl group adjacent to the carbonyl is activated, undergoing an iodination followed by a Kornblum-type oxidation to generate an alpha-dicarbonyl intermediate in situ. This highly reactive species then undergoes a condensation reaction with the trifluoroethylimide hydrazide. The nucleophilic attack of the hydrazide nitrogen on the electrophilic carbonyl carbon leads to the formation of a hydrazone intermediate, accompanied by the elimination of water. Subsequently, under the continued influence of iodine and the basic environment provided by pyridine and sodium dihydrogen phosphate, an intramolecular cyclization occurs. This final ring-closing step constructs the 1,2,4-triazole core, aromatizing the system to yield the stable 3,4,5-trisubstituted product. The synergy between the oxidant (iodine/DMSO) and the base is crucial for driving the equilibrium towards the desired heterocycle while minimizing side reactions.
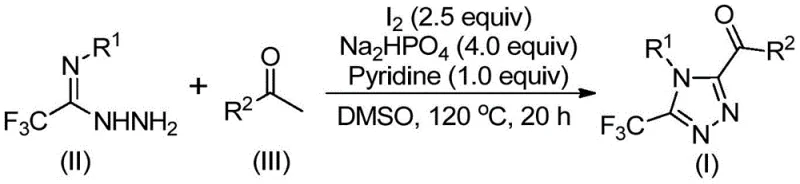
From an impurity control perspective, this mechanism offers distinct advantages. The stepwise nature of the transformation, facilitated by the specific stoichiometry of reagents (e.g., I2 at 2.5 equivalents and Na2HPO4 at 4.0 equivalents), helps to suppress the formation of polymeric byproducts or over-oxidized species that often plague radical-based oxidations. The use of DMSO as both solvent and reactant ensures high solubility of polar intermediates, maintaining a homogeneous reaction phase that promotes consistent kinetics. Furthermore, the tolerance for various substituents on the aryl rings (R1 and R2), including electron-donating groups like methoxy and electron-withdrawing groups like chloro or trifluoromethyl, demonstrates the versatility of this catalytic system. This broad substrate scope allows medicinal chemists to rapidly generate diverse libraries of triazole analogs for structure-activity relationship (SAR) studies without needing to re-optimize reaction conditions for each new derivative, thereby accelerating the drug discovery timeline.
How to Synthesize 3,4,5-Trisubstituted 1,2,4-Triazoles Efficiently
Implementing this synthesis in a practical setting requires careful attention to the sequential addition of reagents and temperature control to maximize yield and purity. The process is designed to be user-friendly, avoiding the pitfalls of air-sensitive chemistry while maintaining high efficiency. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring reproducibility from gram-scale development to pilot plant operations. It is critical to adhere to the specified molar ratios, particularly the excess of aryl ethyl ketone relative to the hydrazide, to drive the reaction to completion and minimize the presence of unreacted hydrazide in the final crude mixture.
- Combine aryl ethyl ketone and elemental iodine in dimethyl sulfoxide (DMSO) and heat to 90-110°C for 4-6 hours to initiate oxidation.
- Add additional iodine, sodium dihydrogen phosphate, pyridine, and trifluoroethylimide hydrazide to the reaction mixture.
- Heat the mixture to 110-130°C for 12-20 hours, then filter and purify via column chromatography to isolate the target triazole compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN113105402B translates directly into tangible operational benefits and risk mitigation. The shift away from precious metal catalysis removes a significant variable cost driver and supply bottleneck. Iodine is a commodity chemical with a stable global supply chain, unlike palladium or rhodium, which are subject to extreme price volatility and geopolitical sourcing risks. This stability allows for more accurate long-term budgeting and reduces the exposure to sudden raw material cost spikes. Additionally, the simplified workup procedure, which involves basic filtration and standard column chromatography rather than complex metal scavenging, reduces the consumption of auxiliary materials and shortens the overall cycle time per batch. This efficiency gain enhances the throughput capacity of existing manufacturing assets without requiring capital-intensive infrastructure upgrades.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and ligands results in a direct decrease in bill of materials (BOM) costs. Furthermore, the avoidance of specialized equipment for inert atmosphere processing lowers both capital expenditure (CAPEX) and operational expenditure (OPEX). The ability to run the reaction in standard vessels under ambient pressure and aerobic conditions simplifies the engineering controls required, leading to substantial cost savings in utility consumption and maintenance. The high atom economy of the iodine-promoted pathway also minimizes waste generation, reducing the costs associated with hazardous waste disposal and environmental compliance.
- Enhanced Supply Chain Reliability: The starting materials, specifically aryl ethyl ketones and trifluoroethylimide hydrazides, are widely available from multiple global suppliers, ensuring a robust and redundant supply chain. This diversity of sourcing options mitigates the risk of single-supplier dependency and ensures continuity of supply even during market disruptions. The robustness of the reaction conditions means that production is less susceptible to delays caused by minor deviations in environmental parameters, such as humidity or oxygen levels, which can often halt sensitive metal-catalyzed processes. This reliability is crucial for maintaining just-in-time delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The use of DMSO, a high-boiling polar aprotic solvent, facilitates heat transfer and mixing on a large scale, making the transition from laboratory to commercial production seamless. The process generates minimal hazardous byproducts, aligning with modern green chemistry principles and easing the burden on wastewater treatment facilities. The absence of heavy metals in the reaction mixture simplifies the regulatory filing process, as there is no need to validate extensive metal clearance protocols. This environmental compatibility not only reduces compliance costs but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-promoted triazole synthesis. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of adopting this route for your specific project needs. Understanding these details is essential for making informed decisions about process integration and resource allocation.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the method described in patent CN113105402B utilizes elemental iodine as a non-metal promoter, completely eliminating the need for costly palladium, copper, or rhodium catalysts and simplifying heavy metal removal processes.
Q: What are the reaction conditions regarding moisture and oxygen sensitivity?
A: Unlike many traditional heterocyclic syntheses, this protocol does not require strict anhydrous or oxygen-free conditions, allowing the reaction to proceed efficiently in standard laboratory or plant equipment without specialized inert atmosphere setups.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method can be easily expanded to the gram level and beyond, utilizing cheap and readily available starting materials like aryl ethyl ketones, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4,5-Trisubstituted 1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in the successful commercialization of new therapeutic agents. Our team of expert process chemists has extensively evaluated the technology disclosed in CN113105402B and is fully prepared to leverage this metal-free iodine-promoted strategy for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to full-scale manufacturing is smooth and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch of high-purity pharmaceutical intermediates we deliver meets the stringent purity specifications required by global regulatory authorities.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to further streamline the synthesis of 3,4,5-trisubstituted 1,2,4-triazoles. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis requirements. Let us help you turn this innovative patent technology into a competitive advantage for your business.
