Advanced One-Step Synthesis of Quinazolinone Derivatives for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to access bioactive heterocyclic scaffolds efficiently. Patent CN111116552B introduces a significant advancement in the synthesis of quinazolinone compounds, a class of molecules renowned for their diverse biological activities including anti-inflammatory, antitumor, and antifungal properties. This intellectual property discloses a novel preparation method that utilizes a direct addition reaction between 4-hydroxyquinazoline compounds and vinyl nitrogen heterocyclic compounds. By leveraging soluble transition metal salts as catalysts, specifically highlighting scandium trifluoromethanesulfonate, the invention achieves a streamlined one-step synthesis that bypasses the cumbersome multi-step procedures typical of prior art. This technological breakthrough not only enriches the structural diversity of available quinazolinone derivatives but also provides a foundational platform for developing next-generation therapeutic agents with unique physiological activities.
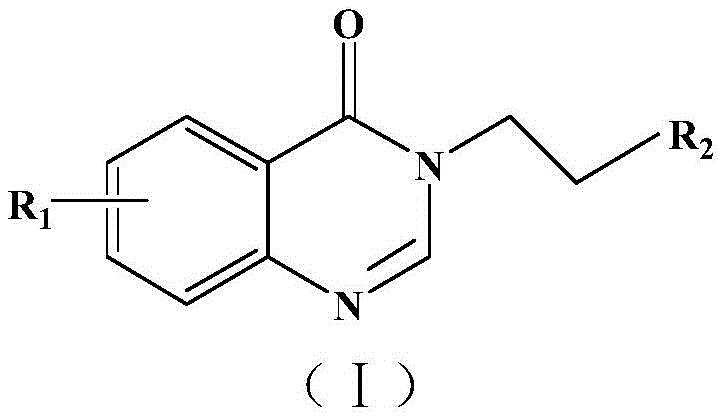
For R&D directors and process chemists, the ability to rapidly generate analogues is crucial for structure-activity relationship (SAR) studies. The disclosed method allows for extensive substitution patterns at the R1 and R2 positions, accommodating halogens, alkoxy groups, nitro groups, and trifluoromethyl groups. This flexibility is paramount for optimizing drug candidates, as minor structural modifications can drastically alter pharmacokinetic profiles. Furthermore, the reaction conditions described are remarkably mild yet effective, operating in common organic solvents such as toluene under inert atmospheres. This combination of structural versatility and operational simplicity positions this patent as a critical asset for any organization aiming to expand its pipeline of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives has been plagued by synthetic inefficiencies that hinder rapid development and commercial viability. Traditional routes often involve multiple discrete steps, requiring the isolation and purification of unstable intermediates which leads to significant material loss and increased waste generation. These conventional methods frequently rely on harsh reaction conditions, including strong acids or bases and elevated temperatures that can degrade sensitive functional groups, thereby limiting the scope of substrates that can be utilized. Additionally, the use of expensive or toxic reagents in older protocols poses significant challenges for environmental compliance and worker safety, necessitating complex waste treatment procedures that drive up operational costs. The cumulative effect of these limitations is a prolonged development timeline and reduced overall yield, making it difficult to secure a reliable supply of complex quinazolinone intermediates for clinical trials or market launch.
The Novel Approach
In stark contrast, the methodology outlined in CN111116552B represents a paradigm shift towards green and efficient chemistry. By employing a direct hydroalkylation strategy, the invention merges the construction of the carbon-nitrogen bond and the functionalization of the quinazolinone core into a single operational step. This atom-economical approach minimizes the generation of by-products and eliminates the need for intermediate workups, drastically simplifying the downstream processing requirements. The use of vinyl nitrogen heterocycles, such as 4-vinylpyridine or 2-vinylquinoline, as coupling partners opens up new chemical space that was previously difficult to access. Moreover, the reaction demonstrates excellent functional group tolerance, allowing for the incorporation of electron-withdrawing and electron-donating groups without compromising yield. This novel approach effectively resolves the bottlenecks of traditional synthesis, offering a scalable pathway that aligns perfectly with the demands of modern industrial manufacturing.
Mechanistic Insights into Scandium-Catalyzed Addition
The core of this innovation lies in the activation of the vinyl heterocycle by a Lewis acid catalyst, specifically scandium trifluoromethanesulfonate (Sc(OTf)3). In this mechanistic pathway, the scandium center coordinates with the nitrogen atom of the vinyl pyridine or quinoline moiety, increasing the electrophilicity of the vinyl double bond. This activation facilitates a nucleophilic attack by the nitrogen atom of the 4-hydroxyquinazoline tautomer. The subsequent proton transfer and aromatization steps lead to the formation of the stable 3-substituted quinazolin-4(3H)-one structure. The choice of Sc(OTf)3 is particularly strategic due to its high water tolerance and strong Lewis acidity, which ensures rapid reaction kinetics even at moderate temperatures. This catalytic cycle is highly efficient, requiring only 10 mol% of the catalyst to drive the reaction to completion, as evidenced by the high yields reported in the experimental examples.
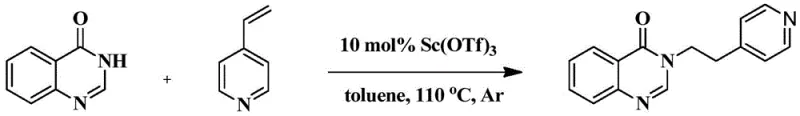
From an impurity control perspective, the mechanism offers distinct advantages over radical-based or high-temperature thermal processes. The mild conditions (60-110°C) prevent the decomposition of the sensitive quinazolinone ring system and minimize polymerization of the vinyl starting materials. The specificity of the Lewis acid interaction ensures high regioselectivity, predominantly yielding the N-alkylated product over potential C-alkylated by-products. This high selectivity simplifies the purification process, often requiring only standard silica gel column chromatography to achieve analytical purity. For quality control teams, this means a cleaner crude reaction profile and a more consistent final product specification, which is essential for meeting the stringent regulatory requirements of the pharmaceutical industry. The ability to tune the electronic properties of the catalyst also allows for further optimization should specific substrate combinations prove challenging.
How to Synthesize 3-(2-(pyridin-4-yl)ethyl)quinazolin-4(3H)-one Efficiently
To implement this synthesis in a laboratory or pilot plant setting, operators should follow a standardized protocol that ensures safety and reproducibility. The process begins by charging a reaction vessel with 4-hydroxyquinazoline, the selected vinyl nitrogen heterocycle, and the scandium catalyst in dry toluene. It is critical to maintain an inert atmosphere, typically using argon or nitrogen, to prevent moisture interference and oxidation of sensitive components. The mixture is then heated to the optimal temperature range of 110°C with vigorous stirring to ensure homogeneous mixing and efficient heat transfer. Reaction progress should be monitored regularly using thin-layer chromatography (TLC) to determine the precise endpoint, preventing over-reaction which could lead to degradation. Upon completion, the reaction is quenched carefully with water, followed by extraction and purification steps to isolate the target compound.
- Combine 4-hydroxyquinazoline, 4-vinylpyridine, and scandium trifluoromethanesulfonate catalyst in toluene under an inert argon atmosphere.
- Heat the reaction mixture to 110°C with stirring and monitor progress via thin-layer chromatography until completion.
- Quench the reaction with water, extract with dichloromethane, wash with brine, dry over sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic benefits that extend beyond simple chemical synthesis. The elimination of multiple synthetic steps directly correlates to a reduction in raw material consumption and labor hours, driving down the overall cost of goods sold (COGS). By utilizing readily available starting materials like 4-hydroxyquinazoline and substituted vinyl pyridines, manufacturers can mitigate supply chain risks associated with exotic or hard-to-source reagents. The robustness of the reaction conditions allows for flexible scheduling and easier integration into existing manufacturing infrastructure without the need for capital-intensive equipment upgrades. Furthermore, the high atom economy and reduced waste generation align with corporate sustainability goals, potentially lowering waste disposal fees and enhancing the company's environmental profile.
- Cost Reduction in Manufacturing: The one-step nature of this process fundamentally alters the cost structure of quinazolinone production. By removing the need for intermediate isolation and purification, facilities can save significantly on solvent usage, filtration media, and energy consumption associated with drying and concentrating multiple batches. The use of catalytic amounts of scandium triflate, which can potentially be recovered or substituted with cheaper alternatives like copper acetate as shown in the patent examples, further optimizes the reagent cost profile. This efficiency allows for a more competitive pricing strategy when supplying high-purity pharmaceutical intermediates to global clients, ensuring margins remain healthy even in a price-sensitive market.
- Enhanced Supply Chain Reliability: Dependence on complex, multi-step syntheses often introduces multiple points of failure in the supply chain, where a delay in one intermediate can halt the entire production line. This streamlined approach consolidates the synthesis into a single unit operation, drastically reducing lead times and improving on-time delivery performance. The broad substrate scope means that if a specific vinyl heterocycle becomes unavailable, alternative analogues can often be synthesized using the same protocol, providing flexibility in raw material sourcing. This resilience is crucial for maintaining continuous supply to downstream API manufacturers who operate on tight Just-In-Time schedules.
- Scalability and Environmental Compliance: Scaling chemical processes from gram to ton scale often reveals hidden complexities, but this method is designed with scalability in mind. The use of toluene, a standard industrial solvent, and moderate temperatures ensures that heat management and mixing remain manageable at larger volumes. The reduction in hazardous waste and the avoidance of heavy metal contaminants (when using Sc or Bi catalysts compared to Pd or Ni) simplify the environmental permitting process. This ease of compliance accelerates the timeline from process development to commercial production, allowing companies to bring new quinazolinone-based drugs to market faster while adhering to strict global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinazolinone synthesis technology. These answers are derived directly from the experimental data and claims within patent CN111116552B, providing a reliable reference for technical teams evaluating this route. Understanding these nuances is essential for making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method utilizes a direct addition reaction between 4-hydroxyquinazoline and vinyl heterocycles, eliminating the need for complex multi-step sequences and intermediate isolation, thereby significantly improving atom economy and reducing production time.
Q: Which catalysts are effective for this transformation according to the patent data?
A: While scandium trifluoromethanesulfonate (Sc(OTf)3) is the preferred catalyst providing optimal yields, the patent also demonstrates the efficacy of bismuth trifluoromethanesulfonate and copper acetate as viable alternatives for cost-sensitive applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process employs common organic solvents like toluene and operates at standard heating temperatures (60-110°C), making it highly adaptable for commercial scale-up without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic routes disclosed in CN111116552B for the production of advanced pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate these laboratory-scale innovations into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of quinazolinone derivative meets the highest industry standards for potency and impurity profiles.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to our deep reservoir of process knowledge and our commitment to quality excellence. Please contact us today to request specific COA data for our available quinazolinone catalog or to discuss route feasibility assessments for your custom synthesis projects, ensuring a seamless path from concept to commercial reality.
