Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinone Derivatives for Commercial Scale-Up
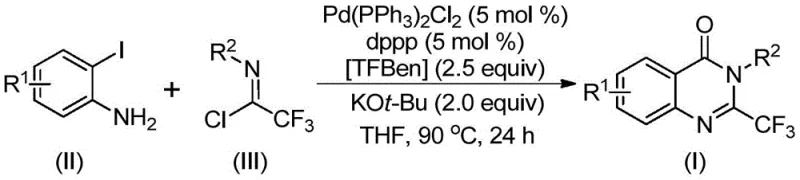
The pharmaceutical industry continuously seeks robust synthetic routes for heterocyclic scaffolds that possess significant biological activity, particularly within the realm of oncology and anti-inflammatory therapeutics. Patent CN112125856A introduces a groundbreaking methodology for the preparation of 2-trifluoromethyl substituted quinazolinone derivatives, a class of compounds renowned for their enhanced metabolic stability and lipophilicity due to the presence of the fluorine atom. This innovation addresses critical bottlenecks in existing synthetic pathways by employing a transition metal palladium-catalyzed carbonylation tandem reaction. By utilizing 1,3,5-tricarboxylate phenol ester (TFBen) as a safe, solid carbon monoxide substitute, this process circumvents the severe safety hazards associated with handling toxic carbon monoxide gas, thereby offering a safer and more operationally convenient alternative for industrial manufacturing. The method boasts exceptional substrate applicability, allowing for the efficient synthesis of diverse derivatives through simple substrate design, which is paramount for medicinal chemistry campaigns aiming to optimize lead compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl substituted quinazolinone derivatives has been plagued by significant operational and safety challenges that hinder large-scale production. Traditional methodologies often rely on the cyclization of anthranilamides with ethyl trifluoroacetate or trifluoroacetic anhydride, processes that frequently require harsh reaction conditions and expensive, pre-activated substrates. Furthermore, alternative routes involving T3P-promoted tandem reactions or the use of unstable trifluoroacetamides often suffer from narrow substrate scopes and inconsistent yields, making them unreliable for commercial supply chains. The reliance on gaseous carbon monoxide in many carbonylation protocols presents a formidable safety barrier, necessitating specialized high-pressure equipment and rigorous safety protocols that drastically increase capital expenditure and operational complexity. These limitations collectively result in higher production costs, extended lead times, and increased environmental burdens due to the generation of difficult-to-treat waste streams, rendering many conventional methods unsuitable for modern, green chemistry standards.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent leverages a sophisticated palladium-catalyzed system that transforms simple, commercially available starting materials into high-value intermediates with remarkable efficiency. By reacting o-iodoaniline derivatives with trifluoroacetimidoyl chlorides in the presence of a palladium catalyst and a phosphine ligand, the process achieves a seamless carbonylation and cyclization sequence. The cornerstone of this innovation is the use of TFBen as a solid carbon monoxide surrogate, which decomposes under heating conditions to release CO in situ, thereby eliminating the need for external gas cylinders and high-pressure reactors. This strategy not only enhances operator safety but also simplifies the reactor setup, allowing the reaction to proceed smoothly at atmospheric pressure in standard organic solvents like tetrahydrofuran. The method demonstrates broad functional group tolerance, accommodating various substituents such as halogens, alkyl groups, and nitro groups, which ensures that a wide library of analogues can be generated rapidly for structure-activity relationship studies without the need for extensive process re-optimization.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Tandem Reaction
The mechanistic pathway of this transformation involves a complex yet elegant series of organometallic steps that ensure high selectivity and yield. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond coupling between the o-iodoaniline and the trifluoroacetimidoyl chloride, generating a trifluoroacetamidine intermediate. Subsequently, the palladium catalyst undergoes oxidative addition into the carbon-iodine bond of the aromatic ring, forming a reactive divalent palladium species. As the temperature rises to 90°C, the TFBen additive decomposes to release carbon monoxide, which then inserts into the carbon-palladium bond to form a crucial acyl-palladium intermediate. This insertion step is pivotal, as it introduces the carbonyl functionality required for the quinazolinone core. Following this, the base facilitates the formation of a palladium-nitrogen bond, leading to a seven-membered ring palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl substituted quinazolinone derivative and regenerates the active palladium catalyst for the next turnover. This well-defined catalytic cycle minimizes side reactions and ensures that the trifluoromethyl group is retained intact throughout the process.
From an impurity control perspective, the mild reaction conditions and the specific choice of ligands play a vital role in maintaining product purity. The use of dppp (1,3-bis(diphenylphosphine)propane) as a bidentate ligand stabilizes the palladium center, preventing the formation of palladium black and other inactive species that could catalyze decomposition pathways. Furthermore, the in situ generation of carbon monoxide from TFBen ensures a steady, controlled concentration of CO, preventing over-carbonylation or the formation of urea byproducts that are common when using excess gaseous CO. The compatibility of the system with various electron-withdrawing and electron-donating groups on the aromatic rings means that electronic effects do not significantly derail the catalytic cycle, resulting in a consistent impurity profile across different substrates. This predictability is essential for downstream purification, as it allows for the development of robust crystallization or chromatography protocols that can consistently deliver high-purity material suitable for clinical trials.
How to Synthesize 2-Trifluoromethyl Quinazolinone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing simplicity and reproducibility. The process begins by charging a reaction vessel with the palladium catalyst, ligand, base, TFBen, and the two primary organic substrates in an aprotic solvent. The mixture is then heated to a moderate temperature, allowing the tandem reaction to proceed to completion over a defined period. Post-reaction workup involves straightforward filtration and silica gel treatment, followed by standard purification techniques. For detailed operational parameters, stoichiometry, and specific troubleshooting tips tailored to your facility's equipment, please refer to the standardized synthesis guide below.
- Combine palladium catalyst, dppp ligand, potassium tert-butoxide, TFBen, trifluoroacetimidoyl chloride, and o-iodoaniline in an organic solvent like THF.
- Heat the reaction mixture to 90°C and stir for 16 to 30 hours to allow the carbonylation and cyclization to proceed.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative advantages in terms of cost structure and logistical reliability. The shift away from hazardous gaseous reagents and expensive activation agents fundamentally alters the cost basis of producing these valuable intermediates. By utilizing cheap and readily available starting materials such as o-iodoanilines and trifluoroacetimidoyl chlorides, the raw material costs are significantly minimized compared to traditional routes that require pre-functionalized or protected precursors. Moreover, the elimination of high-pressure carbon monoxide infrastructure reduces the capital investment required for plant modification, allowing for faster deployment of production lines. The robustness of the reaction conditions also implies a lower risk of batch failures, ensuring a more consistent supply of material to meet downstream manufacturing demands without unexpected interruptions.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous reagents with cost-effective alternatives. The use of TFBen as a solid CO source eliminates the need for specialized gas handling equipment and the associated safety compliance costs, leading to substantial operational savings. Additionally, the high catalytic efficiency allows for lower catalyst loading, reducing the consumption of precious palladium metals. The simplified workup procedure, which avoids complex quenching steps often required for reactive acylating agents, further reduces labor and utility costs. Collectively, these factors contribute to a leaner manufacturing process that delivers a lower cost of goods sold (COGS), enhancing the overall profitability of the final pharmaceutical product.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of commodity chemicals as starting materials. O-iodoanilines and trifluoroacetimidoyl chlorides are widely produced and available from multiple global suppliers, mitigating the risk of single-source dependency. The stability of the solid CO surrogate, TFBen, simplifies storage and transportation logistics, as it does not require the stringent regulatory controls associated with compressed toxic gases. This ease of handling ensures that raw material inventory can be maintained safely on-site, buffering against potential market fluctuations or delivery delays. Consequently, manufacturers can maintain higher service levels and shorter lead times for their customers, fostering stronger long-term partnerships.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the homogeneous nature of the reaction and the absence of gas-liquid mass transfer limitations typical of gaseous CO reactions. The reaction can be performed in standard stirred-tank reactors without the need for autoclaves, simplifying technology transfer from R&D to production. From an environmental standpoint, the process aligns with green chemistry principles by avoiding toxic gas emissions and generating less hazardous waste. The use of THF as a solvent, which is easily recoverable and recyclable, further minimizes the environmental footprint. This compliance with increasingly strict environmental regulations ensures long-term operational viability and reduces the risk of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process development teams. Understanding these nuances is critical for evaluating the feasibility of integrating this technology into existing production workflows.
Q: How does this method improve safety compared to traditional quinazolinone synthesis?
A: This method utilizes 1,3,5-tricarboxylate phenol ester (TFBen) as a solid carbon monoxide surrogate, completely eliminating the need for handling toxic and hazardous carbon monoxide gas cylinders typically required in carbonylation reactions.
Q: What is the substrate scope for this palladium-catalyzed reaction?
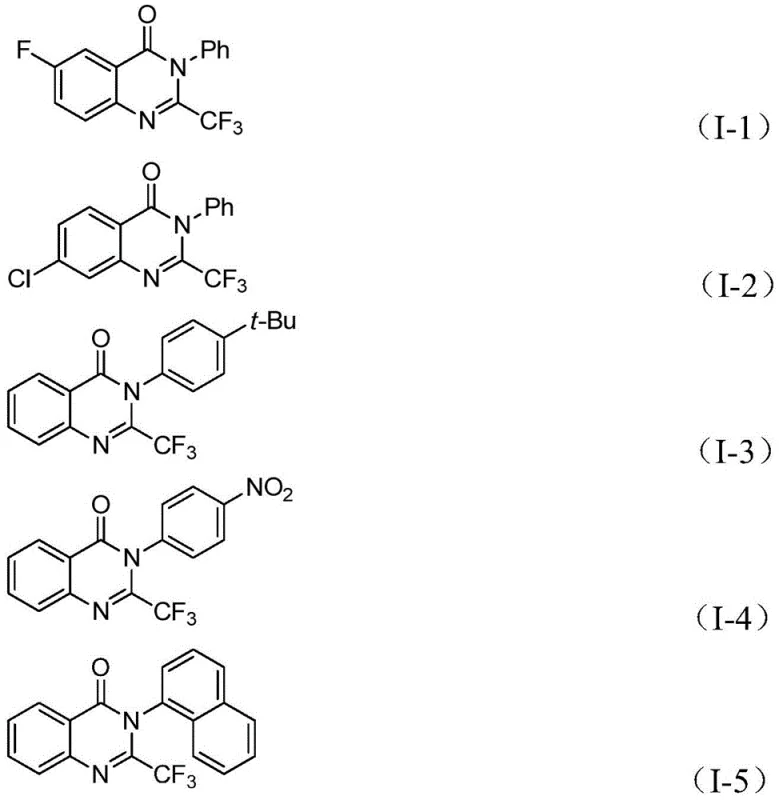
A: The process demonstrates excellent compatibility with various substituents on both the o-iodoaniline and the imidoyl chloride components, including halogens (F, Cl, Br), alkyl groups, and nitro groups, allowing for diverse derivative synthesis.
Q: What are the typical reaction conditions and catalyst loading?
A: The reaction typically proceeds at 90°C in THF solvent for 16-30 hours, utilizing a low catalyst loading of 5 mol% Pd(PPh3)2Cl2 and 5 mol% dppp ligand, ensuring cost-effectiveness for large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient and safe synthetic routes for complex pharmaceutical intermediates. Our team of expert chemists has thoroughly analyzed the potential of this palladium-catalyzed carbonylation technology and is fully prepared to support its translation from benchtop discovery to commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition to larger batches is seamless and compliant with international quality standards. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-trifluoromethyl quinazolinone derivative delivered meets the exacting requirements of global drug developers.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data for our reference standards and to discuss route feasibility assessments for your target molecules. Let us help you optimize your supply chain and accelerate your drug development pipeline with our reliable, high-quality intermediate solutions.
