Advanced Manufacturing of High-Purity Trifluoromethyl Triazoles via Iodine Catalysis for Pharmaceutical Supply Chains
Patent CN110467579B introduces a groundbreaking methodology for preparing 5-trifluoromethyl substituted 1,2,4-triazole compounds representing a significant advancement in heterocyclic chemistry with direct implications for pharmaceutical development pipelines globally This innovative approach leverages readily accessible starting materials while avoiding stringent reaction conditions that have historically constrained industrial adoption of similar processes The method eliminates requirements for anhydrous and anaerobic environments completely bypassing toxic heavy metal catalysts that complicate downstream purification and increase production costs By utilizing elemental iodine as a benign catalytic promoter alongside simple organic solvents like dichloroethane the process achieves high yields across diverse substrate combinations without specialized equipment or hazardous reagents This patent establishes a new paradigm for synthesizing fluorinated triazole derivatives critical as building blocks in modern drug discovery where enhanced metabolic stability and bioavailability are essential characteristics The demonstrated scalability from laboratory demonstrations to commercial production levels further underscores its potential to transform supply chains for high-value pharmaceutical intermediates worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethylated heterocycles typically rely on expensive trifluoromethylation reagents such as Togni's reagent or Umemoto's compounds which require specialized handling under strictly controlled anhydrous and anaerobic conditions due to their extreme moisture sensitivity These methods often employ transition metal catalysts including palladium or copper complexes that necessitate elaborate removal procedures because regulatory agencies impose stringent limits on metal residues in pharmaceutical products The high cost of fluorinating agents combined with limited functional group tolerance significantly restricts substrate scope while increasing production expenses substantially Furthermore many existing protocols generate substantial waste streams requiring costly treatment processes that conflict with modern green chemistry principles The operational complexity safety hazards associated with handling pyrophoric reagents or cryogenic temperatures also contribute to extended production timelines inconsistent batch quality and higher failure rates during scale-up efforts These cumulative challenges have historically prevented widespread industrial implementation despite growing demand for fluorinated triazole compounds in therapeutic applications.
The Novel Approach
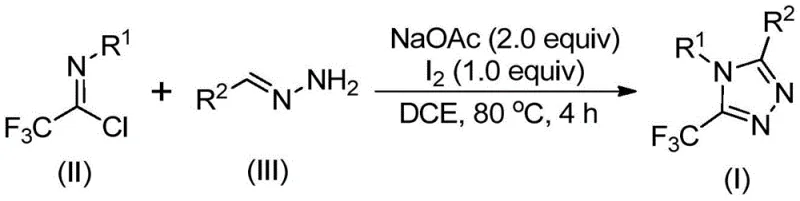
In contrast the patented methodology employs a streamlined one-pot reaction sequence using inexpensive sodium acetate as a base promoter and elemental iodine as a non-toxic catalyst operating effectively under ambient atmospheric conditions without requiring specialized equipment This approach utilizes readily available trifluoroethylimidoyl chloride and hydrazone precursors synthesized from common aldehydes and aromatic amines through straightforward procedures By eliminating heavy metal catalysts and anhydrous environments this method significantly reduces both capital expenditure and operational complexity while maintaining excellent functional group compatibility across diverse substrates The reaction proceeds efficiently in standard organic solvents like dichloroethane at moderate temperatures around eighty degrees Celsius with minimal byproduct formation simplifying purification through conventional column chromatography techniques This innovative strategy achieves comparable or superior yields to existing methods while demonstrating exceptional scalability potential from gram-scale laboratory demonstrations to multi-ton industrial production volumes without requiring extensive process revalidation.
Mechanistic Insights into Iodine-Catalyzed Triazole Formation
The reaction mechanism begins with base-promoted intermolecular carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and hydrazone components generating a key trifluoroacetamidine intermediate through nucleophilic substitution This intermediate subsequently undergoes spontaneous isomerization followed by iodine-mediated oxidative transformation creating a critical iodinated species through electrophilic activation The iodinated intermediate then participates in an intramolecular cyclization event where nitrogen nucleophiles attack electrophilic carbon centers forming triazole ring systems through concerted substitution processes This ring closure is immediately followed by aromatization through proton elimination establishing fully conjugated heterocyclic structures characteristic of final products The mild reaction conditions facilitate excellent control over regioselectivity while minimizing unwanted side reactions that could compromise product purity or yield This mechanistic pathway demonstrates how iodine catalysis enables efficient transformation without requiring harsh reagents or extreme temperatures typically limiting industrial applicability.
The process inherently minimizes impurity formation through its well-defined reaction pathway avoiding common side reactions such as over-halogenation or decomposition pathways observed in alternative methodologies Elemental iodine functions as a catalytic mediator creating a self-limiting system where oxidizing agents operate through regenerative cycles preventing excessive iodination while maintaining high conversion rates across diverse substrate combinations The absence of transition metals eliminates potential sources of metal-induced impurities requiring additional purification steps in pharmaceutical manufacturing settings Furthermore moderate temperature profiles prevent thermal degradation of sensitive functional groups while ensuring complete conversion within practical reaction timescales thus supporting consistent production quality essential for regulatory compliance.
How to Synthesize Trifluoromethyl Triazoles Efficiently
This patented methodology represents a significant advancement in heterocyclic synthesis by providing a robust pathway to access diverse trifluoromethylated triazole derivatives through rational substrate design optimization The process demonstrates exceptional versatility across various functional groups while maintaining high yields under operationally simple conditions readily adaptable to industrial manufacturing environments Detailed standardized synthesis procedures have been developed based on extensive experimental validation across multiple substrate combinations as documented in patent literature The following section provides essential guidance for implementing this innovative approach in laboratory or production settings.
- Combine sodium acetate (2.0 equivalents), trifluoroethylimidoyl chloride (II), and hydrazone (III) in dichloroethane solvent under standard atmospheric conditions.
- Heat the reaction mixture to precisely 80°C and maintain this temperature for four hours to facilitate carbon-nitrogen bond formation.
- Add elemental iodine (1.0 equivalent) and continue heating at constant temperature for one additional hour before initiating purification.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis method directly addresses critical pain points in pharmaceutical supply chains by offering more sustainable economically viable pathways to high-value triazole intermediates essential components in numerous therapeutic agents Elimination of expensive catalysts and specialized reaction conditions translates into substantial operational efficiencies enhancing both cost competitiveness and supply reliability for procurement teams managing complex global sourcing networks Leveraging readily available starting materials simplified processing requirements enables manufacturers greater flexibility meeting fluctuating demand patterns while maintaining stringent quality standards required by regulatory authorities worldwide This approach creates significant strategic advantages through reduced vulnerability to supply chain disruptions common in traditional manufacturing routes.
- Cost Reduction in Manufacturing: Complete avoidance of transition metal catalysts removes significant expenses associated with precious metal procurement complex post-reaction purification processes required to meet regulatory limits on metal residues Elemental iodine functions as an inexpensive catalytic mediator eliminating costly waste treatment requirements while sodium acetate serves as an economical base promoter substantially lowering raw material costs compared to conventional fluorination methodologies.
- Enhanced Supply Chain Reliability: Reliance on commercially available starting materials with broad supplier networks ensures consistent access to critical precursors without vulnerability to single-source dependencies Simplified reaction protocol operating under standard atmospheric conditions reduces sensitivity to environmental factors causing production delays enabling faster batch turnaround times through streamlined processing workflows thus improving overall supply chain resilience.
- Scalability and Environmental Compliance: Demonstrated scalability from laboratory to commercial production levels with minimal process modification requirements provides immediate pathway to large-scale manufacturing without extensive revalidation efforts Reduced environmental footprint resulting from lower energy consumption minimized hazardous waste generation aligns with evolving regulatory frameworks supporting corporate sustainability initiatives across pharmaceutical value chains.
Frequently Asked Questions (FAQ)
The following questions address common concerns regarding implementation of this patented technology based on detailed analysis of technical specifications operational parameters documented in CN110467579B These insights reflect practical considerations derived from extensive experimental validation under diverse manufacturing scenarios providing actionable information for procurement supply chain decision-makers evaluating this innovative approach.
Q: How does this method eliminate expensive heavy metal catalysts?
A: The process utilizes elemental iodine as a non-toxic catalytic promoter instead of transition metals like palladium or copper complexes which require costly removal procedures due to regulatory limits on metal residues.
Q: What advantages does this approach offer regarding reaction conditions?
A: Unlike conventional methods requiring strictly anhydrous and anaerobic environments this synthesis operates effectively under standard atmospheric conditions using common organic solvents without specialized equipment.
Q: How does this process support scalable manufacturing?
A: The simplified procedure with readily available starting materials straightforward purification through column chromatography and demonstrated scalability from gram-scale to commercial volumes enables seamless industrial implementation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Triazole Supplier
Our company possesses extensive experience scaling diverse pathways from one hundred kilograms to one hundred metric tons annual commercial production while maintaining stringent purity specifications through rigorous QC labs equipped with state-of-the-art analytical instrumentation As a leading CDMO specialist in complex heterocyclic synthesis we have successfully implemented this patented methodology across multiple client projects requiring high-purity pharmaceutical intermediates with demanding quality requirements Our integrated manufacturing platform combines deep technical expertise with flexible production capabilities delivering consistent quality at commercial scale while optimizing cost structures through continuous process improvements tailored to specific client needs.
We invite you to request a Customized Cost-Saving Analysis from our technical procurement team to evaluate how this innovative synthesis approach can enhance your specific supply chain requirements Please contact us directly to obtain detailed COA data route feasibility assessments tailored to your manufacturing needs.
