Revolutionizing Amide Bond Formation: Scalable NHC-Catalyzed Routes for High-Purity Intermediates
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct the ubiquitous amide bond, a structural motif present in approximately 25% of all drug molecules. Patent CN108558692B introduces a transformative preparation method for amide compounds that leverages N-heterocyclic carbene (NHC) organocatalysis to overcome the limitations of traditional synthesis. This technology enables the direct conversion of organic acid esters and organic amines into high-purity amides under remarkably mild conditions, typically requiring only 0.1 to 1.5 hours at temperatures ranging from 0 to 40°C. By eliminating the need for harsh activating agents or toxic transition metals, this innovation offers a robust solution for the commercial scale-up of complex pharmaceutical intermediates, ensuring both environmental compliance and operational efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amides has relied heavily on two primary strategies, both of which present significant bottlenecks for modern manufacturing. The first conventional approach involves activating carboxylic acids using reagents such as triphenylphosphine/iodine (Ph3P/I2) or HATU to form active esters prior to amidation. While effective, these methods are plagued by prolonged reaction times, incomplete conversions, and the generation of stoichiometric amounts of phosphine oxide or urea byproducts that are notoriously difficult to separate from the final product. The second strategy employs transition metal-catalyzed oxidative aminolysis of aldehydes; however, this route suffers from narrow substrate adaptability and often requires expensive metal catalysts that necessitate rigorous removal steps to meet stringent purity specifications for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes an N-heterocyclic carbene catalyst to activate organic acid esters directly, facilitating a rapid nucleophilic substitution with organic amines. This organocatalytic cycle bypasses the need for pre-activation of carboxylic acids or the use of aldehyde precursors, thereby streamlining the synthetic sequence. The reaction proceeds efficiently in common organic solvents like tetrahydrofuran (THF) with simple bases such as DBU or potassium carbonate. This shift not only drastically simplifies the workup procedure—often requiring only extraction and standard chromatography—but also ensures high total yields, making it an ideal candidate for cost reduction in amide manufacturing where raw material efficiency and waste minimization are critical.
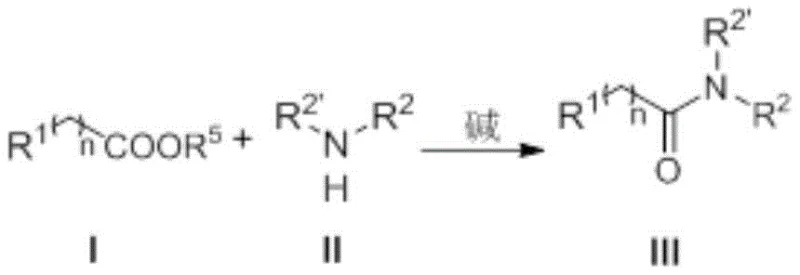
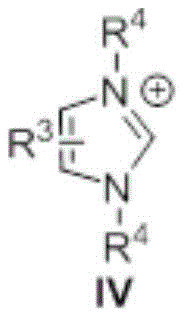
Mechanistic Insights into NHC-Catalyzed Ester Aminolysis
The core of this technological breakthrough lies in the unique ability of the N-heterocyclic carbene to act as a nucleophilic catalyst. As illustrated in the catalyst structure below, the carbene species interacts with the carbonyl carbon of the organic acid ester to form a highly reactive acyl azolium intermediate. This activated species is significantly more electrophilic than the parent ester, allowing it to undergo rapid nucleophilic attack by the amine even under mild thermal conditions. This mechanism effectively lowers the activation energy barrier for the amidation process, explaining the observed reaction times of less than 1.5 hours compared to the multi-hour durations typical of traditional coupling protocols.
From an impurity control perspective, the mildness of the NHC-catalyzed pathway is paramount for R&D teams focused on quality. Traditional activation methods often generate acidic or basic byproducts that can degrade sensitive functional groups on the substrate, leading to complex impurity profiles. By operating at near-neutral pH and low temperatures (0-40°C), this new method preserves the integrity of delicate moieties such as nitro groups or methoxy substituents, as evidenced by the successful synthesis of various substituted benzamides in the patent examples. This inherent selectivity reduces the burden on downstream purification, ensuring that the final high-purity amide compounds meet the rigorous standards required for subsequent drug substance synthesis without extensive recrystallization cycles.
How to Synthesize Amide Compounds Efficiently
The implementation of this NHC-catalyzed protocol is straightforward and aligns well with existing infrastructure in standard chemical processing facilities. The process begins with the preparation of the catalytic system under an inert atmosphere, followed by the sequential addition of substrates. The simplicity of the reagent profile—avoiding moisture-sensitive or pyrophoric materials—enhances operational safety. For detailed procedural specifics regarding molar ratios, solvent choices, and workup parameters, please refer to the standardized synthesis guide provided below, which outlines the exact steps validated in the patent examples to achieve yields exceeding 90%.
- Prepare the reaction system under an inert atmosphere by dissolving the NHC precursor and base in an organic solvent like THF.
- Sequentially add the organic acid ester and organic amine to the stirred mixture, maintaining temperatures between 0-40°C.
- Upon completion, quench with water, extract with organic solvents, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this NHC-catalyzed amidation technology represents a strategic opportunity to optimize the supply of key building blocks. The elimination of expensive coupling reagents like HATU or precious metal catalysts directly translates to a reduction in the bill of materials. Furthermore, the use of stable organic acid esters and amines as starting materials ensures a reliable supply chain, as these commodities are widely available from multiple global vendors, mitigating the risk of single-source dependency that often plagues specialized reagent procurement.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the replacement of stoichiometric activating agents with a catalytic amount of inexpensive organic salts. Traditional methods often require greater than 1:1 equivalents of costly activators, whereas this method uses only 0.1 to 0.2 equivalents of the NHC precursor. Additionally, the simplified purification process reduces solvent consumption and labor hours associated with complex workups, leading to substantial overall cost savings in the production of bulk pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By utilizing robust and readily available starting materials, manufacturers can significantly reduce lead times for high-purity pharmaceutical intermediates. The reaction's tolerance to a wide range of substrates means that a single standardized protocol can be applied to synthesize a diverse library of amide derivatives, simplifying inventory management and allowing for flexible response to changing market demands without the need for specialized equipment or rare catalysts.
- Scalability and Environmental Compliance: The mild reaction conditions (0-40°C) and the absence of heavy metals make this process inherently safer and easier to scale from kilogram to tonne quantities. The reduced generation of hazardous waste, particularly phosphine oxides or metal residues, aligns with increasingly strict environmental regulations. This green chemistry profile not only lowers waste disposal costs but also enhances the sustainability credentials of the final product, a growing requirement for major pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NHC-catalyzed amidation technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the method's versatility and operational parameters for potential partners evaluating this route for their supply chains.
Q: What are the primary advantages of NHC catalysis over traditional coupling reagents?
A: Unlike traditional methods using reagents like HATU or Ph3P/I2 which often suffer from long reaction times and difficult byproduct separation, NHC catalysis operates under mild conditions with significantly shorter reaction times and higher yields.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process utilizes readily available esters and amines, avoids expensive transition metals, and operates at near-room temperature, making it highly scalable and economically viable for commercial manufacturing.
Q: What types of substrates are compatible with this synthesis route?
A: The method demonstrates broad substrate adaptability, successfully converting various organic acid esters (including those with electron-withdrawing or donating groups) and organic amines into corresponding amides with high efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient amide bond formation in the development of next-generation therapeutics. Our technical team has extensively evaluated the NHC-catalyzed pathway described in CN108558692B and confirmed its potential for delivering high-quality intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. Our stringent purity specifications and rigorous QC labs guarantee that every batch of amide compound meets the exacting standards required for API synthesis.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your target molecule. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this innovative catalytic method can enhance your supply chain efficiency and reduce overall manufacturing costs.
