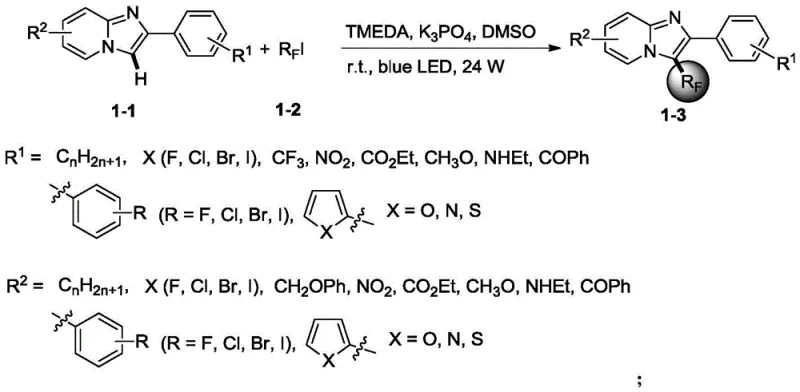
Scalable Photocatalytic Synthesis of 3-Polyfluoroalkyl Imidazo Pyridines for Commercial Drug Development
The pharmaceutical industry continuously seeks efficient pathways to incorporate fluorine atoms into heterocyclic scaffolds, as these modifications significantly enhance metabolic stability and membrane permeability. Patent CN112851670A discloses a groundbreaking simple synthesis method for 3-polyfluoroalkyl substituted imidazo[1,2-a]pyridine derivatives, addressing a critical gap in current drug synthesis technologies. This innovation leverages a visible-light-induced radical mechanism to directly functionalize the C-3 position of the imidazo ring, bypassing the need for pre-functionalized starting materials. The process operates under remarkably mild conditions, utilizing a nitrogen atmosphere and standard laboratory lighting to drive the reaction forward with high efficiency. By enabling the direct coupling of readily available polyfluoroalkyl iodides with diverse imidazo[1,2-a]pyridine cores, this technology offers a versatile platform for generating high-value pharmaceutical intermediates. The structural versatility is evident in the broad scope of substituents tolerated at the R1 and R2 positions, allowing medicinal chemists to rapidly explore structure-activity relationships.
![General structure of imidazo[1,2-a]pyridine derivatives showing variable R1 and R2 substituents](/insights/img/fluoroalkyl-imidazo-pyridine-synthesis-pharma-supplier-20260304233733-01.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for introducing polyfluoroalkyl groups onto nitrogen-containing heterocycles often suffer from significant drawbacks that hinder their application in large-scale manufacturing. Conventional cross-coupling reactions typically require expensive transition metal catalysts, such as palladium or copper, which not only increase raw material costs but also introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient. Furthermore, many existing methods necessitate harsh reaction conditions, including high temperatures and strong bases, which can lead to the decomposition of sensitive functional groups or the formation of complex impurity profiles. The requirement for pre-halogenated substrates adds additional synthetic steps, thereby reducing the overall atom economy and increasing waste generation. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks for procurement teams aiming to secure reliable supplies of complex fluorinated intermediates. Additionally, the use of toxic solvents and difficult-to-remove catalysts complicates the downstream purification process, posing challenges for environmental compliance and operational safety.
The Novel Approach
In stark contrast, the novel photocatalytic approach described in the patent data offers a sustainable and economically viable alternative that directly addresses these historical pain points. By utilizing a simple mixture of TMEDA and potassium phosphate under blue LED irradiation, the method induces the formation of polyfluoroalkyl radicals without the need for exogenous photocatalysts or precious metals. This catalyst-free or organocatalytic nature drastically simplifies the reaction setup and eliminates the costly step of metal scavenging during workup. The reaction proceeds smoothly at temperatures between 20-45°C, preserving the integrity of sensitive moieties such as esters, ethers, and halides that might otherwise degrade under thermal stress. The use of dimethyl sulfoxide (DMSO) as a solvent provides excellent solubility for both organic substrates and inorganic additives, ensuring homogeneous reaction kinetics. This streamlined protocol not only reduces the environmental footprint by minimizing waste but also enhances the overall process safety, making it an attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Photocatalytic Polyfluoroalkylation
The core of this technological breakthrough lies in the unique ability of the amine inducer, specifically TMEDA, to form an electron donor-acceptor (EDA) complex with the polyfluoroalkyl iodide under visible light irradiation. Upon exposure to blue LED light (24-70W), this complex undergoes a single-electron transfer (SET) process, resulting in the homolytic cleavage of the carbon-iodine bond and the generation of a highly reactive polyfluoroalkyl radical. This radical species then selectively attacks the electron-rich C-3 position of the imidazo[1,2-a]pyridine ring, forming a transient radical intermediate. The subsequent oxidation and deprotonation steps, facilitated by the phosphate additive and the ambient oxygen or oxidant present in the system, restore aromaticity and yield the final 3-polyfluoroalkyl substituted product. This mechanistic pathway avoids the high energy barriers associated with thermal radical initiation, allowing the reaction to proceed efficiently at room temperature. The precise control over radical generation ensures high regioselectivity, minimizing the formation of undesired isomers or over-alkylated byproducts.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in maintaining a clean crude profile. Unlike thermal methods that often promote side reactions such as polymerization or decomposition of the fluorinated chain, this photochemical process preserves the structural fidelity of the polyfluoroalkyl group. The absence of transition metals eliminates a major source of inorganic impurities, simplifying the analytical burden on quality control laboratories. Furthermore, the tolerance for various functional groups on the aromatic rings (R1 and R2) means that complex molecules can be synthesized in fewer steps, reducing the cumulative impurity load from previous synthetic stages. The use of potassium phosphate as a mild base prevents the hydrolysis of ester groups, which is a common issue when using stronger alkoxides. This high level of chemoselectivity ensures that the final product meets stringent purity specifications required for clinical applications, thereby reducing the risk of batch rejection and supply chain disruptions.
How to Synthesize 3-Polyfluoroalkyl Imidazo Pyridine Efficiently
To implement this synthesis effectively, operators must adhere to strict protocols regarding atmospheric control and light source calibration to ensure reproducibility. The process begins with the dissolution of the imidazo[1,2-a]pyridine derivative and the polyfluoroalkyl iodide in anhydrous DMSO under a rigorous nitrogen atmosphere to prevent premature quenching of radicals by oxygen. Following the addition of the TMEDA inducer and potassium phosphate additive, the reaction vessel is positioned under a 24W blue LED lamp, maintaining a temperature between 20-45°C for a duration of 24 to 96 hours depending on the specific substrate reactivity. Detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are essential for maintaining consistent yields and purity profiles. For a comprehensive breakdown of the exact molar ratios, workup procedures, and purification techniques, please refer to the technical guide below.
- Dissolve the imidazo[1,2-a]pyridine derivative and polyfluoroalkyl iodide in anhydrous DMSO under a nitrogen atmosphere.
- Add TMEDA as an inducer and potassium phosphate as an additive to the reaction mixture.
- Irradiate the solution with a 24W blue LED lamp at 20-45°C for 24-96 hours, followed by extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The elimination of expensive noble metal catalysts represents a direct reduction in raw material costs, while the simplified workup procedure lowers the consumption of auxiliary chemicals and solvents. The mild reaction conditions reduce energy consumption associated with heating and cooling, contributing to a lower overall carbon footprint and aligning with corporate sustainability goals. Moreover, the robustness of the reaction across a wide range of substrates ensures a stable supply of diverse intermediates without the need for frequent process re-optimization. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts from the process flow eliminates the need for specialized metal scavenging resins and extensive filtration steps, which are traditionally cost-intensive. By relying on inexpensive organic amines and inorganic salts as promoters, the bill of materials is significantly optimized, leading to substantial cost savings per kilogram of produced intermediate. Additionally, the high yields reported in the patent examples minimize the loss of valuable starting materials, further enhancing the economic efficiency of the process. The simplified purification requirements also reduce the man-hours and equipment time needed for downstream processing, driving down the overall cost of goods sold.
- Enhanced Supply Chain Reliability: The starting materials, including various imidazo[1,2-a]pyridine derivatives and polyfluoroalkyl iodides, are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The stability of the reagents under ambient storage conditions simplifies inventory management and reduces the need for specialized cold-chain logistics. The scalability of the LED-based reactor setup allows for flexible production capacity, enabling manufacturers to quickly ramp up output in response to surges in market demand. This agility ensures that procurement teams can secure reliable supplies of high-purity pharmaceutical intermediates even during periods of global supply chain volatility.
- Scalability and Environmental Compliance: The use of visible light as the energy source is inherently scalable, as LED arrays can be easily configured to illuminate larger reaction vessels without the heat transfer limitations associated with traditional thermal heating. The process generates minimal hazardous waste, as the primary byproducts are inorganic salts that can be easily disposed of or treated in standard wastewater facilities. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the environmental compliance burden on manufacturing sites. The ability to run the reaction at near-room temperature also enhances operational safety, lowering the risk of thermal runaway incidents and ensuring a safer working environment for plant personnel.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method in an industrial setting. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide accurate guidance for decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines. We encourage technical teams to review these points thoroughly to assess the potential impact on their specific project requirements.
Q: What are the key advantages of this photocatalytic method over traditional fluorination?
A: This method utilizes visible light and mild temperatures (20-45°C) to induce radical formation, eliminating the need for harsh thermal conditions or expensive transition metal catalysts often required in conventional cross-coupling reactions.
Q: What is the typical yield range for these fluorinated imidazo pyridine derivatives?
A: According to the experimental data in patent CN112851670A, the process consistently achieves high yields ranging from 45% to 92% across a diverse library of substrates, demonstrating robust efficiency.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the use of standard LED lighting and common solvents like DMSO, combined with a simple workup procedure involving extraction and chromatography, makes this route highly amenable to scale-up for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Polyfluoroalkyl Imidazo Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic technology in accelerating the development of next-generation fluorinated drugs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions seamlessly from the laboratory to the marketplace. Our state-of-the-art facilities are equipped with advanced photoreactors capable of handling large-scale LED-driven syntheses, guaranteeing consistent quality and throughput. We maintain stringent purity specifications through our rigorous QC labs, employing cutting-edge analytical techniques to verify the identity and purity of every batch. Our commitment to excellence ensures that you receive high-purity pharmaceutical intermediates that meet the highest international standards.
We invite you to collaborate with us to leverage this innovative synthesis route for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us help you optimize your supply chain and reduce time-to-market with our expert manufacturing capabilities.
