Advanced Palladium-Catalyzed Carbonylation for High-Purity 2,3-Dihydroquinolone Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocyclic scaffolds, particularly those exhibiting significant biological activity. A recent technological breakthrough documented in patent CN112239456B introduces a highly efficient preparation method for substituted 2,3-dihydroquinolone compounds, a core structure prevalent in numerous therapeutic agents. This innovation leverages a transition metal palladium-catalyzed carbonylation reaction, utilizing N-pyridine sulfonyl-o-iodoaniline and olefins as primary starting materials to achieve high reaction efficiency and broad substrate compatibility. The significance of this chemical architecture cannot be overstated, as derivatives of this skeleton have demonstrated potent anti-cancer and analgesic properties in prior medical research, making them critical targets for drug discovery pipelines. 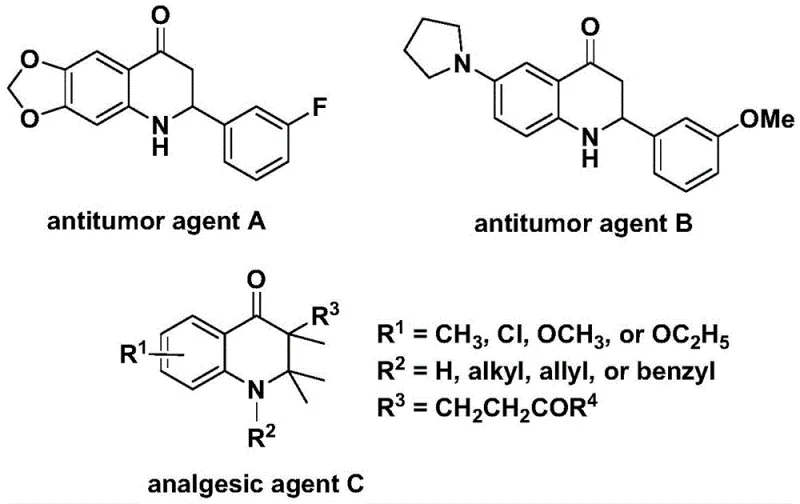
For R&D directors and procurement specialists, understanding the provenance and scalability of such intermediates is vital for securing a reliable pharmaceutical intermediates supplier partnership. The disclosed method addresses long-standing challenges in heterocycle synthesis by providing a route that is not only chemically elegant but also practically viable for commercial manufacturing. By employing a carbon monoxide substitute rather than toxic gas directly, the process enhances operational safety while maintaining high yields. This strategic advancement positions the technology as a cornerstone for cost reduction in pharma manufacturing, allowing companies to bypass complex multi-step sequences that traditionally plague the production of these valuable molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3-dihydroquinolone skeletons has relied on methodologies that often suffer from significant drawbacks regarding operational complexity and environmental impact. Traditional routes frequently require harsh reaction conditions, expensive reagents, or multiple protection and deprotection steps that drastically reduce overall atom economy. Furthermore, many conventional carbonylation methods necessitate the use of high-pressure carbon monoxide gas, which poses severe safety risks and requires specialized infrastructure that many contract development and manufacturing organizations (CDMOs) may lack. These limitations often result in prolonged lead times and inflated production costs, creating bottlenecks in the supply chain for high-purity intermediates. Additionally, the substrate scope in older methods is frequently narrow, failing to accommodate diverse functional groups required for modern medicinal chemistry optimization campaigns.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a palladium-catalyzed system that operates under relatively mild thermal conditions, specifically between 100°C and 120°C, using 1,3,5-trimesic acid phenol ester as a safe and solid carbon monoxide surrogate. This substitution eliminates the hazards associated with gaseous CO while ensuring a steady release of the carbonyl source throughout the reaction duration of 24 to 48 hours. The use of bis(acetylacetone)palladium alongside the dppp ligand creates a highly active catalytic species that facilitates the insertion of carbon monoxide and subsequent cyclization with remarkable efficiency. 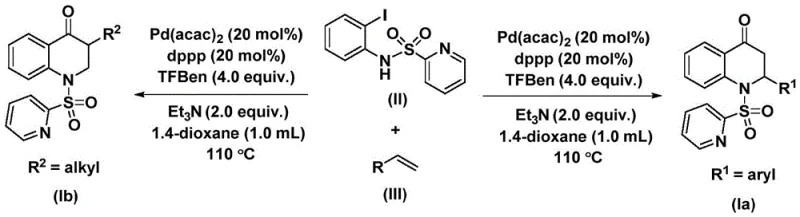
This methodology allows for the direct synthesis of both 2-aryl and 3-alkyl substituted variants through simple substrate design, significantly widening the applicability of the process. The operational simplicity extends to the work-up procedure, which involves standard filtration and column chromatography, making it accessible for both laboratory-scale optimization and commercial scale-up of complex heterocycles. By streamlining the synthetic pathway, this approach effectively reduces the number of unit operations, thereby minimizing waste generation and enhancing the overall sustainability profile of the manufacturing process.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cyclization
To fully appreciate the robustness of this synthesis, one must examine the intricate mechanistic pathway that drives the transformation of simple olefins and iodoanilines into complex bicyclic systems. The catalytic cycle initiates with the oxidative addition of the palladium catalyst into the carbon-iodine bond of the N-pyridine sulfonyl-o-iodoaniline, generating a reactive aryl-palladium intermediate. This step is crucial as it activates the aromatic ring for subsequent functionalization. Following this activation, the carbon monoxide released from the phenol ester surrogate inserts into the palladium-carbon bond, forming an acyl-palladium species. This acyl intermediate is the key precursor to the carbonyl functionality found in the final 2,3-dihydroquinolone ring system. The precision of this insertion step dictates the regioselectivity and purity of the final product, ensuring that impurities arising from alternative coupling pathways are minimized.
Subsequently, the olefin substrate coordinates with the palladium center and undergoes migratory insertion into the acyl-palladium bond, creating a new carbon-carbon bond and extending the molecular framework. This alkene insertion step is highly sensitive to steric and electronic factors, yet the chosen ligand system provides sufficient flexibility to accommodate various substituted olefins, including those with aryl, alkyl, or silyl groups. The cycle concludes with a reductive elimination step that releases the substituted 2,3-dihydroquinolone compound and regenerates the active palladium catalyst for another turnover. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the molar ratio of catalyst to ligand (optimized at 0.2:0.2), to maximize yield and ensure consistent quality across different batches of high-purity 2,3-dihydroquinolone.
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
Implementing this synthesis requires careful attention to reagent quality and reaction monitoring to ensure optimal outcomes. The process begins with the precise weighing of palladium bis(acetylacetonate), the dppp ligand, and the solid CO surrogate, which are then combined with the amine and olefin substrates in an anhydrous organic solvent such as 1,4-dioxane. The choice of solvent is critical, as it must fully dissolve the reactants while remaining stable at the elevated reaction temperature of 110°C. Detailed standardized synthesis steps are provided below to guide technical teams in replicating this high-efficiency route.
- Prepare the reaction mixture by adding palladium bis(acetylacetonate), dppp ligand, triethylamine, CO surrogate, N-pyridine sulfonyl-o-iodoaniline, and olefin into an organic solvent like dioxane.
- Heat the reaction mixture to 110°C and maintain stirring for 24 to 48 hours to ensure complete conversion of the starting materials into the intermediate species.
- Upon completion, filter the mixture, mix with silica gel, and perform column chromatography purification to isolate the high-purity substituted 2,3-dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits that directly address the pain points of procurement managers and supply chain heads. The reliance on commercially available starting materials, such as simple olefins and iodinated anilines, ensures a stable and resilient supply chain that is not dependent on exotic or hard-to-source reagents. This availability translates into reduced risk of supply disruptions and allows for more accurate forecasting of production timelines. Furthermore, the elimination of hazardous gaseous carbon monoxide simplifies regulatory compliance and facility safety requirements, potentially lowering the overhead costs associated with specialized equipment maintenance and safety protocols.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot carbonylation strategy significantly reduces the number of synthetic steps required to access the target scaffold compared to traditional multi-step routes. By avoiding expensive transition metal removal steps often associated with other catalytic systems and utilizing a recyclable solvent system, the overall cost of goods sold (COGS) is drastically optimized. The high reaction efficiency means less raw material is wasted, contributing to substantial cost savings without compromising on the quality of the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of functional groups, ensures consistent batch-to-batch reproducibility. This reliability is paramount for reducing lead time for high-purity intermediates, as it minimizes the need for re-processing or troubleshooting failed batches. The ability to source key reagents like olefins from multiple global vendors further diversifies the supply base, mitigating the risk of single-source dependency and ensuring continuous availability for downstream drug manufacturing processes.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, having already demonstrated success at the gram level with a clear path to industrial large-scale production. The use of a solid CO surrogate and the absence of high-pressure gas requirements make the technology easier to scale in standard reactor vessels. Additionally, the simplified post-treatment involving filtration and chromatography reduces the volume of chemical waste generated, aligning with modern green chemistry principles and facilitating easier environmental compliance for manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational capabilities and strategic advantages of this method for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production workflows.
Q: What are the key advantages of this carbonylation method over traditional synthesis?
A: This method utilizes readily available olefins and a robust palladium catalyst system, eliminating the need for harsh conditions often found in conventional cyclization routes. It offers superior substrate compatibility and simplifies post-treatment.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is simple to operate and has been expanded to the gram level with potential for industrial large-scale production due to the stability of the catalyst system and ease of purification.
Q: What types of substituents are tolerated in this reaction?
A: The reaction demonstrates excellent functional group tolerance, accommodating various aryl groups with methyl, tert-butyl, methoxy, fluorine, or chlorine substituents, as well as linear or branched alkyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Dihydroquinolone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug development timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into market-ready supplies. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards, guaranteeing that the 2,3-dihydroquinolone intermediates you receive are fit for purpose in sensitive pharmaceutical applications.
We invite you to collaborate with our technical procurement team to explore how this innovative palladium-catalyzed route can optimize your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this technology for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in delivering high-value chemical solutions.
