Advanced Palladium-Catalyzed Carbonylation for Commercial-Scale Production of High-Purity Substituted 2,3-Dihydroquinolone Intermediates
The patent CN112239456B introduces a groundbreaking palladium-catalyzed carbonylation methodology for synthesizing substituted 2,3-dihydroquinolone compounds, representing a significant advancement in heterocyclic chemistry for pharmaceutical applications. This innovative approach addresses critical limitations in existing synthetic routes by utilizing readily accessible starting materials including N-pyridine sulfonyl-o-iodoaniline and various olefins under mild reaction conditions. The process achieves high substrate compatibility across diverse functional groups while maintaining operational simplicity through standard laboratory equipment and procedures. Crucially, the method enables precise structural modification at both the 2 and 3 positions of the quinolone core, facilitating tailored molecular design for specific biological activities. This strategic flexibility positions the technology as a versatile platform for developing next-generation pharmaceutical intermediates with enhanced therapeutic profiles. The patent demonstrates robust scalability from laboratory to industrial production scales without requiring specialized infrastructure or hazardous reagents.
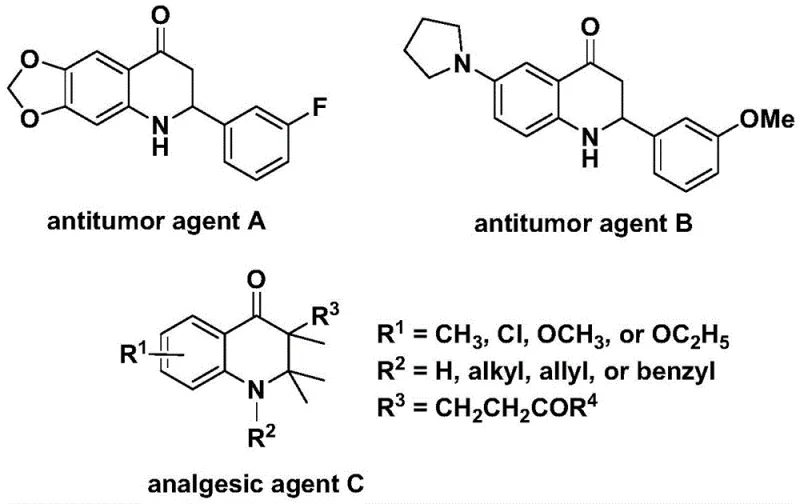
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic approaches for constructing the 2,3-dihydroquinolone scaffold often require multi-step sequences involving harsh reaction conditions such as strong acids or high temperatures that compromise functional group tolerance. These methods typically suffer from low yields due to competing side reactions and necessitate extensive purification procedures that increase production costs and reduce overall efficiency. The limited substrate scope in conventional routes restricts structural diversity at critical positions, hindering the development of optimized compounds for specific therapeutic applications. Furthermore, many existing protocols rely on expensive or unstable reagents that create supply chain vulnerabilities and complicate large-scale manufacturing operations. The absence of regioselective control in traditional syntheses frequently results in complex product mixtures requiring sophisticated separation techniques that diminish process economics. These cumulative challenges have historically constrained the commercial viability of dihydroquinolone-based pharmaceutical intermediates despite their significant therapeutic potential.
The Novel Approach
The patented methodology overcomes these limitations through a single-step palladium-catalyzed carbonylation process that operates under controlled thermal conditions between 100°C and 120°C with reaction times ranging from 24 to 48 hours. By employing N-pyridine sulfonyl-o-iodoaniline as a directing group precursor, the reaction achieves unprecedented regioselectivity while maintaining compatibility with a wide array of functional groups including halogens, alkyl chains, and silyl moieties. The use of commercially available bis(acetylacetonato)palladium catalyst with dppp ligand ensures high conversion rates without requiring specialized handling procedures or exotic reagents. This streamlined approach eliminates multiple intermediate isolation steps while delivering consistent yields across diverse substrate combinations as demonstrated in the patent examples. The simplified workup procedure involving standard filtration and column chromatography significantly reduces processing time and material waste compared to conventional multi-step syntheses. Most importantly, the method's inherent scalability from milligram to kilogram quantities provides a direct pathway to commercial production without process re-engineering.
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The catalytic cycle begins with oxidative addition of palladium(0) into the carbon-nitrogen bond of N-pyridine sulfonyl-o-iodoaniline, forming a key aryl palladium intermediate that undergoes subsequent carbon monoxide insertion from the TFBen source to generate an acyl palladium species. This critical step establishes the carbonyl functionality essential for quinolone ring formation while maintaining stereochemical control through chelation effects from the pyridine sulfonyl directing group. The olefin substrate then coordinates with the acyl palladium complex, facilitating migratory insertion that constructs the six-membered heterocyclic ring system with precise regiochemistry at both the 2 and 3 positions. Reductive elimination completes the catalytic cycle by releasing the substituted dihydroquinolone product while regenerating the active palladium(0) species for subsequent turnovers. This mechanism operates efficiently under mild thermal conditions due to the synergistic effects between the palladium catalyst and dppp ligand that stabilize all intermediate species throughout the reaction sequence.
Impurity control is achieved through multiple built-in mechanisms within this catalytic system, starting with the selective activation of the C-N bond that minimizes competing side reactions at other molecular sites. The pyridine sulfonyl group serves as both a directing element and a temporary protecting group that prevents unwanted oxidation or decomposition pathways during the reaction. Precise control over carbon monoxide release from TFBen ensures consistent carbonylation without over-insertion or byproduct formation that could complicate purification. The solvent system using anhydrous dioxane provides optimal polarity for intermediate stabilization while facilitating easy separation during workup. Post-reaction purification through silica gel filtration effectively removes residual catalysts and ligands without requiring additional scavenging steps, resulting in high-purity products suitable for pharmaceutical applications without extensive chromatographic processing.
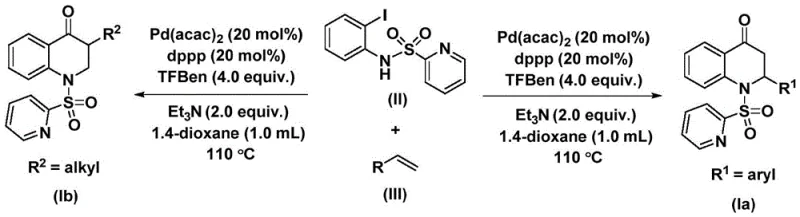
How to Synthesize Substituted 2,3-Dihydroquinolone Efficiently
This innovative synthesis route represents a paradigm shift in dihydroquinolone production by replacing complex multi-step sequences with a single catalytic transformation that maintains exceptional control over molecular architecture. The methodology leverages commercially available starting materials and standard laboratory equipment to deliver high-value pharmaceutical intermediates with remarkable efficiency and reproducibility across diverse structural variants. By eliminating traditional limitations in substrate scope and functional group compatibility, this approach enables rapid development of customized compounds tailored to specific therapeutic requirements while maintaining stringent quality standards. The patent demonstrates robust performance across fifteen distinct examples with varying substituents, confirming the method's versatility for creating diverse compound libraries essential for drug discovery programs. Detailed standardized synthesis steps are provided below to facilitate immediate implementation in research and manufacturing environments.
- Prepare reaction mixture by combining N-pyridine sulfonyl-o-iodoaniline (II), olefin (III), bis(acetylacetonato)palladium catalyst (20 mol%), dppp ligand (20 mol%), triethylamine (2.0 equiv.), and TFBen (4.0 equiv.) in anhydrous dioxane under inert atmosphere.
- Heat the sealed reaction vessel at 110°C for 48 hours with continuous stirring to ensure complete conversion through palladium-mediated carbonylation and alkene insertion.
- Perform post-treatment by filtration through silica gel followed by column chromatography purification using standard elution protocols to isolate the substituted 2,3-dihydroquinolone product.
Commercial Advantages for Procurement and Supply Chain Teams
This patented methodology delivers substantial strategic advantages for procurement and supply chain operations by addressing critical pain points in pharmaceutical intermediate sourcing through fundamental process improvements rather than incremental optimizations. The elimination of multi-step syntheses reduces raw material requirements while enhancing supply chain resilience through reliance on widely available commercial reagents instead of specialized or restricted compounds. This approach significantly simplifies vendor qualification processes and mitigates single-source dependency risks that commonly plague complex intermediate production. The streamlined manufacturing workflow directly translates to reduced production cycle times without requiring capital-intensive equipment upgrades or specialized technical expertise. These operational enhancements collectively strengthen supply chain continuity while providing greater flexibility to respond to fluctuating market demands for high-value pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The single-step catalytic process eliminates multiple intermediate isolation and purification stages required in conventional syntheses, substantially reducing solvent consumption, labor requirements, and waste disposal costs. By utilizing commercially available catalysts and ligands without requiring precious metal recovery systems, the method achieves significant material cost savings while maintaining high product quality standards through inherent reaction selectivity that minimizes impurity formation.
- Enhanced Supply Chain Reliability: The reliance on globally sourced starting materials with established supply networks ensures consistent availability regardless of regional disruptions, while the simplified process design enables rapid technology transfer between manufacturing sites without extensive revalidation procedures. This operational flexibility allows for dynamic allocation of production capacity across multiple facilities to maintain uninterrupted supply even during peak demand periods or unexpected logistical challenges.
- Scalability and Environmental Compliance: The methodology demonstrates seamless scalability from laboratory to commercial production volumes using standard reactor configurations without requiring specialized equipment modifications. The reduced number of processing steps significantly lowers energy consumption and waste generation per unit output while eliminating hazardous reagents that complicate environmental compliance procedures. This inherently greener process design aligns with evolving regulatory requirements for sustainable manufacturing practices without compromising production efficiency or product quality.
Frequently Asked Questions (FAQ)
The following questions address key technical considerations raised by procurement teams regarding implementation of this patented synthesis methodology in commercial production environments. Each response is derived directly from experimental data and process descriptions documented in patent CN112239456B to ensure technical accuracy and practical relevance for manufacturing decision-makers. These insights provide essential context for evaluating the technology's suitability for specific production requirements while highlighting its competitive advantages over conventional approaches.
Q: How does this method improve upon conventional synthesis of dihydroquinolone compounds?
A: The palladium-catalyzed carbonylation approach eliminates multi-step sequences required in traditional methods by directly constructing the quinolone core through C-N bond activation. This reduces intermediate handling and purification steps while accommodating diverse functional groups without protective group strategies.
Q: What are the scalability advantages of this palladium-catalyzed process?
A: The reaction demonstrates consistent yields from milligram to gram scale with straightforward solvent and catalyst recovery protocols. The use of commercially available starting materials and standard purification techniques enables seamless transition to industrial production volumes without specialized equipment.
Q: How does the N-pyridine sulfonyl directing group enhance reaction efficiency?
A: The pyridine sulfonyl moiety facilitates regioselective C-N bond cleavage and stabilizes key palladium intermediates during carbonylation. This design allows broad substrate scope with aryl and alkyl olefins while maintaining high functional group tolerance across diverse reaction conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted 2,3-Dihydroquinolone Supplier
Our patented methodology represents a significant advancement in dihydroquinolone synthesis that directly addresses critical challenges in pharmaceutical intermediate manufacturing through innovative catalytic design and process optimization. NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production while maintaining stringent purity specifications required for pharmaceutical applications through rigorous QC labs equipped with advanced analytical capabilities. Our integrated manufacturing platform combines deep technical expertise in heterocyclic chemistry with robust quality management systems to deliver consistent product quality across all production scales without compromising on regulatory compliance or environmental stewardship standards.
We invite you to initiate a Customized Cost-Saving Analysis tailored to your specific production requirements by contacting our technical procurement team today. Request specific COA data and route feasibility assessments to evaluate how our patented synthesis methodology can enhance your supply chain resilience while delivering substantial operational efficiencies for your pharmaceutical intermediate sourcing needs.
