Advanced Electrochemical Synthesis of Aromatic Amines for Commercial Scale-up
The landscape of organic synthesis is undergoing a transformative shift towards greener, more sustainable methodologies, particularly in the production of high-value nitrogen-containing heterocycles. Patent CN113789527B, published in early 2023, introduces a groundbreaking electrochemical synthesis method for aromatic amines that fundamentally alters the traditional reliance on transition metal catalysis. This innovation leverages electrochemical oxidative dehydrogenation to convert cyclohexanone derivatives directly into aromatic amines, bypassing the need for stoichiometric oxidants or expensive palladium and copper catalysts. The core structural versatility of this approach is illustrated by the general formula (I), which encompasses a wide array of substituents including nitriles, esters, and trifluoromethyl groups, demonstrating its broad applicability across diverse chemical spaces. 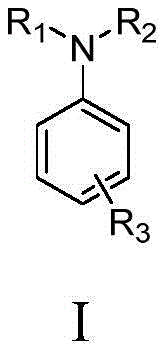 . For R&D directors and procurement specialists alike, this technology represents a significant leap forward in achieving high-purity aromatic amines while simultaneously addressing the critical industry demand for cost reduction in pharmaceutical intermediates manufacturing.
. For R&D directors and procurement specialists alike, this technology represents a significant leap forward in achieving high-purity aromatic amines while simultaneously addressing the critical industry demand for cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aromatic amine scaffolds has been dominated by classical cross-coupling reactions such as the Buchwald-Hartwig, Ullmann, and Chan-Lam protocols. While these methods are well-established in academic literature, they suffer from severe inherent limitations when applied to large-scale industrial production. Primarily, these traditional routes necessitate the use of precious metal catalysts like palladium or copper, which not only drive up raw material costs but also introduce complex downstream purification challenges to meet stringent regulatory limits on heavy metal residues in active pharmaceutical ingredients (APIs). Furthermore, these reactions often require harsh conditions, strong bases, and stoichiometric oxidants that generate substantial amounts of toxic waste, complicating environmental compliance and waste disposal logistics. Another critical drawback is the limited regioselectivity; conventional methods often struggle to differentiate between ortho, meta, and para positions without extensive protecting group strategies, thereby increasing the step count and reducing overall atom economy.
The Novel Approach
In stark contrast, the electrochemical method disclosed in the patent offers a paradigm shift by utilizing electricity as the primary reagent to drive the oxidative aromatization process. This novel approach eliminates the requirement for external metal catalysts and chemical oxidants, resulting in a reaction system where the main byproducts are merely water and hydrogen gas, aligning perfectly with the principles of green chemistry. The operational simplicity is remarkable, as the reaction proceeds under mild temperatures ranging from 25°C to 130°C and uses readily available cyclohexanone and amine starting materials. By avoiding the use of transition metals, the process inherently produces a cleaner crude reaction mixture, significantly simplifying the workup procedure and reducing the burden on quality control laboratories. This method effectively solves the regioselectivity issue by allowing chemists to pre-install the desired substitution pattern on the cyclohexanone ring prior to the electrochemical step, ensuring precise control over the final aromatic amine structure.
Mechanistic Insights into Electrochemical Oxidative Dehydrogenation
The mechanistic pathway of this electrochemical transformation is a sophisticated interplay of acid-base catalysis and anodic oxidation events that drive the aromatization of the cyclohexane ring. The process initiates with the condensation of the cyclohexanone carbonyl group with the amine nucleophile, a step facilitated by the presence of a weak acid in the electrolyte solution which protonates the carbonyl oxygen to enhance its electrophilicity. This leads to the formation of an enamine intermediate, which serves as the crucial substrate for the subsequent electrochemical steps. Once formed, the enamine migrates to the anode surface where it undergoes a single-electron transfer (SET) to generate a cationic radical species. This highly reactive intermediate is then deprotonated by a weak base present in the solution, yielding a neutral carbon-centered radical.  . This radical undergoes a second anodic oxidation to form a cation, which is subsequently deprotonated again by the weak base to establish a new double bond. Repetition of this oxidation-deprotonation cycle completes the aromatization process, converting the saturated ring into the stable aromatic system observed in the final product.
. This radical undergoes a second anodic oxidation to form a cation, which is subsequently deprotonated again by the weak base to establish a new double bond. Repetition of this oxidation-deprotonation cycle completes the aromatization process, converting the saturated ring into the stable aromatic system observed in the final product.
From an impurity control perspective, this mechanism offers distinct advantages over thermal oxidation methods. Because the oxidation potential is precisely controlled by the applied current (typically 3-30 mA), over-oxidation of sensitive functional groups is minimized, preserving the integrity of moieties like nitriles and esters which might otherwise degrade under harsh chemical oxidants. The weak base plays a dual role not only in facilitating the deprotonation steps but also in shuttling protons to the cathode where they are reduced to hydrogen gas, effectively regenerating the base and maintaining the pH balance of the system. This catalytic cycle ensures that the reaction proceeds with high efficiency and minimal side reactions, resulting in a cleaner impurity profile that is critical for pharmaceutical applications. The absence of metal catalysts also means there is no risk of metal-mediated side reactions such as homocoupling or catalyst decomposition products, further enhancing the purity of the final aromatic amine.
How to Synthesize Aromatic Amines Efficiently
The practical implementation of this electrochemical synthesis route is straightforward and relies on standard electrolytic equipment available in most modern process development laboratories. The protocol involves dissolving the cyclohexanone precursor, the amine coupling partner, an electrolyte salt, and catalytic amounts of weak acid and base in a polar aprotic solvent such as acetonitrile or DMF. The detailed standardized synthesis steps, including specific molar ratios and electrode configurations, are outlined below to ensure reproducibility and optimal yield.
- Dissolve cyclohexanone compounds, amine compounds, electrolyte, weak acid, and weak base in a suitable solvent to form a mixed solution.
- Place the mixture in an electrolytic device and perform electrolysis at 25-130°C with a current of 3-30 mA for 2-24 hours.
- Cool the reaction, extract with organic solvents, dry, and purify via column chromatography to obtain the high-purity aromatic amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into tangible strategic benefits that extend far beyond the laboratory bench. The elimination of precious metal catalysts removes a significant variable from the raw material cost structure, insulating the production process from the volatile market prices of palladium and other rare earth metals. Furthermore, the simplified downstream processing reduces the consumption of solvents and scavenging agents, leading to substantial cost savings in waste management and utility usage. The robustness of the reaction conditions ensures a reliable aromatic amine supplier can maintain consistent output even when facing fluctuations in raw material quality, as the electrochemical parameters can be finely tuned to compensate.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the complete removal of expensive transition metal catalysts and ligands from the bill of materials. In traditional synthesis, these components can account for a significant portion of the COGS (Cost of Goods Sold), and their removal drastically lowers the entry barrier for production. Additionally, the generation of hydrogen and water as the sole byproducts eliminates the need for costly disposal of heavy metal waste streams, further enhancing the economic viability of the process. The mild reaction conditions also reduce energy consumption compared to high-temperature thermal processes, contributing to a leaner and more efficient manufacturing footprint.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as cyclohexanones and simple amines ensures a stable and diversified supply base, reducing the risk of bottlenecks associated with specialized reagents. Since the process does not depend on air-sensitive or moisture-sensitive catalysts, the storage and handling requirements are significantly relaxed, simplifying logistics and inventory management. This robustness allows for longer campaign runs and reduces the frequency of reactor turnarounds, ensuring a continuous flow of high-purity aromatic amines to meet downstream demand without interruption.
- Scalability and Environmental Compliance: Electrochemical reactors are inherently scalable, allowing for the seamless transition from gram-scale optimization to multi-ton commercial production without the need for extensive re-engineering. The green nature of the synthesis, characterized by the absence of toxic oxidants and heavy metals, aligns perfectly with increasingly stringent global environmental regulations, future-proofing the supply chain against regulatory shocks. This sustainability profile not only mitigates compliance risks but also enhances the brand value of the final pharmaceutical products by associating them with eco-friendly manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of aromatic amines, providing clarity on the feasibility and advantages of this innovative technology. These answers are derived directly from the experimental data and technical specifications detailed in the patent documentation to ensure accuracy and relevance for industry stakeholders.
Q: Does this electrochemical method leave metal residues in the final product?
A: No, the process described in patent CN113789527B operates without additional metal catalysts, eliminating the risk of heavy metal contamination and the need for expensive scavenging steps.
Q: Can this method achieve specific regioselectivity for ortho, meta, or para positions?
A: Yes, by selecting various substituted cyclohexanones as starting materials, the method allows for the convenient preparation of arylamines functionalized at ortho, meta, and para positions.
Q: Is this electrochemical process suitable for large-scale industrial production?
A: Absolutely. The patent highlights that the reaction conditions are mild, raw materials are easily accessible, and the method is explicitly designed for easy amplification and strong practical utility in commercial settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in reshaping the supply chain for critical pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of aromatic amine meets the highest international standards for safety and efficacy.
We invite forward-thinking partners to collaborate with us to leverage this cutting-edge technology for their next-generation drug candidates. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule, along with specific COA data and route feasibility assessments. Let us help you navigate the transition to greener, more cost-effective manufacturing solutions that secure your supply chain for the future.
