Advanced Copper-Catalyzed Selective Halogenation for High-Purity Pharmaceutical Intermediates
Advanced Copper-Catalyzed Selective Halogenation for High-Purity Pharmaceutical Intermediates
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the need for more sustainable, cost-effective, and selective methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN112047879B, which introduces a novel method for selectively synthesizing halogenated arylamine compounds utilizing copper catalysis. This technology represents a paradigm shift from traditional precious metal-catalyzed processes, offering a robust pathway for generating high-value building blocks used in the production of kinase inhibitors and other bioactive molecules. By leveraging inexpensive copper salts and mild reaction conditions, this approach addresses critical pain points in process chemistry, such as heavy metal contamination and harsh reaction environments. For R&D directors and procurement specialists alike, understanding the nuances of this copper-catalyzed C-H activation is essential for optimizing supply chains and reducing the overall cost of goods for complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
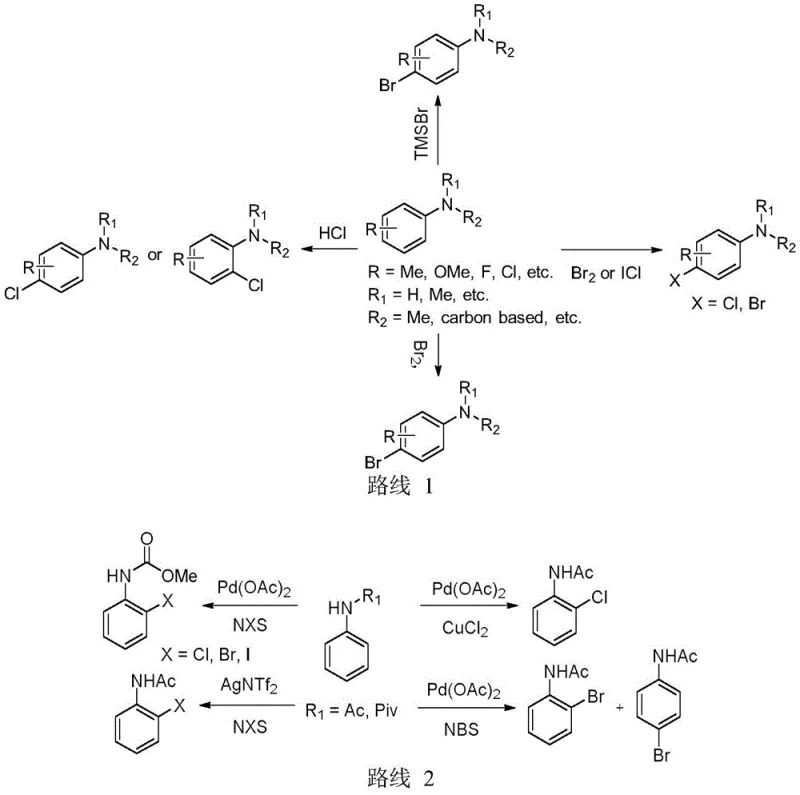
Historically, the synthesis of halogenated arylamines has relied heavily on classical electrophilic halogenation or palladium-catalyzed C-H functionalization, both of which present substantial drawbacks for large-scale manufacturing. Classical electrophilic halogenation often suffers from poor regioselectivity, leading to complex mixtures of isomers that are difficult and expensive to separate, thereby lowering overall yield and purity. Furthermore, the reagents used in these traditional methods, such as molecular halogens, are highly toxic and corrosive, posing significant safety hazards and environmental compliance challenges for production facilities. On the other hand, while palladium-catalyzed methods offer better selectivity, they are plagued by the high cost of the catalyst and the necessity for specialized, often air-sensitive ligands. These factors contribute to elevated production costs and supply chain vulnerabilities, making the reliance on precious metals a strategic risk for long-term commercial manufacturing of pharmaceutical intermediates.
The Novel Approach
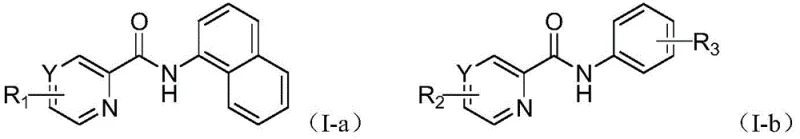
The methodology outlined in patent CN112047879B circumvents these historical limitations by employing a copper-catalyzed system that utilizes pyridine or pyrazine as a directing group to achieve precise C-H halogenation. This innovative route allows for the direct conversion of inert C-H bonds into C-X bonds (where X is Cl, Br, or I) with exceptional regioselectivity, specifically targeting the ortho-position relative to the amide nitrogen. The process operates under remarkably mild conditions, typically between 0°C and 80°C, and utilizes benign oxidants like iodobenzene diacetate alongside commercially available N-halosuccinimides as the halogen source. This not only simplifies the operational protocol but also significantly reduces the generation of hazardous waste. The ability to achieve both mono- and di-halogenation on naphthylamine derivatives with high fidelity demonstrates the versatility of this system, providing a powerful tool for constructing diverse chemical libraries required for modern drug discovery and development.
Mechanistic Insights into Copper-Catalyzed C-H Halogenation
The core of this technological advancement lies in the intricate interplay between the copper catalyst, the oxidant, and the directing group, which collectively facilitate a highly selective C-H activation cycle. The mechanism likely involves the coordination of the copper species to the nitrogen atom of the pyridine or pyrazine ring, which serves as a transient directing group to position the metal center in proximity to the target C-H bond on the aromatic ring. Upon oxidation by agents such as PhI(OAc)2, the copper center enters a higher oxidation state, enabling the cleavage of the C-H bond through a concerted metalation-deprotonation (CMD) or similar pathway. Subsequent reaction with the N-halosuccinimide reagent transfers the halogen atom to the activated carbon center, regenerating the catalyst and releasing the halogenated product. This catalytic cycle is highly efficient, minimizing the stoichiometric requirement for the metal and ensuring that the reaction proceeds with minimal formation of side products, which is crucial for maintaining high purity standards in pharmaceutical synthesis.
Furthermore, the selectivity of this reaction is profoundly influenced by the electronic and steric properties of the substrate, allowing for fine-tuning of the outcome based on specific synthetic needs. For instance, the presence of electron-donating or withdrawing groups on the aromatic ring can modulate the reactivity of the C-H bond, yet the copper system maintains robust performance across a wide range of substrates. The use of N-halosuccinimides as the halogen source is particularly advantageous as it provides a controlled release of the halogen species, preventing over-halogenation or non-selective radical pathways that often plague free-radical halogenation methods. This level of control ensures that the resulting halogenated arylamines possess the precise substitution patterns required for downstream coupling reactions, such as Suzuki or Buchwald-Hartwig aminations, which are staple transformations in the construction of complex therapeutic agents.
How to Synthesize Halogenated Arylamine Efficiently
Implementing this copper-catalyzed halogenation strategy requires careful attention to reaction parameters to maximize yield and selectivity while ensuring operational safety. The general procedure involves dissolving the N-aryl amide substrate in a polar aprotic solvent such as N,N-dimethylformamide (DMF) or 1,2-dichloroethane, followed by the addition of the copper catalyst, typically copper(II) bromide or chloride, and the oxidant. The halogenating agent is then introduced, and the mixture is stirred at a controlled temperature, usually around 60°C, for a duration of 3 to 6 hours. Monitoring the reaction progress via TLC or HPLC is recommended to determine the optimal endpoint, after which the mixture is cooled, filtered to remove insoluble salts, and concentrated. The crude product is subsequently purified using standard silica gel column chromatography to afford the target halogenated compound in high purity. For detailed standardized synthesis steps and specific optimization parameters, please refer to the guide below.
- Prepare the reaction mixture by combining the N-aryl amide substrate, copper catalyst (e.g., CuBr2), and oxidant in an organic solvent like DMF.
- Add the halogenating reagent (N-halosuccinimide) and stir the mixture at 0-80°C for 0.5 to 6 hours under air atmosphere.
- Upon completion, cool the reaction, filter, concentrate, and purify the crude product via silica gel column chromatography to obtain the halogenated arylamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this copper-catalyzed methodology offers transformative benefits for procurement managers and supply chain leaders focused on cost reduction and reliability in pharmaceutical intermediate manufacturing. The substitution of expensive palladium catalysts with abundant and inexpensive copper salts results in a drastic reduction in raw material costs, directly impacting the bottom line of production budgets. Moreover, the elimination of specialized ligands and the use of air-stable reagents simplify the procurement process, reducing the lead time for sourcing critical materials and mitigating the risk of supply disruptions associated with precious metal markets. The mild reaction conditions also translate to lower energy consumption and reduced wear on reactor equipment, contributing to overall operational efficiency and sustainability goals within the manufacturing facility.
- Cost Reduction in Manufacturing: The economic impact of switching to a copper-based catalytic system is profound, primarily driven by the significant price differential between base metals and precious metals. By eliminating the need for costly palladium complexes and their associated ligands, manufacturers can achieve substantial savings on catalyst expenses, which often constitute a major portion of the variable costs in fine chemical synthesis. Additionally, the high selectivity of the reaction minimizes the formation of impurities, thereby reducing the burden on downstream purification processes and increasing the overall yield of the desired product. This efficiency gain means less raw material is wasted, and fewer resources are expended on separation and waste treatment, further enhancing the cost-effectiveness of the entire production workflow.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the widespread availability of copper salts and N-halosuccinimides, which are commodity chemicals produced at scale by numerous global suppliers. Unlike specialized palladium catalysts that may have limited sources and long lead times, these copper reagents can be sourced reliably from multiple vendors, ensuring continuity of supply even during market fluctuations. The robustness of the reaction conditions, which tolerate air and moisture better than many sensitive organometallic processes, also reduces the logistical complexities associated with storage and handling. This stability allows for more flexible inventory management and reduces the risk of batch failures due to reagent degradation, ensuring a steady flow of high-quality intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the simplicity of the reaction setup and the use of standard organic solvents that are already prevalent in the fine chemical industry. The absence of highly toxic halogen gases and the use of solid N-halosuccinimide reagents significantly improve the safety profile of the operation, making it easier to meet stringent environmental and safety regulations. Furthermore, the reduced generation of heavy metal waste simplifies effluent treatment and disposal, aligning with green chemistry principles and corporate sustainability mandates. This environmental compatibility not only reduces compliance costs but also enhances the marketability of the final product to eco-conscious pharmaceutical clients who prioritize sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed halogenation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing platforms.
Q: What are the primary advantages of using copper catalysis over palladium for arylamine halogenation?
A: Copper catalysis offers significant cost advantages due to the abundance and lower price of copper salts compared to precious palladium complexes. Additionally, the method described in CN112047879B operates under milder conditions with high selectivity, reducing the formation of toxic byproducts often associated with traditional electrophilic halogenation.
Q: Can this method achieve double halogenation on naphthylamine derivatives?
A: Yes, the patented method uniquely enables both C2 and C4 site double halogenation as well as C4 site single halogenation of 1-naphthylamine derivatives. This level of regioselectivity is difficult to achieve with conventional methods and expands the utility of these intermediates for complex drug synthesis.
Q: Is the pyridine directing group removable after the reaction?
A: Absolutely. A key feature of this synthetic route is that the pyridine guiding group installed on the substrate can be easily removed via simple hydrolysis post-reaction. This allows for further functionalization or conversion into aromatic heterocyclic compounds without retaining the directing moiety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Halogenated Arylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the copper-catalyzed halogenation described in CN112047879B can be seamlessly transitioned from benchtop to plant. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality and process optimization ensures that every batch of halogenated arylamine we produce adheres to the highest industry standards, providing our partners with the reliability they need to advance their drug development pipelines.
We invite you to collaborate with us to leverage these cutting-edge synthetic technologies for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how our optimized processes can reduce your overall manufacturing expenses. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you navigate the complexities of fine chemical synthesis and secure a stable, cost-effective supply of critical intermediates for your business growth.
