Scalable Synthesis of Obeticholic Acid Intermediates via Novel Weinreb Amide Protection Strategy
Scalable Synthesis of Obeticholic Acid Intermediates via Novel Weinreb Amide Protection Strategy
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for complex bile acid derivatives, particularly for treating liver disorders such as primary biliary cirrhosis. Patent CN111285914B, published in early 2023, discloses a groundbreaking preparation method for Obeticholic Acid that addresses critical bottlenecks in existing manufacturing technologies. This innovation centers on the strategic use of a Weinreb amide intermediate, specifically Compound III, which serves as a pivotal junction for controlling stereochemistry and ensuring high purity throughout the synthesis. By shifting away from direct alkylation strategies that have historically plagued the field with low selectivity, this new methodology offers a streamlined pathway that is both operationally simple and economically viable for large-scale production. The technical breakthrough lies in the ability to maintain structural integrity while introducing the crucial ethyl group at the C6 position with exceptional stereocontrol. For R&D directors and process chemists, this patent represents a significant leap forward in the reliable supply of high-purity API intermediates, promising to redefine the standard for hepatic drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Obeticholic Acid has been fraught with challenges related to selectivity and yield, particularly when relying on early methodologies such as those described in WO2002072598 and WO2006122977. The conventional approach typically involves direct alkylation with ethyl iodide under strongly alkaline conditions, a process notorious for its poor regioselectivity and the formation of numerous by-products. Furthermore, subsequent steps involving silyl-protected enol compounds often result in unstable intermediates that tend to separate as oils rather than crystalline solids, making purification extremely difficult and inconsistent. These issues lead to a cumulative loss of material at every stage, resulting in a total synthesis yield that is often commercially unviable, frequently hovering below 50%. The difficulty in controlling impurities not only increases the cost of goods sold due to extensive chromatography requirements but also poses significant risks to supply chain continuity, as batch-to-batch variability becomes a persistent operational headache for procurement teams managing complex API supply chains.
The Novel Approach
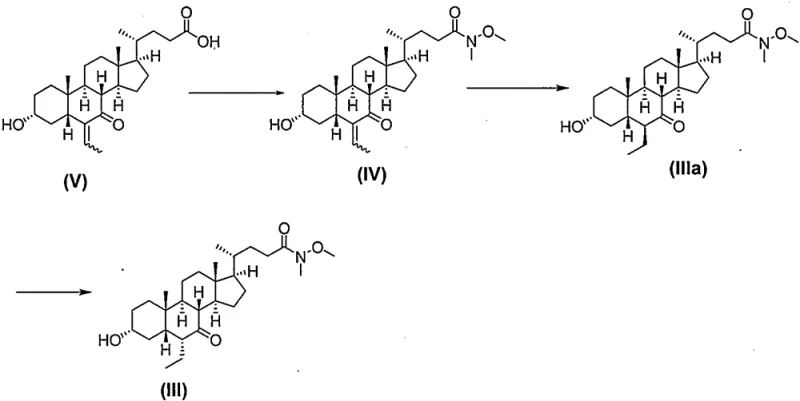
In stark contrast, the novel approach detailed in CN111285914B introduces a sophisticated protection-deprotection strategy that fundamentally alters the reaction landscape. By converting the carboxylic acid precursor (Compound V) into a Weinreb amide (Compound IV) at the very outset, the synthesis effectively masks the reactive acid functionality, preventing unwanted side reactions during subsequent reduction and alkylation steps. This modification allows for the use of milder reagents and more controlled reaction conditions, drastically reducing the formation of complex impurity profiles. The process flow is optimized to ensure that intermediates such as Compound III and Compound II can be isolated as stable, high-purity crystalline solids through simple solvent exchanges, eliminating the need for resource-intensive chromatographic separations. This shift from an oil-prone, low-yield process to a crystallization-driven, high-yield workflow represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, offering a clear path to scalable and reliable production.
Mechanistic Insights into Weinreb Amide Mediated Stereocontrol
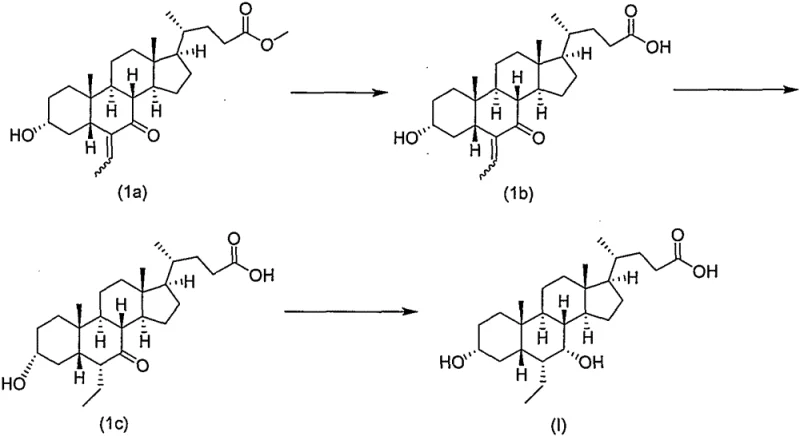
The core mechanistic advantage of this synthesis lies in the unique reactivity of the Weinreb amide functionality and its influence on the stereochemical outcome of the hydrogenation and reduction steps. When Compound IV undergoes catalytic hydrogenation using palladium on carbon, the presence of the Weinreb amide group stabilizes the molecular conformation, facilitating the selective reduction of the exocyclic double bond without affecting the amide linkage. Following this, the critical epimerization step converts the 6-beta isomer (Compound IIIa) into the desired 6-alpha configuration (Compound III) under basic conditions. This stereochemical inversion is thermodynamically driven and is significantly more efficient in the Weinreb amide series compared to free acid or ester analogues, where competing hydrolysis or decarboxylation might occur. The mechanism ensures that the ethyl group is locked into the biologically active alpha orientation with high fidelity, which is essential for the drug's efficacy as an FXR receptor agonist. Understanding this mechanistic nuance is vital for process chemists aiming to replicate these results, as it highlights the importance of maintaining the amide protection until the final stages of the synthesis to preserve chiral integrity.
Furthermore, the final reduction of the 7-keto group in Compound III to the 7-alpha hydroxyl group in Compound II demonstrates exceptional chemoselectivity. Using sodium borohydride in methanol at low temperatures allows for the precise reduction of the ketone without reducing the Weinreb amide or causing epimerization at other chiral centers. This level of control is difficult to achieve in unprotected acid forms, where the carboxylate anion can interfere with the reducing agent or alter the solvation shell around the ketone. The subsequent hydrolysis of the Weinreb amide in Compound II to release the free carboxylic acid of Obeticholic Acid (Compound I) is performed under alkaline conditions that are mild enough to prevent degradation of the sensitive steroid backbone. This careful orchestration of functional group transformations ensures that the final API meets stringent purity specifications, minimizing the risk of genotoxic impurities or structurally related by-products that could complicate regulatory filings.
How to Synthesize Obeticholic Acid Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters, particularly temperature control and solvent selection during the coupling and reduction phases. The initial formation of the Weinreb amide is best conducted in dichloromethane using EDCI and HOBt as coupling agents, ensuring complete conversion of the starting acid before proceeding to hydrogenation. The hydrogenation step benefits from the use of ethyl acetate as a solvent, which facilitates the subsequent crystallization of the intermediate. For the epimerization and ketone reduction steps, maintaining low temperatures is critical to preventing thermal degradation and ensuring high diastereoselectivity. The detailed standardized synthesis steps, including specific reagent ratios, reaction times, and workup procedures required to achieve the reported high yields and purities, are outlined in the guide below.
- Protect the carboxyl group of Compound V using N,O-dimethylhydroxylamine and EDCI/HOBt coupling agents to form the Weinreb amide Compound IV.
- Perform catalytic hydrogenation on Compound IV using Pd/C to reduce the exocyclic double bond, followed by base-mediated epimerization to obtain Compound III with the correct 6-alpha configuration.
- Reduce the 7-keto group of Compound III using sodium borohydride to form Compound II, followed by alkaline hydrolysis to remove the protecting group and yield Obeticholic Acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers profound advantages for procurement managers and supply chain heads looking to optimize their API sourcing strategies. The most significant benefit is the drastic improvement in overall process yield, which directly translates to substantial cost savings in raw material consumption and waste disposal. By eliminating the need for complex purification techniques like preparative HPLC and replacing them with robust crystallization steps, the manufacturing process becomes significantly more scalable and less prone to batch failures. This reliability is crucial for maintaining continuous supply lines, especially for high-demand therapeutics like Obeticholic Acid where market shortages can have severe clinical implications. Additionally, the use of common, non-proprietary reagents such as palladium carbon and sodium borohydride reduces dependency on specialized catalyst suppliers, further enhancing supply chain resilience and reducing lead time for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The implementation of Weinreb amide protection eliminates the expensive and inefficient steps associated with direct alkylation, such as the use of excess alkylating agents and extensive cleanup procedures. By achieving near-quantitative yields in the protection step and maintaining high efficiency through subsequent reductions, the overall material throughput is maximized. This efficiency gain means that less starting material is required to produce the same amount of final API, effectively lowering the variable cost per kilogram. Furthermore, the ability to isolate intermediates as pure solids reduces the inventory holding costs associated with unstable oils and minimizes the loss of valuable material during storage and handling, contributing to a leaner and more cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: The robustness of the crystallization processes described in this patent ensures that production schedules can be met with high predictability. Unlike previous methods where the formation of oily residues could delay batches for days or weeks while attempting to induce solidification, this new route provides consistent solid forms that are easy to filter, dry, and analyze. This consistency reduces the risk of out-of-specification results and the need for re-processing, which are common causes of supply disruptions. For supply chain planners, this means more accurate forecasting and the ability to commit to longer-term delivery contracts with confidence, knowing that the underlying chemistry is stable and reproducible on a multi-ton scale.
- Scalability and Environmental Compliance: The synthetic route is designed with green chemistry principles in mind, utilizing solvents like ethyl acetate and methanol which are easier to recover and recycle compared to more hazardous alternatives. The high atom economy of the coupling and reduction steps minimizes the generation of chemical waste, simplifying effluent treatment and reducing the environmental footprint of the manufacturing facility. This alignment with environmental compliance standards is increasingly important for multinational corporations seeking to partner with suppliers who demonstrate a commitment to sustainability. The process is inherently scalable, moving seamlessly from laboratory benchtop to pilot plant and finally to commercial production without the need for fundamental re-engineering, ensuring a smooth technology transfer and rapid time-to-market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical benefits of the Weinreb amide strategy. Understanding these details helps stakeholders evaluate the feasibility of adopting this route for their own manufacturing needs or sourcing requirements. The focus is on resolving uncertainties regarding yield, purity, and operational complexity to facilitate informed decision-making.
Q: What is the primary advantage of using Weinreb amide protection in Obeticholic Acid synthesis?
A: The use of Weinreb amide protection significantly improves the purification efficiency of intermediates and prevents over-reduction or side reactions at the carboxyl terminus, leading to higher overall yields and purity compared to direct alkylation methods.
Q: How does this new process improve impurity control compared to prior art?
A: Unlike previous methods that often resulted in oily, unstable intermediates difficult to purify, this process utilizes stable crystallization conditions for intermediates like Compound III and IV, allowing for effective removal of impurities and achieving final product purity exceeding 99%.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process employs mild reaction conditions, common reagents like Pd/C and sodium borohydride, and robust crystallization steps, making it highly suitable for industrial scale-up with consistent quality and reduced operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Obeticholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route disclosed in CN111285914B and are fully equipped to leverage this technology for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield process are realized in actual manufacturing output. Our state-of-the-art facilities are designed to handle the specific requirements of steroid chemistry, including dedicated hydrogenation suites and controlled temperature reactors necessary for the sensitive reduction steps. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Obeticholic Acid intermediate meets the highest international standards, providing our clients with the assurance of quality they need for their regulatory submissions.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain and reduce your overall cost of goods. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to this Weinreb amide-based route can impact your bottom line. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a stable, high-quality supply of this critical hepatic therapeutic intermediate, ensuring your projects move forward without interruption.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
