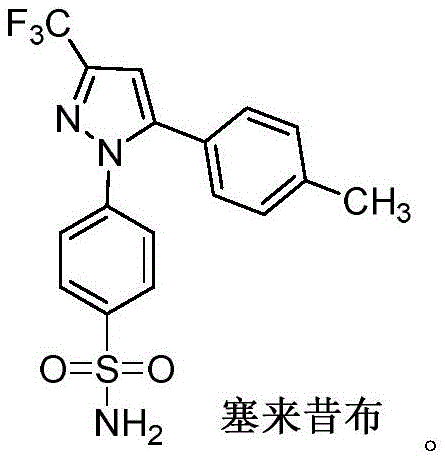
Advanced Copper-Catalyzed Synthesis of Fluoroalkylpyrazole Intermediates for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value anti-inflammatory agents, particularly COX-2 inhibitors like Celecoxib and Deracoxib. Patent CN112521290B introduces a groundbreaking methodology for synthesizing fluoroalkylpyrazole compounds, which serve as critical scaffolds in modern medicinal chemistry. This innovation centers on a novel intermediate, the β-amino ketoene compound, derived from the catalytic ring-opening of isoxazole derivatives. Unlike traditional approaches that often rely on hazardous reagents or energy-intensive conditions, this protocol leverages copper salt catalysis to achieve high efficiency and operational simplicity. For R&D directors and procurement specialists, understanding this technology is vital for securing a reliable pharmaceutical intermediates supplier capable of delivering high-purity materials. The strategic shift towards this copper-catalyzed route represents a significant advancement in process chemistry, offering a viable solution for the commercial scale-up of complex pharmaceutical intermediates while addressing longstanding safety and efficiency concerns in the supply chain.
Historically, the synthesis of 1,5-diarylpyrazole derivatives has been plagued by significant technical limitations that hinder industrial adoption. Early methodologies, such as those described by Isakson et al. in 1997, laid the groundwork but often lacked the efficiency required for modern manufacturing demands. More recent attempts, like the microwave-assisted substitution reactions reported by Britton and Jamison, utilize excessive amounts of iodide reagents, reaching up to 4 equivalents, which drastically reduces atom economy and increases waste disposal costs. Furthermore, the reliance on microwave energy sources creates a bottleneck for thermal scalability, rendering such processes unsuitable for multi-ton production campaigns. Other existing patents, such as CN110526868A, depend on acetaldehyde, a volatile and carcinogenic substance classified by the WHO, posing severe safety risks and regulatory hurdles for large-scale chemical synthesis. Similarly, routes involving Friedel-Crafts acylation with aluminum chloride, as seen in CN1076865A, suffer from extreme sensitivity to moisture and exothermic hazards, making temperature control difficult and yield consistency poor. These conventional methods collectively highlight the urgent need for a safer, more controllable, and economically viable synthetic strategy.
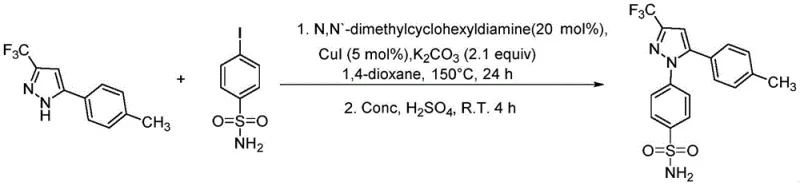
The novel approach disclosed in the patent data fundamentally reimagines the construction of the pyrazole core by utilizing an isoxazole ring-opening strategy. This method bypasses the need for hazardous acetaldehyde or moisture-sensitive Lewis acids, instead employing stable isoxazole precursors that undergo smooth transformation under copper catalysis. The process initiates with the ring-opening of the isoxazole compound in the presence of copper salts, such as Cu(OAc)2 or CuI, and specific organic ligands like trans-N,N'-dimethyl 1,2-cyclohexanediamine. This catalytic system facilitates the formation of the key β-amino ketoene intermediate with exceptional precision. Subsequent cyclization with hydrazine derivatives under mild acidic conditions yields the target fluoroalkylpyrazole structures. By shifting the synthetic logic from direct substitution or acylation to a ring-opening/cyclization sequence, the new method achieves superior control over reaction parameters. This transition not only enhances the purity profile of the final API intermediate but also simplifies the downstream purification processes, thereby reducing the overall cost reduction in pharmaceutical manufacturing significantly.
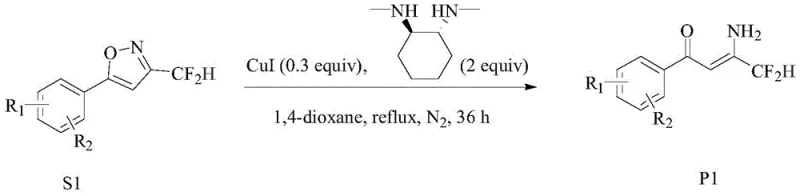
Mechanistically, the success of this synthesis relies on the precise coordination between the copper catalyst and the organic ligand to activate the isoxazole ring. The copper species, whether in the +1 or +2 oxidation state, coordinates with the nitrogen atoms of the ligand, creating a reactive complex that promotes the transfer of hydrogen atoms to the isoxazole ring. This activation lowers the energy barrier for ring cleavage, allowing the transformation to proceed efficiently at temperatures between 120°C and 140°C. The choice of solvent plays a critical role, with 1,4-dioxane demonstrating superior performance compared to THF or toluene, likely due to its ability to stabilize the transition state and solubilize the catalytic species. The resulting β-amino ketoene intermediate possesses a unique electronic structure that predisposes it to rapid cyclization upon exposure to hydrazine salts. This mechanistic pathway ensures high regioselectivity, minimizing the formation of unwanted isomers that often complicate the purification of pyrazole drugs. For technical teams, this level of mechanistic clarity translates to predictable batch-to-batch consistency and a robust impurity profile, which are essential criteria for regulatory approval.
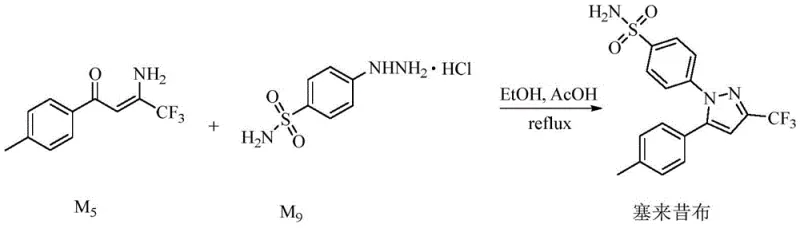
The commercial implications of adopting this synthetic route are profound for procurement and supply chain management teams seeking stability and efficiency. By eliminating the need for microwave reactors and exotic reagents, the process aligns perfectly with standard industrial infrastructure, facilitating the commercial scale-up of complex pharmaceutical intermediates without capital-intensive equipment upgrades. The use of stable, non-carcinogenic reagents significantly reduces safety compliance costs and insurance liabilities associated with handling hazardous materials like acetaldehyde or aluminum chloride. Moreover, the high yields reported, approaching 95% under optimal conditions, directly contribute to substantial cost savings by maximizing raw material utilization and minimizing waste generation. The operational simplicity of the reaction conditions, which do not require cryogenic temperatures or strict anhydrous environments, enhances supply chain reliability by reducing the risk of batch failures due to environmental fluctuations. This robustness ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands. Ultimately, this technology offers a sustainable pathway for producing essential pain management medications with a lower environmental footprint.
The technical superiority of this method is best understood through a structured analysis of its operational steps and optimization parameters. The synthesis is divided into two distinct phases: the catalytic generation of the enone intermediate and the subsequent heterocyclic closure. Each phase has been rigorously optimized to balance reaction kinetics with practical handling requirements. Understanding these steps is crucial for any contract development and manufacturing organization (CDMO) aiming to replicate or license this technology for production. The following sections detail the specific procedural logic that underpins the high efficiency of this route, providing a clear roadmap for technical implementation.
- Perform ring-opening reaction of isoxazole compounds using copper salts (CuI or Cu(OAc)2) and organic ligands in 1,4-dioxane at 120-140°C.
- Isolate the resulting β-amino ketoene intermediate through solvent removal and column chromatography.
- Conduct ring-closing reaction with p-hydrazinobenzenesulfonamide hydrochloride using acetic acid catalyst in ethanol at 50-80°C.
Frequently Asked Questions (FAQ)
Q: What are the advantages of the isoxazole ring-opening method over traditional Friedel-Crafts acylation?
A: The isoxazole ring-opening method avoids the use of water-sensitive aluminum chloride and corrosive acetyl chloride, offering milder reaction conditions and easier temperature control compared to traditional Friedel-Crafts routes.
Q: Which copper catalyst provides the highest yield for this synthesis?
A: According to patent data, Copper(II) acetate (Cu(OAc)2) provides the highest yield of nearly 95%, outperforming other copper salts like Cu(acac)2 or cuprous iodide under optimized conditions.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the method utilizes stable reagents, avoids microwave irradiation, and operates at controllable temperatures (120-140°C), making it highly feasible for commercial scale-up compared to laboratory-specific methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Celecoxib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly evaluated the copper-catalyzed isoxazole ring-opening technology and confirmed its potential for delivering high-quality fluoroalkylpyrazole intermediates. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of validating the low impurity profiles achieved by this novel route. We understand that for R&D directors, the consistency of the chemical structure and the absence of toxic residuals are paramount, and our quality systems are designed to guarantee these outcomes consistently.
We invite procurement managers and supply chain heads to collaborate with us to leverage this cutting-edge synthesis for your product pipelines. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this safer, more efficient process. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. Together, we can secure a sustainable and cost-effective supply of essential anti-inflammatory intermediates, driving value for your organization while adhering to the highest standards of safety and quality.
