Revolutionizing Agrochemical Intermediates via Green Electrochemical Dechlorination Technology
The global demand for high-performance agrochemicals continues to drive innovation in the synthesis of key molecular building blocks, specifically pyridine carboxylates which serve as critical intermediates for next-generation herbicides and insecticides. Patent CN110172710B introduces a groundbreaking electrochemical methodology that fundamentally shifts the paradigm from harsh chemical stoichiometry to precise electron-driven synthesis. This technology enables the direct conversion of trichloromethylpyridine derivatives into valuable carboxylic esters through a streamlined, one-pot electrochemical dechlorination and alcoholysis process. By leveraging the power of electricity as a clean reagent, this method addresses long-standing inefficiencies in heterocyclic chemistry, offering a pathway that is not only chemically elegant but also industrially robust. For R&D leaders and procurement strategists alike, understanding this patent is crucial as it represents a viable route to cost reduction in agrochemical manufacturing while simultaneously adhering to increasingly stringent environmental regulations regarding waste discharge and hazardous reagent usage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of pyridine carboxylates has relied heavily on multi-step synthetic routes that involve significant safety hazards and environmental liabilities. Traditional protocols, such as those disclosed in earlier patents, often necessitate the use of concentrated sulfuric acid at elevated temperatures ranging from 110 to 160 degrees Celsius to effect acylation, followed by alcoholysis. This approach generates massive quantities of high-COD acidic wastewater that requires complex and expensive neutralization and treatment processes before discharge. Furthermore, alternative pathways frequently employ thionyl chloride as a chlorinating agent to activate carboxylic acids, a practice that introduces severe toxicity risks due to the release of sulfur dioxide gas and the handling of corrosive liquids. These legacy methods create substantial bottlenecks in production throughput and inflate the total cost of ownership due to the heavy infrastructure required for corrosion resistance and waste management, making them less attractive for modern sustainable manufacturing initiatives.
The Novel Approach
In stark contrast, the electrochemical method described in the patent offers a transformative solution by integrating dechlorination and esterification into a single, cohesive unit operation. This novel approach utilizes a trichloromethylpyridine derivative, as shown in Formula (I), which undergoes cathodic reduction in the presence of an alcohol or phenol solvent to directly yield the corresponding polychlorinated pyridine carboxylate derivative, depicted in Formula (II). The reaction proceeds efficiently at normal temperature and pressure, eliminating the need for energy-intensive heating cycles and high-pressure reactors. By replacing dangerous chemical oxidants and chlorinating agents with electrons, the process inherently minimizes the generation of hazardous byproducts. This simplification of the synthetic route not only accelerates the timeline from raw material to finished intermediate but also drastically reduces the physical footprint of the manufacturing facility, positioning it as an ideal candidate for facilities seeking to modernize their production capabilities.
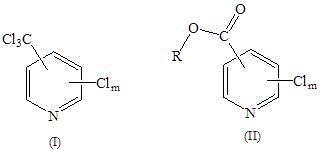
Mechanistic Insights into Electrochemical Dechlorination and Alcoholysis
The core of this technology lies in the sophisticated interplay between the electrode surface and the organic substrate within the electrolytic cell. The mechanism initiates at the cathode, where the trichloromethyl group attached to the pyridine ring undergoes a stepwise reductive dechlorination. Electrons supplied by the power source facilitate the cleavage of the carbon-chlorine bonds, generating reactive intermediates that are immediately trapped by the nucleophilic alcohol or phenol molecules present in the solution. The use of specific electrolytes, such as lithium acetate or various tetraalkylammonium salts, is critical for maintaining high conductivity and stabilizing the transition states during the electron transfer process. The patent highlights that the presence of dissolved oxygen or air in the electrolyte can further enhance the reaction efficiency, likely by participating in the regeneration of active species or preventing the over-reduction of the pyridine ring, thereby ensuring high selectivity for the desired ester product over potential side products.
Impurity control is another significant advantage derived from the mechanistic precision of this electrochemical route. In traditional acid-catalyzed reactions, the harsh conditions often lead to the degradation of the sensitive pyridine ring or the formation of polymeric tars, which are difficult to separate and lower the overall purity of the final API intermediate. The mild conditions of the electrochemical process, typically operating between 25 and 70 degrees Celsius, preserve the structural integrity of the heterocyclic core. Furthermore, the use of sacrificial anodes made of zinc, magnesium, or aluminum helps to balance the charge without introducing foreign metal contaminants that could complicate downstream purification. This inherent cleanliness of the reaction profile means that the crude product often requires minimal workup, such as simple distillation or recrystallization, to achieve the high-purity pyridine carboxylate specifications demanded by top-tier pharmaceutical and agrochemical clients.
How to Synthesize Polychlorinated Pyridine Carboxylates Efficiently
Implementing this electrochemical synthesis requires careful attention to cell configuration and parameter optimization to maximize yield and current efficiency. The patent outlines a versatile protocol where the substrate concentration, electrolyte composition, and current density are tuned to match the specific electronic properties of the starting trichloromethylpyridine derivative. Operators can choose between batch processing in stirred tanks or continuous flow systems depending on the required production volume, with the flexibility to adjust the cathode material between silver mesh, copper foam, or nickel-plated foam to suit economic constraints. The detailed standardized synthesis steps below provide a foundational guide for replicating the high conversion rates observed in the patent examples, ensuring that technical teams can rapidly validate the process in their own pilot facilities.
- Dissolve the polychlorinated pyridine derivative substrate into an alcohol or phenol solution containing a supporting electrolyte such as lithium acetate or tetraalkylammonium salts to form the electrolytic reaction liquid.
- Utilize the prepared solution as the catholyte in an electrochemical cell equipped with a porous silver or copper cathode and a sacrificial zinc, magnesium, or aluminum anode.
- Perform electrolytic reduction dechlorination and alcoholysis at the cathode under controlled current density and temperature, optionally introducing oxygen or air to enhance reaction efficiency.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology translates into tangible strategic benefits that extend far beyond simple reaction yields. The elimination of concentrated sulfuric acid and thionyl chloride from the supply chain removes the volatility associated with sourcing and transporting these highly regulated hazardous materials. This shift significantly de-risks the procurement portfolio, ensuring a more stable and predictable flow of raw materials. Moreover, the reduction in waste generation directly correlates to lower disposal costs and reduced regulatory scrutiny, allowing manufacturing sites to operate with greater flexibility and fewer interruptions due to environmental compliance audits. The ability to run the reaction at ambient pressure also lowers the capital expenditure required for reactor vessels, as there is no need for expensive high-pressure rated equipment, leading to substantial long-term savings in facility maintenance and depreciation.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling when analyzing the total cost of production. By removing the need for stoichiometric amounts of expensive and hazardous reagents like thionyl chloride, the direct material costs are significantly lowered. Additionally, the avoidance of concentrated sulfuric acid eliminates the downstream costs associated with neutralizing and treating large volumes of acidic wastewater, which is often a hidden but massive expense in traditional chemical manufacturing. The energy efficiency of running the reaction at near-ambient temperatures further contributes to a leaner cost structure, as less utility consumption is required for heating and cooling cycles compared to the high-temperature processes of the past.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and robustness of the electrochemical process. Since the method relies on electricity as the primary driver rather than a complex sequence of distinct chemical reagents, the risk of production stoppages due to the shortage of a specific catalyst or activating agent is minimized. The use of common alcohols and readily available electrolyte salts ensures that the supply base is broad and competitive, preventing vendor lock-in. This flexibility allows procurement teams to negotiate better terms and secure consistent inventory levels, ensuring that the production of critical agrochemical intermediates remains uninterrupted even during periods of global supply chain disruption.
- Scalability and Environmental Compliance: Scaling electrochemical processes is inherently modular, allowing manufacturers to increase capacity by adding more electrode pairs or cells rather than building massive new reactors. This modularity supports a agile manufacturing strategy where production can be ramped up quickly to meet market demand spikes. From an environmental perspective, the process aligns perfectly with green chemistry principles by preventing pollution at the source rather than treating it after formation. The absence of sulfur dioxide emissions and high-COD liquid waste simplifies the permitting process for new facilities and ensures long-term operational continuity in regions with strict environmental enforcement, safeguarding the company's social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical dechlorination technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when adopting this methodology. Understanding these nuances is essential for making informed decisions about process integration and technology transfer.
Q: How does this electrochemical method improve environmental compliance compared to traditional acid hydrolysis?
A: Traditional methods often rely on concentrated sulfuric acid at high temperatures, generating large volumes of high-COD acidic waste liquid that is difficult and expensive to treat. This electrochemical approach operates under mild conditions without concentrated acids, significantly reducing the environmental burden and waste treatment costs associated with hazardous effluent disposal.
Q: Does this process eliminate the need for toxic thionyl chloride reagents?
A: Yes, unlike conventional acylation routes that utilize highly toxic thionyl chloride and produce corrosive sulfur dioxide gas as a byproduct, this electrochemical dechlorination strategy avoids these hazardous reagents entirely. This enhances operational safety for plant personnel and removes the requirement for specialized scrubbing systems to handle toxic off-gases.
Q: Is this electrochemical synthesis suitable for large-scale commercial production?
A: The patent demonstrates high conversion rates and yields across various substrates using scalable electrode configurations like silver mesh and foam copper. The process can be conducted in batch, semi-continuous, or continuous flow electrolytic cells, making it highly adaptable for commercial scale-up from pilot plants to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trichloromethylpyridine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of complex heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110172710B can be successfully translated into industrial reality. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of pyridine carboxylate delivered meets the exacting standards required for global agrochemical and pharmaceutical applications.
We invite you to collaborate with us to leverage this green technology for your specific product portfolio. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your current manufacturing challenges, demonstrating exactly how switching to this electrochemical route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data for our electrochemically synthesized intermediates and to discuss route feasibility assessments for your upcoming projects, ensuring a secure and sustainable supply chain for your critical materials.
