Revolutionizing Aromatic Amine Production: Metal-Free Electrochemical Synthesis for Commercial Scale
The landscape of organic synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, and patent CN113789527A stands at the forefront of this revolution by introducing a novel electrochemical synthesis method for aromatic amines. This groundbreaking technology addresses the critical industry demand for environmentally benign processes that do not rely on scarce or toxic transition metals. By leveraging electrochemical catalysis, this method achieves the direct conversion of cyclohexanone derivatives and amines into valuable aromatic amine structures under remarkably mild conditions. The core innovation lies in the ability to drive oxidative dehydrogenation solely through electrical energy, thereby eliminating the need for stoichiometric chemical oxidants and metal catalysts that traditionally plague amine synthesis. This approach not only aligns with the principles of green chemistry by producing water and hydrogen as the primary by-products but also offers a versatile platform for generating a wide array of functionalized anilines essential for pharmaceutical and agrochemical applications. 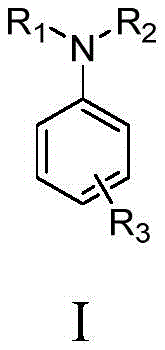
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-N bonds to form aromatic amines has relied heavily on transition-metal-catalyzed cross-coupling reactions, such as the Buchwald-Hartwig, Ullmann, and Chan-Lam couplings. While effective, these conventional methods suffer from significant drawbacks that impact both economic efficiency and environmental sustainability. Firstly, they necessitate the use of expensive and often toxic precious metal catalysts, typically based on palladium or copper, which introduces substantial raw material costs and complex supply chain dependencies. Secondly, these reactions frequently require harsh conditions, including high temperatures and strong bases, which can limit substrate scope and lead to the degradation of sensitive functional groups. Furthermore, the removal of residual metal catalysts from the final product is a rigorous and costly process, especially for pharmaceutical intermediates where strict limits on heavy metal impurities are enforced by regulatory bodies. The generation of stoichiometric amounts of metal-containing waste also poses severe challenges for waste management and environmental compliance, making these traditional routes increasingly untenable for modern large-scale manufacturing.
The Novel Approach
In stark contrast, the electrochemical synthesis method disclosed in the patent offers a transformative solution by replacing chemical oxidants and metal catalysts with electrons as the primary reagent. This novel approach utilizes a simple electrolytic cell where the oxidative dehydrogenation is driven by an applied current, typically ranging from 3 to 30 mA, at moderate temperatures between 25 and 130°C. The reaction system is elegantly simple, comprising a cyclohexanone compound, an amine, an electrolyte, and a combination of weak acids and weak bases in a common organic solvent. This metal-free protocol not only drastically reduces the cost of goods by eliminating precious metal inputs but also simplifies the work-up procedure, as there are no metal residues to remove. The versatility of this method is demonstrated by its ability to accommodate various substituted cyclohexanones, allowing for the precise functionalization of the resulting aromatic ring at ortho, meta, or para positions, which is often difficult to achieve with a single conventional method. 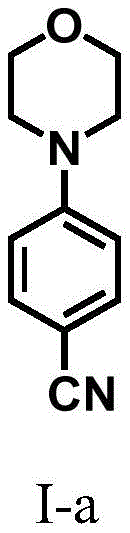
Mechanistic Insights into Electrochemical Oxidative Dehydrogenation
The mechanistic pathway of this electrochemical transformation is a sophisticated interplay of acid-base catalysis and anodic oxidation, distinct from traditional radical or ionic mechanisms. The process initiates with the condensation of the cyclohexanone carbonyl group with the amine nucleophile, a step that is critically accelerated by the presence of a weak acid. The weak acid protonates the carbonyl oxygen, increasing the electrophilicity of the carbonyl carbon and facilitating the nucleophilic attack by the amine to form an enamine intermediate. Once the enamine is formed, it migr至 the anode of the electrolytic cell, where it undergoes a single-electron oxidation to generate a cation radical species. This highly reactive intermediate is then deprotonated by the weak base present in the solution, yielding a neutral radical. This radical subsequently undergoes a second single-electron oxidation at the anode to form a cation, which finally loses a proton to the weak base to establish the aromatic double bond system. This sequential loss of electrons and protons effectively drives the aromatization of the cyclohexane ring without the need for external chemical oxidants.
Crucially, the role of the weak base extends beyond mere deprotonation; it acts as a proton shuttle that transports protons from the anodic compartment to the cathode. At the cathode, these protons are reduced to form hydrogen gas, which is released as a harmless by-product, while the weak base is regenerated and recycled back into the reaction cycle. This internal recycling mechanism ensures that the base is not consumed stoichiometrically, further enhancing the atom economy of the process. The choice of electrolyte, such as lithium perchlorate or tetrabutylammonium tetrafluoroborate, is also pivotal in maintaining conductivity and stabilizing the charged intermediates without interfering with the delicate redox potentials required for the selective oxidation of the enamine. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as current density and electrode material (graphite, RVC, or platinum), to optimize yields and selectivity for specific substrate classes, ensuring robust performance across a diverse library of aromatic amine targets.
How to Synthesize 4-(Morpholin-4-yl)benzonitrile Efficiently
The synthesis of high-value intermediates like 4-(morpholin-4-yl)benzonitrile (Compound I-a) exemplifies the practical application of this electrochemical protocol. The procedure involves dissolving 4-oxocyclohexanecarbonitrile and morpholine in acetonitrile along with lithium perchlorate as the electrolyte, collidine as the weak base, and acetic acid as the weak acid promoter. The mixture is subjected to electrolysis using graphite electrodes at a constant current of 20 mA and a temperature of 40°C for approximately 24 hours. This mild set of conditions avoids the thermal stress often associated with thermal coupling reactions, preserving the integrity of the nitrile group. Following the reaction, the work-up is straightforward, involving simple extraction and purification via flash column chromatography to afford the product in high purity. For detailed standardized synthetic steps and specific parameter optimization, please refer to the guide below.
- Prepare the electrolyte solution by dissolving cyclohexanone compounds, amine compounds, electrolyte (e.g., LiClO4), weak acid, and weak base in a suitable solvent like acetonitrile.
- Place the mixed solution into an electrolytic device equipped with graphite or platinum electrodes and apply a constant current of 3-30mA.
- Maintain the reaction temperature between 25-130°C for 2-24 hours to complete the oxidative dehydrogenation and aromatization process.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrochemical synthesis route offers profound advantages for procurement managers and supply chain directors seeking to optimize cost structures and mitigate supply risks. The most immediate impact is the drastic reduction in raw material costs associated with the elimination of precious metal catalysts. Traditional palladium-catalyzed reactions require not only the expensive metal itself but also specialized ligands, which can be cost-prohibitive at multi-ton scales. By removing these components entirely, the direct material cost of the synthesis is significantly lowered. Furthermore, the simplicity of the reagents—using commodity chemicals like cyclohexanones, morpholine, and common salts—ensures a stable and diversified supply base, reducing the risk of shortages that often plague specialized catalyst markets. The operational simplicity also translates to lower capital expenditure, as standard electrolytic equipment can be utilized instead of specialized high-pressure reactors often needed for hydrogenation or coupling reactions.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes the necessity for expensive metal scavenging and purification steps, which are traditionally resource-intensive and time-consuming. Without the need to reduce metal residues to parts-per-million levels, the downstream processing becomes much more efficient, leading to substantial savings in both time and consumables. Additionally, the use of electricity as the oxidant replaces costly chemical oxidants, further driving down the variable costs of production. The overall process efficiency is enhanced by the high atom economy, where the only waste products are benign hydrogen and water, minimizing waste disposal fees.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks such as substituted cyclohexanones and simple amines ensures a robust supply chain that is less susceptible to geopolitical disruptions compared to rare earth metals or complex ligands. The modular nature of the electrochemical setup allows for flexible production scaling, enabling manufacturers to respond quickly to fluctuating market demands without significant retooling. The mild reaction conditions also reduce the wear and tear on equipment, extending the lifecycle of production assets and ensuring consistent uptime for continuous manufacturing operations.
- Scalability and Environmental Compliance: The inherent safety of operating at ambient pressure and moderate temperatures makes this process highly scalable from laboratory benchtop to industrial kiloton production. The absence of toxic metal waste simplifies environmental compliance, as there is no need for complex heavy metal wastewater treatment facilities. The generation of hydrogen gas as a by-product can even be captured and utilized as a fuel source within the facility, contributing to a circular economy model. This alignment with green chemistry principles not only meets regulatory standards but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations. By addressing these key areas, we aim to facilitate informed decision-making for partners looking to integrate this innovative methodology into their production pipelines.
Q: What are the primary advantages of this electrochemical method over traditional cross-coupling?
A: Unlike traditional Buchwald-Hartwig or Ullmann couplings, this method eliminates the need for expensive transition metal catalysts (like Palladium or Copper) and external oxidants. The only by-products are hydrogen and water, significantly simplifying downstream purification and waste treatment.
Q: Can this process be scaled for industrial production of API intermediates?
A: Yes, the patent explicitly states the method is easy to carry out for large-scale reactions. The use of standard electrolytic cells and commercially available reagents like morpholine and cyclohexanone derivatives ensures high scalability and supply chain stability.
Q: What types of functional groups are tolerated in this synthesis?
A: The method demonstrates excellent tolerance for various functional groups, including nitriles, esters, trifluoromethyl groups, and heterocycles. This allows for the direct synthesis of diverse ortho-, meta-, and para-functionalized aromatic amines from substituted cyclohexanones.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Amines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in the production of high-purity aromatic amines and are committed to bringing this technology to commercial fruition. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and stringent purity specifications, supported by rigorous QC labs that guarantee every batch meets the highest international standards for pharmaceutical and agrochemical intermediates. We understand that consistency and quality are paramount, and our dedicated technical team is prepared to handle the nuances of electrochemical process optimization to deliver superior results.
We invite global partners to collaborate with us to leverage this cutting-edge synthesis method for your specific project needs. Whether you require custom synthesis of complex aniline derivatives or scale-up of existing routes, our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in green electrochemical manufacturing can drive value and efficiency for your supply chain.
