Advanced Pd-Catalyzed Synthesis of Indole Acetyl Imino Sulfones for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks efficient pathways to access complex heterocyclic scaffolds that serve as core structures for bioactive molecules. Patent CN113173877A introduces a groundbreaking transition metal-catalyzed multicomponent synthesis technology specifically designed for the preparation of indole acetyl imino sulfone series compounds. These compounds are of immense interest due to their potential applications in treating psychosis, hypertension, and as tyrosine kinase inhibitors. The disclosed method leverages readily available starting materials, including ortho-halogen-substituted aromatic amines, propargyl halides, and sulfoxide sulfonimides, to construct the target molecular architecture with high efficiency. By utilizing atmospheric carbon monoxide as a carbonyl source, the process eliminates the need for high-pressure equipment, significantly enhancing operational safety and feasibility for large-scale production. This innovation represents a substantial leap forward in the synthetic accessibility of these valuable pharmacophores.
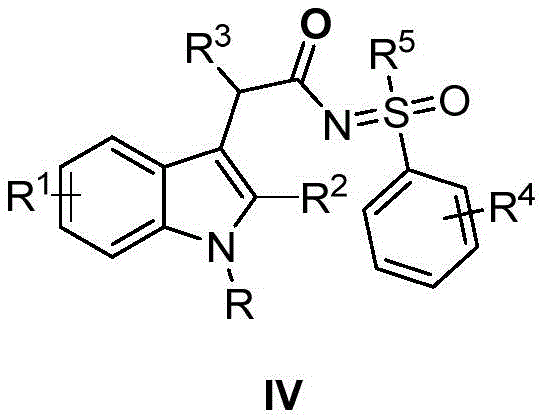
The structural versatility of the resulting indole acetyl imino sulfone derivatives allows for extensive modification at multiple positions, enabling medicinal chemists to fine-tune biological activity. The general structure, as depicted in the patent, accommodates a wide range of substituents on the indole nitrogen and the aromatic rings, including alkyl, aryl, acyl, and various halogen groups. This flexibility is crucial for structure-activity relationship (SAR) studies during drug discovery phases. Furthermore, the presence of the sulfoxide sulfimide moiety adds a layer of chemical stability and multifunctionality, which is often desirable in clinical candidates. The ability to synthesize such complex molecules in a streamlined manner addresses a critical bottleneck in the development of new therapeutic agents targeting central nervous system disorders and other indications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to indole-3-acetamide derivatives and related sulfonimide structures often suffer from significant drawbacks that hinder their industrial application. Conventional methods typically involve multi-step sequences requiring the isolation and purification of sensitive intermediates, which leads to accumulated yield losses and increased waste generation. Many existing protocols rely on pre-functionalized indole starting materials that are expensive and difficult to source commercially, thereby inflating the overall cost of goods. Additionally, carbonylation reactions in older methodologies frequently necessitate the use of high-pressure carbon monoxide reactors, posing serious safety hazards and requiring specialized infrastructure that many contract manufacturing organizations lack. The harsh reaction conditions often associated with these traditional approaches can also lead to poor chemoselectivity, resulting in complex impurity profiles that are challenging to remove during downstream processing.
The Novel Approach
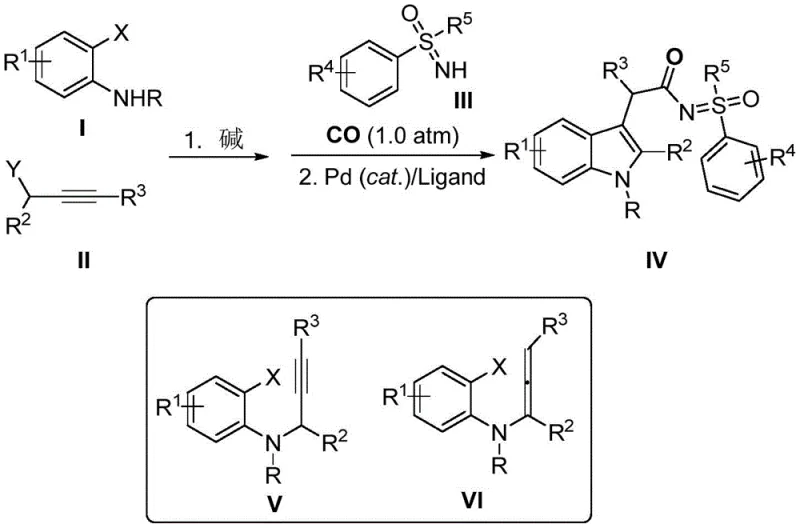
In stark contrast, the methodology described in CN113173877A offers a transformative solution by integrating the formation of the indole core and the installation of the acetyl imino sulfone group into a seamless, telescoped process. This novel approach utilizes a palladium-catalyzed cascade reaction that proceeds under atmospheric pressure of carbon monoxide, drastically reducing equipment costs and safety risks. The reaction design allows for the in situ generation of key intermediates from simple anilines and alkynes, which then immediately undergo carbonyl insertion and cyclization without the need for intermediate isolation. This one-pot strategy not only simplifies the operational workflow but also maximizes atom economy and overall yield. The mild reaction conditions, often proceeding at room temperature or slightly elevated temperatures, ensure compatibility with a broad spectrum of functional groups, making it a robust platform for synthesizing diverse libraries of biologically active compounds.
Mechanistic Insights into Pd-Catalyzed Carbonylative Cyclization
The core of this synthetic breakthrough lies in the sophisticated interplay between the palladium catalyst, the phosphine ligand, and the reaction substrates. The mechanism initiates with the oxidative addition of the ortho-halogenated aromatic amine to the palladium center, followed by the coordination and insertion of the alkyne moiety from the propargyl halide. This sequence generates a reactive organopalladium species that is poised for intramolecular cyclization to form the indole ring system. Subsequently, the introduction of carbon monoxide facilitates a migratory insertion step, creating an acyl-palladium intermediate. This acyl species then undergoes nucleophilic attack by the sulfoxide sulfonimide, ultimately releasing the final indole acetyl imino sulfone product and regenerating the active catalyst. The choice of ligand, such as triphenylphosphine, is critical in stabilizing the palladium species and promoting the desired reductive elimination step.
Controlling the impurity profile in such multicomponent reactions is paramount for pharmaceutical applications, and this protocol demonstrates exceptional selectivity. The use of specific bases like potassium tert-butoxide or cesium carbonate helps to deprotonate the amine and facilitate the initial coupling without promoting side reactions such as polymerization of the alkyne. Furthermore, the mild conditions prevent the decomposition of the sensitive sulfinimide functionality, which can be prone to hydrolysis or rearrangement under acidic or highly thermal conditions. The high yields reported across various examples, often exceeding 80-90%, indicate that competing pathways like homocoupling of the halide or incomplete carbonylation are effectively suppressed. This level of control ensures that the crude reaction mixture is relatively clean, simplifying the purification process and reducing the burden on quality control laboratories.
How to Synthesize Indole Acetyl Imino Sulfone Efficiently
The practical implementation of this synthesis involves a straightforward two-stage procedure that can be easily adapted for batch processing. Initially, the ortho-halogenated aniline and propargyl halide are combined in a solvent like acetone with a base to form the intermediate. Following the consumption of starting materials, the solvent may be exchanged or adjusted, and the sulfinimide along with the palladium catalyst system is introduced under a carbon monoxide atmosphere.
- React ortho-halogenated aromatic amines with propargyl halides in the presence of a base to generate key intermediates without isolation.
- Introduce sulfoxide sulfonimides and atmospheric carbon monoxide to the reaction mixture containing the intermediates.
- Apply a palladium catalyst and phosphine ligand to facilitate carbonyl insertion and cyclization, yielding the target indole acetyl imino sulfone compounds.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers compelling advantages that directly impact the bottom line and operational resilience. The reliance on commodity chemicals such as ortho-iodoanilines, propargyl bromide, and simple sulfinimides ensures a stable and diversified supply base, mitigating the risk of raw material shortages. The elimination of high-pressure carbon monoxide equipment removes a significant capital expenditure barrier, allowing for production in standard glass-lined or stainless steel reactors found in most multipurpose facilities. This accessibility translates to a broader network of potential manufacturing partners, fostering competition and driving down processing costs. Moreover, the telescoped nature of the reaction reduces the total processing time and labor hours required per kilogram of product, contributing to substantial operational efficiency gains.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for intermediate isolation and purification steps, which are traditionally resource-intensive. By avoiding the use of expensive pre-functionalized indole building blocks and instead constructing the core from cheaper aniline precursors, the raw material cost is drastically lowered. The use of atmospheric CO rather than pressurized gas cylinders or generators further reduces utility and safety compliance costs. Additionally, the high conversion rates minimize the loss of valuable starting materials, ensuring that the maximum amount of input is converted into saleable product, thereby improving the overall cost-efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain by reducing the likelihood of batch failures due to sensitive parameters. Since the reaction tolerates a wide range of substrates and proceeds under mild conditions, it is less susceptible to variations in raw material quality or minor fluctuations in temperature and pressure. This consistency ensures predictable lead times and dependable delivery schedules for downstream customers. The ability to source starting materials from multiple global suppliers further strengthens supply security, preventing bottlenecks that could arise from reliance on single-source specialty reagents.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the simplicity of the unit operations involved, which do not require exotic engineering controls. The reduced solvent usage and higher atom economy align with green chemistry principles, lowering the volume of hazardous waste generated per unit of product. This environmental benefit simplifies waste disposal logistics and reduces associated regulatory compliance costs. The mild reaction profile also enhances workplace safety, reducing the risk of accidents and ensuring continuous operation without unplanned shutdowns, which is critical for maintaining long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. Understanding these details is essential for project managers and technical teams evaluating the feasibility of adopting this route for their specific pipeline candidates.
Q: What are the critical reaction conditions for this carbonylation process?
A: The process operates under mild conditions using atmospheric pressure carbon monoxide (1.0 atm) and room temperature to 60°C, utilizing palladium catalysts like Pd(OAc)2 and phosphine ligands.
Q: Does this method require isolation of unstable intermediates?
A: No, the patent describes a telescoped one-pot procedure where the intermediate formed from the amine and alkyne reacts directly in the subsequent carbonylation step without separation.
Q: What is the substrate scope for the indole ring substituents?
A: The method demonstrates wide applicability, tolerating various electron-donating and electron-withdrawing groups such as fluoro, chloro, bromo, methyl, and methoxy on the aromatic rings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Acetyl Imino Sulfone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the one described in CN113173877A to deliver high-quality pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trial material and commercial launch volumes. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest standards of quality and consistency required by global regulatory agencies.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By leveraging our expertise, you can obtain a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this efficient manufacturing process. We encourage you to request specific COA data and route feasibility assessments to validate the suitability of our indole acetyl imino sulfone derivatives for your drug development programs, ensuring a successful and accelerated path to market.
