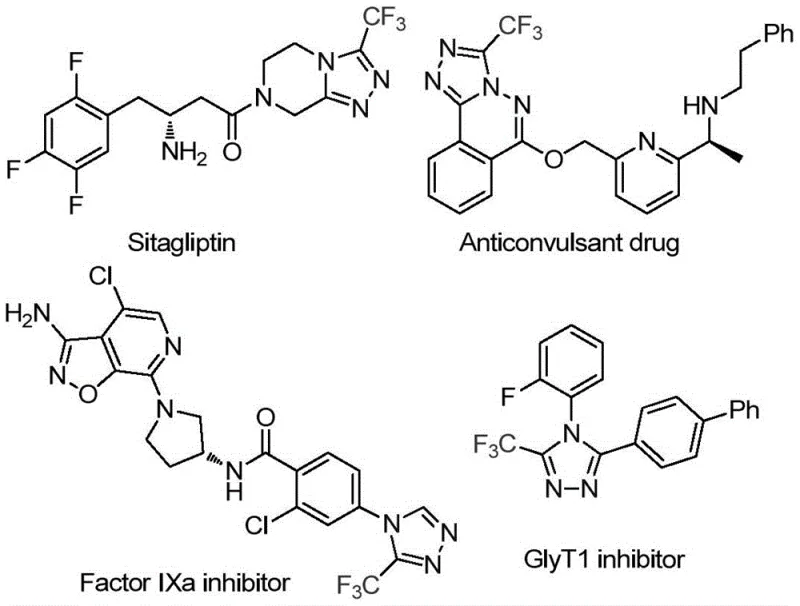
Efficient Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles via Glucose Cascade Cyclization for Pharma
Efficient Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles via Glucose Cascade Cyclization for Pharma
The pharmaceutical industry continuously seeks robust and cost-effective methodologies for constructing nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which are pivotal in modern drug design. Patent CN113880781A introduces a groundbreaking approach for synthesizing 3-trifluoromethyl-substituted 1,2,4-triazole compounds by utilizing glucose as a sustainable carbon source. This innovation addresses critical challenges in pharmaceutical intermediate manufacturing by replacing expensive or unstable aldehyde precursors with abundant biomass. The trifluoromethyl-1,2,4-triazole scaffold is a privileged structure found in numerous bioactive molecules, including antiviral agents and kinase inhibitors, making efficient access to this core essential for R&D pipelines. By leveraging a trifluoromethanesulfonic acid-catalyzed cascade reaction, this method achieves high atom economy and operational simplicity, positioning it as a superior alternative for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to 3-trifluoromethyl-1,2,4-triazoles often rely on the condensation of specific aldehydes with hydrazides, followed by oxidative cyclization. These conventional pathways frequently suffer from significant drawbacks, including the requirement for harsh reaction conditions, the use of expensive transition metal catalysts, and the necessity for strictly anhydrous and anaerobic environments. Furthermore, the aldehyde starting materials required for these reactions can be unstable, difficult to store, or costly to procure in bulk quantities, creating bottlenecks in the supply chain reliability for large-scale production. The reliance on sensitive reagents often necessitates specialized equipment and rigorous safety protocols, which drastically increases the capital expenditure and operational complexity for chemical manufacturers aiming to produce these valuable heterocycles.
The Novel Approach
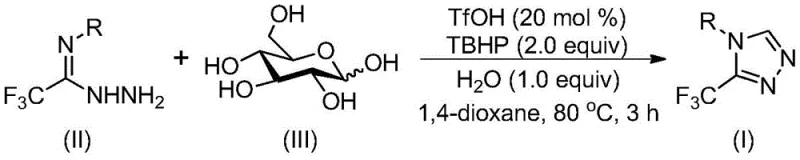
In stark contrast, the methodology disclosed in the patent utilizes glucose, a naturally occurring and inexpensive carbohydrate, as the primary carbon source. Under the catalytic action of trifluoromethanesulfonic acid (TfOH), glucose undergoes acid-catalyzed cleavage to generate reactive aldehyde intermediates in situ. These intermediates immediately condense with trifluoroethylimide hydrazide to form a hydrazone, which subsequently undergoes intramolecular nucleophilic addition and oxidative aromatization facilitated by tert-butyl hydroperoxide (TBHP). This cascade process occurs under mild thermal conditions (70-90°C) in common organic solvents like 1,4-dioxane, eliminating the need for inert atmospheres. This shift not only simplifies the operational workflow but also significantly enhances the cost reduction in API manufacturing by utilizing commodity chemicals instead of specialized fine chemicals.
Mechanistic Insights into Acid-Catalyzed Cascade Cyclization
The mechanistic pathway of this transformation is a sophisticated example of biomimetic chemistry, where the robust structure of glucose is dismantled to build complex heterocycles. Initially, the strong acid catalyst, trifluoromethanesulfonic acid, protonates the hydroxyl groups of the glucose molecule, facilitating the cleavage of carbon-carbon bonds to release reactive aldehyde species. These aldehydes then engage in a condensation reaction with the amino group of the trifluoroethylimide hydrazide, forming a hydrazone intermediate. This step is crucial as it sets the stage for the subsequent ring closure. The presence of water in the reaction system, rather than being detrimental, actually aids in the hydrolysis and activation steps, demonstrating the tolerance of this system to moisture which is rare in traditional heterocycle synthesis.
Following hydrazone formation, the mechanism proceeds through an intramolecular nucleophilic attack where the nitrogen atom attacks the electrophilic center, leading to cyclization. The final aromatization step is driven by the oxidizing agent, TBHP, which removes hydrogen atoms to establish the aromatic 1,2,4-triazole ring system. This oxidative step is highly efficient and avoids the formation of over-oxidized byproducts, ensuring a clean impurity profile. The ability to control the reaction pathway through simple adjustment of acid loading and oxidant equivalents allows for precise tuning of the process, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications without the need for extensive downstream purification.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
The synthesis protocol outlined in the patent provides a straightforward procedure accessible to most chemical laboratories and pilot plants. The process involves mixing the hydrazide substrate, glucose, catalyst, and oxidant in a solvent system, followed by heating. The robustness of the reaction allows for a wide substrate scope, accommodating various aryl and alkyl substituents on the hydrazide nitrogen. For detailed operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- Combine trifluoroethylimide hydrazide, glucose, trifluoromethanesulfonic acid, TBHP, and water in an organic solvent like 1,4-dioxane.
- Heat the reaction mixture to 70-90°C and stir for 2-4 hours to allow for glucose cleavage, condensation, and cyclization.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this glucose-based synthesis route offers transformative benefits regarding cost structure and logistical stability. The substitution of specialized aldehyde precursors with glucose, a globally traded commodity, decouples the production of these intermediates from the volatility of the fine chemical market. This strategic shift ensures a more predictable cost base and mitigates the risk of supply disruptions associated with niche reagents. Furthermore, the elimination of stringent anhydrous and anaerobic requirements reduces the dependency on specialized infrastructure, allowing for production in standard multipurpose reactors.
- Cost Reduction in Manufacturing: The utilization of glucose as a carbon source represents a significant departure from expensive synthetic aldehydes, leading to substantial raw material savings. Additionally, the use of aqueous TBHP and common acid catalysts avoids the high costs associated with noble metal catalysts or exotic oxidants. The simplified workup procedure, which involves filtration and standard chromatography, further reduces processing time and solvent consumption, contributing to overall operational efficiency and lower cost of goods sold.
- Enhanced Supply Chain Reliability: Glucose is produced on a massive industrial scale for the food and fermentation industries, ensuring an uninterrupted and abundant supply for chemical synthesis. This abundance contrasts sharply with the limited availability of many custom-synthesized building blocks used in traditional routes. By anchoring the synthesis on such a stable feedstock, manufacturers can guarantee consistent production schedules and reduce lead times for high-purity pharmaceutical intermediates, thereby strengthening the resilience of the entire supply network.
- Scalability and Environmental Compliance: The reaction conditions are inherently safer and more environmentally benign, operating at moderate temperatures without the generation of hazardous heavy metal waste. The use of water as an additive and the potential for using greener solvents align with modern sustainability goals. The process has been demonstrated to be scalable from gram levels to larger batches without loss of efficiency, making it an ideal candidate for technology transfer from R&D to commercial production facilities focused on green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this glucose-mediated triazole synthesis. These insights are derived directly from the experimental data and mechanistic understanding provided in the patent documentation, offering clarity for technical teams evaluating this route for adoption.
Q: What are the key advantages of using glucose in this synthesis?
A: Glucose serves as an inexpensive, widely available biomass feedstock that eliminates the need for complex aldehyde precursors, significantly reducing raw material costs and simplifying the supply chain.
Q: Does this method require strict anhydrous or anaerobic conditions?
A: No, the process operates under mild conditions without the need for rigorous anhydrous or oxygen-free environments, making it highly suitable for standard industrial reactor setups.
Q: What is the typical reaction temperature and time?
A: The reaction typically proceeds at temperatures between 70°C and 90°C for a duration of 2 to 4 hours, ensuring high conversion rates with minimal energy input.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of innovative synthetic routes like the glucose-based cascade cyclization for producing high-value heterocycles. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that novel academic discoveries are successfully translated into robust industrial processes. We maintain rigorous QC labs and adhere to stringent purity specifications to deliver intermediates that meet the exacting standards of global pharmaceutical clients, guaranteeing consistency and quality in every batch.
We invite you to collaborate with us to leverage this cutting-edge technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can optimize your supply chain for 3-trifluoromethyl-1,2,4-triazole derivatives.
