Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for High-Value Pharmaceutical Intermediates
Advanced Synthesis of 3-Trifluoromethyl-1,2,4-Triazoles for High-Value Pharmaceutical Intermediates
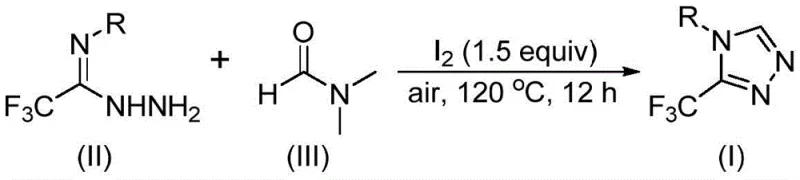
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct fluorinated heterocycles, which serve as critical scaffolds in modern drug design. Patent CN114920707B, published in April 2023, introduces a groundbreaking preparation method for 3-trifluoromethyl substituted 1,2,4-triazole compounds that addresses many longstanding synthetic challenges. This technology leverages the ubiquitous solvent N,N-dimethylformamide (DMF) not merely as a medium, but as an active carbon source, facilitating a tandem cyclization reaction promoted by molecular iodine. For R&D directors and procurement specialists alike, this innovation represents a significant shift towards more atom-economical and operationally simple processes. The ability to synthesize these high-value intermediates under air atmosphere, without stringent anhydrous conditions, drastically lowers the barrier for entry in terms of equipment requirements and safety protocols. As we delve into the technical specifics, it becomes clear that this methodology offers a robust solution for the commercial scale-up of complex fluorinated intermediates required for next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of 1,2,4-triazole rings bearing trifluoromethyl groups has been a laborious endeavor involving multi-step sequences that often suffer from poor atom economy and harsh reaction conditions. Conventional routes frequently rely on the use of specialized, expensive reagents to introduce the triazole moiety, followed by separate fluorination steps or the use of pre-fluorinated building blocks that are costly and difficult to source on a large scale. Many existing protocols necessitate the use of transition metal catalysts, which introduce the risk of heavy metal contamination in the final Active Pharmaceutical Ingredient (API), requiring additional and expensive purification steps to meet regulatory standards. Furthermore, traditional methods often demand strictly anhydrous and anaerobic environments, utilizing inert gas lines and gloveboxes that increase capital expenditure and operational complexity. These factors collectively contribute to extended lead times for high-purity pharmaceutical intermediates and inflate the overall cost of goods sold, creating bottlenecks in the supply chain for critical drug candidates.
The Novel Approach
In stark contrast, the method disclosed in CN114920707B utilizes a streamlined, one-pot strategy that transforms readily available trifluoroethyliminohydrazide and DMF into the desired triazole products with remarkable efficiency. By employing molecular iodine as a promoter, the reaction activates the DMF molecule to serve as a C1 synthon, effectively building the triazole ring in a single operational step. This approach eliminates the need for exotic reagents or complex catalytic systems, relying instead on commodity chemicals that are accessible globally. The reaction proceeds smoothly at temperatures between 110°C and 130°C under ambient air, removing the necessity for expensive inert atmosphere setups. This simplification of the reaction environment not only enhances safety by reducing the handling of pyrophoric reagents but also facilitates easier technology transfer from laboratory to pilot plant. The versatility of this method allows for the introduction of various functional groups on the aromatic ring, enabling the rapid generation of diverse libraries for structure-activity relationship (SAR) studies.
Mechanistic Insights into Iodine-Promoted Tandem Cyclization
The mechanistic pathway of this transformation is fascinating and highlights the dual reactivity of the DMF solvent. According to the patent data, the reaction can proceed via two potential pathways depending on which part of the DMF molecule acts as the carbon source. In the first pathway, the formyl group of DMF undergoes a condensation reaction with the hydrazide moiety of the starting material to generate a hydrazone intermediate. This is followed by an intramolecular cyclization event where the nitrogen attacks the electrophilic center, eventually leading to the elimination of a dimethylamine molecule to yield the aromatic triazole product. Alternatively, the N-methyl group of DMF can be activated by molecular iodine to form an amine salt species. This activated intermediate then participates in a nucleophilic addition with the hydrazide, followed by the elimination of N-methylformamide to generate a reactive azadiene intermediate. Subsequent intramolecular nucleophilic addition and oxidative aromatization finalize the formation of the 3-trifluoromethyl-1,2,4-triazole core. Understanding these pathways is crucial for R&D teams optimizing impurity profiles, as controlling the ratio of reagents and temperature can steer the reaction towards the most efficient cycle.
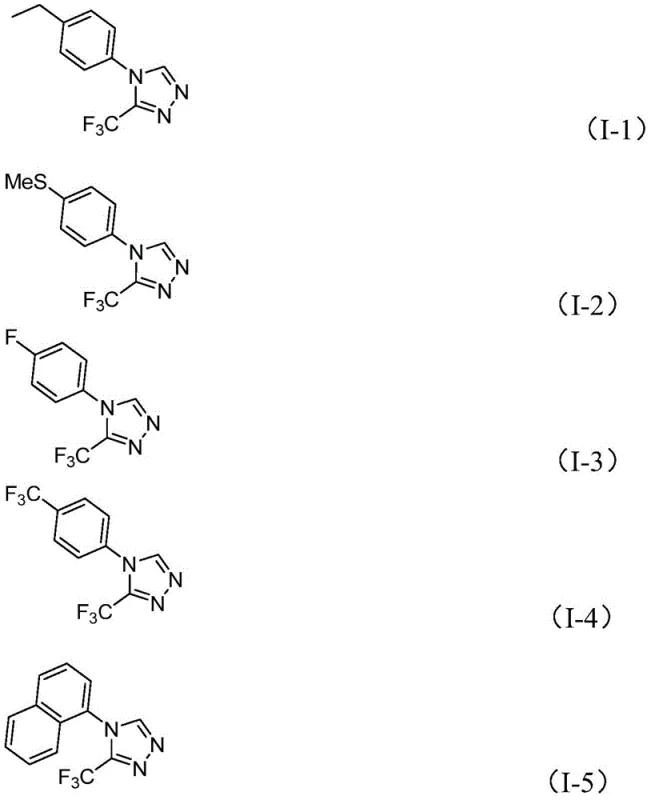
From an impurity control perspective, the mild nature of the iodine-promoted oxidation helps minimize the formation of over-oxidized byproducts or polymeric tars that are common in harsher oxidative cyclizations. The use of DMF as both solvent and reactant ensures a high local concentration of the carbon source, driving the equilibrium towards product formation and suppressing side reactions associated with intermolecular coupling of the hydrazide. The patent data indicates that substrates with varying electronic properties, such as electron-withdrawing trifluoromethyl groups or electron-donating methoxy groups, are well-tolerated, suggesting a robust catalytic cycle that is not overly sensitive to steric or electronic perturbations. This mechanistic resilience is key for ensuring consistent quality across different batches, a critical factor for reliable pharmaceutical intermediate suppliers aiming to support clinical and commercial manufacturing campaigns.
How to Synthesize 3-Trifluoromethyl-1,2,4-Triazole Efficiently
Implementing this synthesis in a laboratory or pilot setting requires careful attention to the stoichiometry of the iodine promoter and the thermal profile of the reaction. The patent outlines a straightforward protocol where trifluoroethyliminohydrazide is mixed with 1.5 equivalents of molecular iodine in DMF. The mixture is then heated to a range of 110-130°C for a duration of 10 to 15 hours. This extended heating period ensures complete conversion of the starting materials, which is vital for maximizing yield and minimizing the burden on downstream purification. The detailed standardized synthesis steps below provide a clear roadmap for chemists looking to replicate this high-efficiency route.
- Combine trifluoroethyliminohydrazide, molecular iodine (1.5 equivalents), and DMF solvent in a reaction vessel.
- Heat the mixture to 110-130°C and stir under air atmosphere for 10-15 hours to facilitate tandem cyclization.
- Upon completion, filter the mixture, wash, dry, and purify via column chromatography to isolate the target triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The reliance on DMF, a solvent produced on a massive industrial scale, ensures a stable and cost-effective supply of the primary carbon source, mitigating risks associated with the volatility of specialty reagent markets. By eliminating the need for precious metal catalysts or complex ligand systems, the process significantly reduces the raw material costs associated with each batch. Furthermore, the absence of heavy metals simplifies the purification workflow, potentially reducing the number of processing steps and the consumption of silica or other chromatographic media. This streamlining of the manufacturing process translates directly into substantial cost savings in API manufacturing, allowing companies to allocate resources to other critical areas of drug development.
- Cost Reduction in Manufacturing: The economic impact of replacing expensive, specialized reagents with commodity chemicals like DMF and iodine cannot be overstated. Traditional methods often require multiple distinct steps, each incurring its own set of material and labor costs. By consolidating the ring formation and trifluoromethylation into a single pot, this method drastically cuts down on solvent usage, energy consumption, and man-hours. The removal of transition metal catalysts also eliminates the costly and time-consuming step of metal scavenging, which is often required to meet strict regulatory limits for residual metals in pharmaceutical products. Consequently, the overall cost of goods is optimized, providing a competitive edge in pricing for the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials. Trifluoroethyliminohydrazide can be synthesized from readily accessible precursors such as aromatic amines and trifluoroacetic acid, ensuring that raw material shortages are unlikely to disrupt production schedules. The robustness of the reaction conditions, specifically the tolerance to air and moisture, means that the process can be executed in standard glass-lined reactors without the need for specialized inert atmosphere equipment. This flexibility allows for greater agility in scheduling production runs and facilitates easier scaling from kilogram to tonne quantities without significant re-engineering of the process infrastructure.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, the process aligns well with green chemistry principles by maximizing atom economy and minimizing waste generation. The use of DMF, while requiring proper handling due to its reproductive toxicity, is a well-understood solvent with established recovery and recycling protocols in most modern facilities. The reaction produces dimethylamine or N-methylformamide as byproducts, which are manageable within standard waste treatment streams. The simplicity of the workup, involving filtration and standard chromatography, reduces the volume of hazardous waste generated compared to multi-step sequences involving extractions and complex quenches. This makes the process not only scalable but also compliant with increasingly stringent environmental regulations governing chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and descriptions provided in the patent documentation, offering clarity on the practical aspects of the synthesis. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the unique role of DMF in this synthesis?
A: In this patented process, DMF serves a dual function as both the reaction solvent and the critical C1 carbon source. It provides either the formyl or N-methyl group necessary for constructing the triazole ring, eliminating the need for separate, expensive carbon donors.
Q: Does this reaction require strict anhydrous conditions?
A: No, one of the primary advantages of this method is its operational simplicity. The reaction proceeds efficiently under standard air atmosphere without the need for rigorous anhydrous or oxygen-free environments, significantly reducing equipment and operational costs.
Q: What is the substrate scope for this triazole synthesis?
A: The method demonstrates excellent versatility, accommodating various substituted aryl groups including those with electron-donating or electron-withdrawing substituents such as halogens, alkyls, and trifluoromethyl groups, making it suitable for diverse drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN114920707B for the development of advanced pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly translated into reliable commercial supply. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 3-trifluoromethyl-1,2,4-triazole meets the exacting standards required by global regulatory bodies. Our commitment to quality and consistency makes us a trusted partner for companies seeking to secure their supply chains for critical drug substances.
We invite you to collaborate with us to explore the full potential of this innovative synthesis for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this DMF-based route can optimize your budget. Please contact us to request specific COA data for our available intermediates or to discuss route feasibility assessments for your custom synthesis projects. Together, we can accelerate the delivery of life-saving medicines to the market through superior chemical manufacturing.
