Advanced One-Pot Manufacturing of Olmesartan Medoxomil Intermediates for Global Pharmaceutical Supply Chains
Advanced One-Pot Manufacturing of Olmesartan Medoxomil Intermediates for Global Pharmaceutical Supply Chains
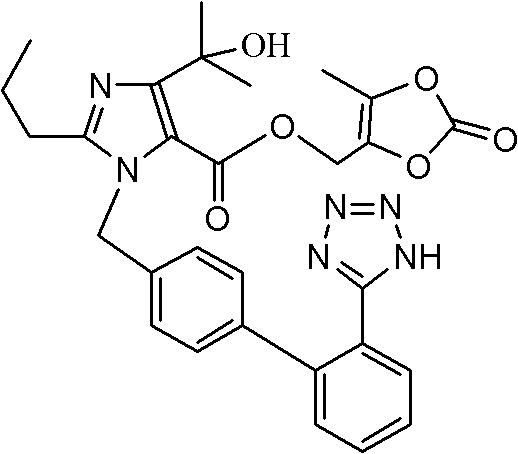
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with operational efficiency, particularly for blockbuster antihypertensive agents like Olmesartan Medoxomil. Patent CN102351849B introduces a transformative preparation method that addresses critical bottlenecks in the synthesis of key imidazole-biphenyl intermediates. This technology leverages a novel one-pot cascade reaction, merging the alkylation and hydrolysis steps into a seamless operation. By bypassing the traditional isolation of the trityl-protected ester intermediate, this approach significantly streamlines the manufacturing workflow. For R&D directors and supply chain managers, this represents a pivotal shift towards more sustainable and cost-effective production of high-purity pharmaceutical intermediates. The method utilizes mild alkaline conditions in polar aprotic solvents, ensuring safety while maintaining exceptional conversion rates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Olmesartan Medoxomil has relied on multi-step sequences that introduce significant inefficiencies. As illustrated in prior art, such as the work by Yanagisawa, the standard route involves reacting an imidazole ester with a biphenyl tetrazole bromide to form a trityl-protected ester intermediate. Crucially, this intermediate must be isolated, purified, and dried before undergoing hydrolysis to the corresponding acid or salt. This isolation step is not only labor-intensive but also a major source of yield loss and impurity generation. The physical handling of sticky or hygroscopic intermediates often leads to mechanical losses and requires substantial volumes of solvents for washing and recrystallization. Furthermore, the extended processing time increases the exposure of sensitive functional groups to potential degradation, complicating the impurity profile and necessitating rigorous downstream purification.
The Novel Approach
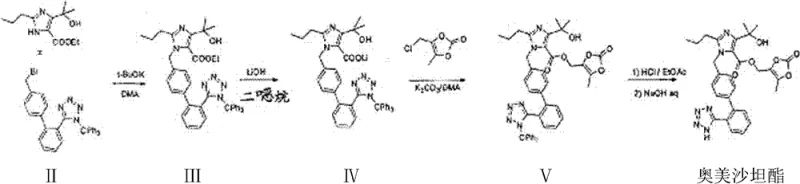
In stark contrast, the methodology disclosed in CN102351849B revolutionizes this workflow by employing a direct one-pot strategy. Instead of isolating the ethyl ester intermediate (Compound III), the process proceeds immediately to hydrolysis within the same reaction vessel. After the initial alkylation between the imidazole derivative and the trityl-tetrazole biphenyl bromide is complete, a lithium hydroxide solution is introduced directly to the reaction mixture. This eliminates the need for filtration, drying, and re-dissolution of the intermediate. The result is a drastic reduction in unit operations, solvent usage, and overall cycle time. This streamlined approach not only enhances the total yield but also simplifies the equipment requirements, making it highly attractive for reliable pharmaceutical intermediate supplier operations aiming for lean manufacturing.
Mechanistic Insights into the One-Pot Alkylation-Hydrolysis Cascade
The core of this technological advancement lies in the compatibility of the reaction conditions for both alkylation and saponification. The process initiates with the nucleophilic substitution of the bromide on the biphenyl tetrazole by the nitrogen of the imidazole ring. This step is facilitated by sodium hydroxide in dimethylformamide (DMF) at temperatures ranging from 60 to 80°C. The choice of DMF is critical as it solubilizes both the organic halide and the ionic base, promoting efficient collision frequency. Once the alkylation reaches completion, monitored typically by TLC, the system is primed for the second transformation. The addition of aqueous lithium hydroxide triggers the hydrolysis of the ethyl ester moiety. The use of lithium hydroxide specifically targets the formation of the lithium carboxylate salt (Compound IV), which exhibits favorable solubility characteristics for subsequent extraction and crystallization. This sequential addition of reagents without intermediate workup prevents the accumulation of byproducts that often arise during the storage or handling of the free acid or ester forms.
From an impurity control perspective, this mechanism offers distinct advantages. In traditional routes, the isolation of the trityl-protected ester can lead to partial detritylation or ester hydrolysis if moisture control is not perfect. By maintaining the reaction in a controlled, homogeneous phase until the final hydrolysis step, the process minimizes side reactions. The patent data indicates that this method consistently achieves high purity levels, with HPLC analysis showing purities of 99.7% to 99.8% in pilot examples. The absence of solid-liquid separation steps reduces the risk of introducing particulate matter or cross-contamination. Furthermore, the mild thermal conditions (60-70°C for hydrolysis) prevent thermal degradation of the sensitive tetrazole ring, ensuring the structural integrity of the final API precursor.
How to Synthesize Olmesartan Medoxomil Lithium Salt Efficiently
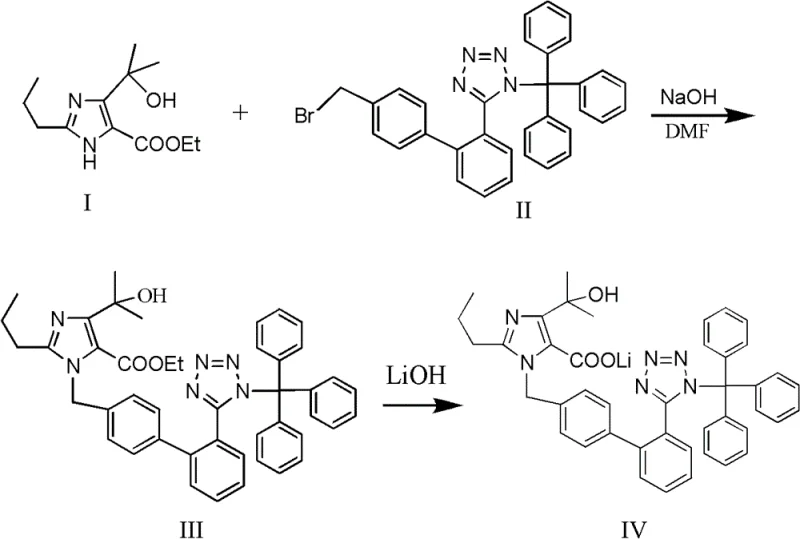
Implementing this synthesis route requires precise control over stoichiometry and temperature profiles to maximize the benefits of the one-pot design. The process begins by charging the imidazole ester and sodium hydroxide into a reactor with DMF, followed by the addition of the trityl-tetrazole biphenyl bromide. The mixture is heated to promote alkylation, after which lithium hydroxide solution is added for the hydrolysis phase. The detailed standardized operating procedures, including specific molar ratios and extraction protocols, are essential for reproducibility. For a comprehensive guide on executing this synthesis with optimal parameters, please refer to the technical instructions below.
- React ethyl 4-(1-hydroxy-1-methylethyl)-2-propylimidazole-5-carboxylate with N-(triphenylmethyl)-5-(4'-bromoethylbiphenyl-2-)tetrazole in DMF using NaOH at 60-80°C.
- Without isolating the intermediate ester, directly add lithium hydroxide solution to the reaction mixture and heat to 60-70°C for hydrolysis.
- Cool the system, extract with ethyl acetate and water, wash, dry, and evaporate to obtain the pure lithium salt intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis translates into tangible strategic benefits beyond mere chemical elegance. The reduction in unit operations directly correlates with lower manufacturing costs and reduced dependency on complex infrastructure. By eliminating the isolation step, facilities can achieve higher throughput with existing reactor capacity, effectively increasing asset utilization without capital expenditure. This efficiency is crucial for maintaining competitive pricing in the global market for angiotensin II receptor antagonists. Moreover, the simplified workflow reduces the demand for auxiliary materials such as filtration media and drying trays, contributing to a leaner supply chain.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the elimination of the intermediate isolation step. In conventional processing, filtering, washing, and drying a bulk intermediate consumes substantial energy and labor hours. By removing this step, the process drastically cuts down on utility costs and man-hours. Additionally, the improved total yield means that less raw material is required to produce the same amount of final product, directly lowering the cost of goods sold (COGS). The reduction in solvent volume for washing and recrystallization further decreases waste disposal costs, enhancing the overall economic viability of the production line.
- Enhanced Supply Chain Reliability: Shorter cycle times inherently improve supply chain responsiveness. With fewer steps to manage, the lead time from raw material intake to finished intermediate is significantly compressed. This agility allows manufacturers to react more swiftly to fluctuations in market demand for Olmesartan Medoxomil. Furthermore, the robustness of the one-pot method reduces the likelihood of batch failures associated with complex isolation procedures. A more predictable production schedule ensures a steady flow of high-purity pharmaceutical intermediates to downstream API manufacturers, mitigating the risk of stockouts and production delays.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies the challenges of heat transfer and mixing, particularly during solid handling. This one-pot method, being largely homogeneous until the final extraction, scales up more predictably than multi-step heterogeneous processes. The reduced solvent usage and waste generation align perfectly with modern environmental regulations and green chemistry principles. Lower effluent volumes simplify wastewater treatment requirements, reducing the environmental footprint of the facility. This compliance is increasingly vital for maintaining operational licenses and meeting the sustainability goals of multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into their supply chains. The following questions address common concerns regarding scalability, purity, and operational safety. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear picture of the method's capabilities.
Q: What is the primary advantage of the one-pot process for Olmesartan Medoxomil?
A: The primary advantage is the elimination of the isolation step for the trityl-protected ester intermediate. By performing alkylation and hydrolysis in a single vessel, the process reduces solvent consumption, operational time, and potential product loss, leading to higher overall yields and purity.
Q: How does this method improve impurity control compared to conventional routes?
A: Conventional methods require isolating the intermediate ester, which can introduce impurities during filtration and drying. The one-pot method minimizes handling, thereby reducing the risk of contamination and degradation, resulting in end-product purity exceeding 99.7% as demonstrated in patent examples.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial production due to mild reaction conditions (60-80°C), high safety profile, and simplified operational steps that facilitate easier scale-up and waste management.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olmesartan Medoxomil Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists has extensively analyzed advanced methodologies like the one-pot synthesis described in CN102351849B to ensure we deliver the highest quality intermediates. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Olmesartan Medoxomil intermediate meets the exacting standards required for FDA-approved drug manufacturing.
We invite global partners to collaborate with us to leverage these technological advancements for their product pipelines. By optimizing the synthesis of complex intermediates, we help our clients achieve significant operational efficiencies. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain resilience and profitability.
