Advanced Palladium-Catalyzed Synthesis of 2-Trifluoromethyl Quinazolinones for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust synthetic methodologies to access privileged heterocyclic scaffolds, particularly those containing fluorine atoms which often enhance metabolic stability and bioavailability. A significant breakthrough in this domain is detailed in patent CN113045503B, which discloses a novel preparation method for 2-trifluoromethyl substituted quinazolinone compounds. These structures are not merely academic curiosities; they form the core backbone of numerous bioactive agents, ranging from sedatives like Methaqualone to potent anticancer and anti-inflammatory agents such as Rutaecarpine. The introduction of the trifluoromethyl group at the 2-position is strategically critical, yet historically challenging due to the instability of precursors and harsh reaction conditions required for cyclization. This new technology offers a transformative approach by utilizing a transition metal palladium-catalyzed carbonylation cascade reaction, starting from cheap and readily available trifluoroethylimidoyl chloride and amines.
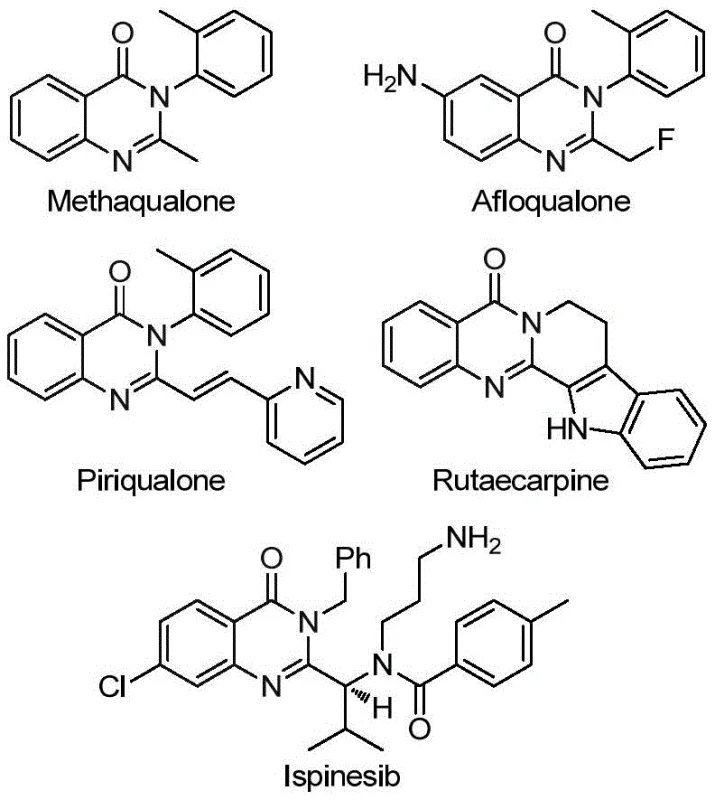
For R&D directors evaluating new routes, the versatility of this method is paramount. The patent highlights that the process is operationally simple, tolerates a wide range of functional groups, and achieves high reaction efficiency. Unlike traditional methods that might require pre-activated substrates or unstable intermediates, this protocol leverages a solid carbon monoxide substitute, TFBen, which simplifies the engineering controls needed for scale-up. As a reliable pharmaceutical intermediate supplier, understanding such mechanistic advancements allows us to offer clients superior quality intermediates with cleaner impurity profiles, directly addressing the stringent requirements of modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-trifluoromethyl-substituted quinazolinones has been plagued by significant technical hurdles that limit their widespread adoption in industrial manufacturing. Conventional literature reports typically rely on the cyclization of anthranilamide with ethyl trifluoroacetate, trifluoroacetic anhydride, or trifluoroacetic acid under varying conditions. Alternatively, methods involving the reaction of anthranilates with unstable trifluoroacetamides or the cyclization of isatoic anhydride with trifluoroacetic anhydride have been employed. Another reported pathway involves T3P-promoted cascade reactions of anthranilic acid, trifluoroacetic acid, and amines. However, these legacy approaches suffer from severe drawbacks: they often necessitate harsh reaction conditions that degrade sensitive functional groups, require expensive or difficult-to-handle starting materials that need pre-activation, and frequently result in low yields due to side reactions. Furthermore, the substrate scope in these traditional methods is notoriously narrow, failing to accommodate the diverse structural complexity required for modern medicinal chemistry campaigns.
The Novel Approach
In stark contrast, the methodology described in patent CN113045503B represents a paradigm shift towards efficiency and practicality. By employing a palladium-catalyzed carbonylation cascade, this novel approach utilizes trifluoroethylimidoyl chloride and amines as direct starting materials. The reaction proceeds smoothly in an organic solvent, specifically 1,4-dioxane, at a moderate temperature of 110°C. The use of TFBen (1,3,5-tricarboxylic acid phenol ester) as a solid carbon monoxide source is a masterstroke in process safety, eliminating the need for handling toxic CO gas cylinders. This method boasts exceptional substrate compatibility, allowing for the synthesis of various substituted trifluoromethyl quinazolinone compounds simply by modifying the R1 and R2 groups on the starting materials. The operational simplicity, combined with high conversion rates and the ability to scale to gram levels without loss of efficiency, makes this route highly attractive for cost reduction in pharmaceutical intermediate manufacturing.
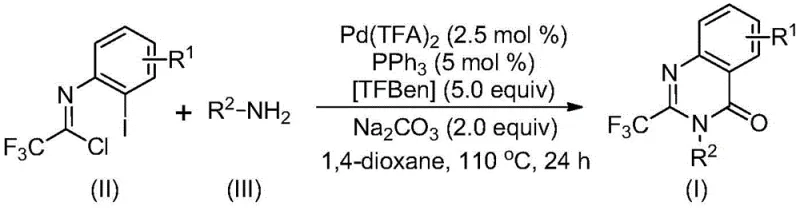
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
Understanding the catalytic cycle is essential for optimizing the production of high-purity pharmaceutical intermediates. The proposed mechanism begins with a base-promoted intermolecular carbon-nitrogen bond coupling between the trifluoroethylimidoyl chloride and the amine, generating a trifluoroacetamidine derivative in situ. Subsequently, the palladium catalyst, specifically palladium trifluoroacetate coordinated with triphenylphosphine, undergoes oxidative addition into the carbon-iodine bond of the aromatic ring, forming a divalent palladium intermediate. At this critical juncture, TFBen decomposes under heating to release carbon monoxide, which then inserts into the carbon-palladium bond to form an acyl palladium species. Under the influence of the base (sodium carbonate), the nitrogen atom coordinates to the palladium center, facilitating the formation of a seven-membered ring palladium intermediate. The cycle concludes with a reductive elimination step that releases the final 2-trifluoromethyl-substituted quinazolinone product and regenerates the active palladium catalyst. This intricate dance of organometallic steps ensures high atom economy and minimizes the formation of polymeric byproducts often seen in radical-based cyclizations.
From an impurity control perspective, the choice of ligands and the precise molar ratios of the catalyst system are vital. The patent specifies a molar ratio of palladium trifluoroacetate to triphenylphosphine to sodium carbonate of 0.025:0.05:2. Deviating from these optimized parameters can lead to incomplete conversion or the formation of dehalogenated side products. The use of aprotic solvents like 1,4-dioxane is also mechanistically significant, as they effectively dissolve the organic substrates while stabilizing the charged intermediates without interfering with the coordination sphere of the palladium. This level of mechanistic control allows for the commercial scale-up of complex pharmaceutical intermediates with consistent quality, ensuring that the final API meets rigorous regulatory standards for residual metals and organic impurities.
How to Synthesize 2-Trifluoromethyl Quinazolinones Efficiently
The execution of this synthesis requires careful attention to reagent quality and reaction monitoring to maximize yield. The process is designed to be robust, utilizing commercially available reagents that do not require specialized storage conditions beyond standard laboratory practices. The reaction time is optimized between 16 to 30 hours; extending beyond this window increases energy costs without significant gains in conversion, while shorter times risk incomplete reaction. Post-reaction processing is straightforward, involving filtration to remove inorganic salts followed by silica gel treatment and column chromatography. For detailed operational parameters, stoichiometry, and specific purification protocols, please refer to the standardized guide below.
- Combine palladium trifluoroacetate, triphenylphosphine, TFBen, sodium carbonate, trifluoroethylimidoyl chloride, and amine in 1,4-dioxane.
- Heat the reaction mixture to 110°C and stir for 16 to 30 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target quinazolinone compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material supply chain. Traditional methods often rely on custom-synthesized, unstable, or hazardous reagents that create bottlenecks in production scheduling. In contrast, this method utilizes trifluoroethylimidoyl chloride and various amines, which are either commodity chemicals or easily synthesized from abundant aromatic amines. This shift significantly enhances supply chain reliability by reducing dependency on niche suppliers and mitigating the risk of raw material shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is profound. By eliminating the need for high-pressure carbon monoxide gas infrastructure, capital expenditure for reactor setup is significantly lowered. Furthermore, the use of a solid CO surrogate (TFBen) improves safety profiles, potentially lowering insurance and compliance costs associated with hazardous gas handling. The high yields reported (ranging from 74% to 98% across various substrates) mean less waste generation and lower cost per kilogram of the final product. The catalyst loading is relatively low (2.5 mol %), and the ligand (triphenylphosphine) is inexpensive, contributing to a favorable overall cost structure for large-scale production.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions translates directly to supply security. The tolerance for diverse functional groups means that a single platform technology can produce a library of different intermediates without requiring extensive re-optimization for each new analog. This flexibility allows manufacturers to respond rapidly to changing market demands or clinical trial requirements. Additionally, the reaction can be performed in standard glass-lined or stainless steel reactors without the need for exotic materials of construction, ensuring that production capacity can be easily allocated across existing manufacturing suites without dedicated line investment.
- Scalability and Environmental Compliance: Environmental, Health, and Safety (EHS) considerations are increasingly critical in the fine chemical sector. This method generates fewer hazardous byproducts compared to anhydride-based cyclizations which produce stoichiometric amounts of acidic waste. The workup procedure is simple, involving filtration and chromatography, which reduces the volume of solvent waste compared to complex extraction sequences. The ability to scale this reaction from milligram to gram levels, as demonstrated in the patent examples including the synthesis of Rutaecarpine, indicates a clear path to multi-kilogram and ton-scale production. This scalability ensures that the supply of these critical intermediates can grow in lockstep with the commercial success of the downstream drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of adopting this route for your specific project needs.
Q: What are the advantages of using TFBen over carbon monoxide gas?
A: TFBen acts as a solid carbon monoxide surrogate, eliminating the need for high-pressure gas equipment and significantly improving operational safety and scalability in standard laboratory or plant settings.
Q: What is the substrate scope for this quinazolinone synthesis?
A: The method demonstrates excellent compatibility with various functional groups, including halogens (F, Cl, Br), alkyl groups, and trifluoromethyl groups on the aromatic ring, as well as diverse amine substrates.
Q: Can this method be applied to complex drug molecules like Rutaecarpine?
A: Yes, the patent explicitly demonstrates the successful application of this methodology in the efficient total synthesis of Rutaecarpine with a high overall yield of 77%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Quinazolinone Supplier
The technological potential of this palladium-catalyzed carbonylation route is immense, offering a streamlined path to valuable heterocyclic building blocks. At NINGBO INNO PHARMCHEM, we pride ourselves on being at the forefront of adopting such innovative synthetic methodologies to serve our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs capable of detecting trace impurities, ensuring that every batch of 2-trifluoromethyl quinazolinone we deliver meets stringent purity specifications required by top-tier pharmaceutical companies.
We invite you to leverage our expertise to optimize your supply chain and reduce your overall development costs. Whether you are in the early stages of drug discovery or preparing for commercial launch, our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary targets. Let us collaborate to bring your next breakthrough therapy to market faster and more efficiently.
