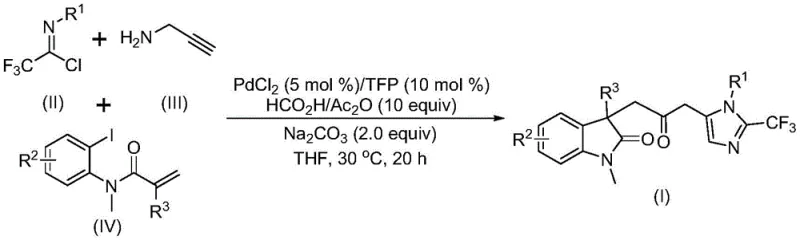
Advanced Pd-Catalyzed Multi-Component Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds efficiently. Patent CN115353511A introduces a groundbreaking multi-component approach for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technology represents a significant leap forward in organic synthesis by replacing hazardous gaseous reagents with safe, liquid surrogates while maintaining high atom economy. For R&D directors and process chemists, the ability to install a carbonyl bridge alongside a trifluoromethyl group in a single operational step offers unparalleled versatility for drug discovery programs. The method leverages a palladium-catalyzed cascade that tolerates a wide range of functional groups, ensuring that diverse molecular libraries can be accessed without extensive protecting group strategies. As a reliable pharmaceutical intermediate supplier, understanding such innovations is critical for maintaining a competitive edge in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of carbonyl-bridged biheterocycles has been fraught with significant operational challenges and safety hazards. Conventional carbonylation reactions typically rely on the direct use of carbon monoxide gas, which is highly toxic and requires specialized high-pressure equipment and rigorous safety protocols to handle effectively. Furthermore, existing synthetic routes often involve multi-step sequences where the heterocyclic rings are constructed separately and then coupled, leading to lower overall yields and increased waste generation. These stepwise approaches not only inflate the cost of goods but also extend the lead time for producing high-purity intermediates, creating bottlenecks in the supply chain. Additionally, many traditional methods suffer from poor substrate compatibility, failing to accommodate sensitive functional groups that are increasingly common in modern drug candidates. The reliance on harsh conditions and expensive, specialized reagents further limits the scalability of these processes, making them less attractive for commercial manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN115353511A utilizes a sophisticated yet operationally simple multi-component reaction strategy that circumvents these historical limitations. By employing a formic acid and acetic anhydride mixture as an in situ carbon monoxide source, the process eliminates the need for handling toxic CO gas, thereby drastically simplifying the reactor setup and safety requirements. This novel approach enables the simultaneous formation of multiple chemical bonds—including carbon-carbon and carbon-nitrogen bonds—in a single pot, significantly enhancing step economy. The reaction proceeds under remarkably mild conditions, typically at 30°C, which preserves the integrity of sensitive functional groups and reduces energy consumption. The use of readily available starting materials such as trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives ensures that the process is both cost-effective and scalable. This paradigm shift allows for the rapid assembly of complex biheterocyclic architectures with high efficiency.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation is a testament to the elegance of modern transition metal catalysis, involving a intricate sequence of organometallic steps initiated by the palladium catalyst. The cycle likely begins with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive aryl-palladium intermediate. This is followed by an intramolecular Heck-type reaction or migratory insertion that sets the stage for ring closure. Crucially, the carbon monoxide generated from the decomposition of the formic acid/acetic anhydride mixture inserts into the palladium-carbon bond to form an acyl-palladium species. This acyl intermediate is pivotal, as it serves as the electrophilic center for the subsequent nucleophilic attack. Concurrently, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine generates a trifluoroacetamidine species in situ. The activation of this amidine by the acyl-palladium complex triggers a final cyclization event, forging the imidazole ring and completing the carbonyl-bridged biheterocyclic framework. This cascade mechanism ensures high regioselectivity and minimizes the formation of side products.
From an impurity control perspective, the mild reaction temperature of 30°C plays a vital role in suppressing thermal degradation pathways that often plague high-temperature syntheses. The specific choice of ligands, such as trifurylphosphine (TFP), stabilizes the palladium center and accelerates the turnover frequency, preventing the accumulation of inactive palladium black which can act as a sink for reactants. Furthermore, the stoichiometric balance of the reagents, particularly the excess of propargylamine and acrylamide relative to the imidoyl chloride, drives the equilibrium towards the desired product, minimizing unreacted starting materials. The use of sodium carbonate as a mild base ensures that acid-sensitive groups remain intact while effectively neutralizing the hydrochloric acid byproduct generated during the amidine formation. Post-reaction processing involves standard filtration and silica gel chromatography, which effectively removes palladium residues and inorganic salts, yielding high-purity products suitable for downstream pharmaceutical applications without the need for complex purification protocols.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
To implement this synthesis effectively, precise control over reaction parameters and reagent quality is essential to maximize yield and reproducibility. The protocol dictates the use of anhydrous tetrahydrofuran (THF) as the preferred solvent due to its ability to solubilize all organic components while supporting the catalytic cycle. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below to ensure consistent results across different batches.
- Combine palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source mixture in an organic solvent such as THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction vessel under stirring.
- Maintain the reaction mixture at 30°C for 12 to 20 hours to allow the carbonylation cascade to complete, followed by filtration and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The elimination of toxic carbon monoxide gas from the process inventory removes a major regulatory and safety burden, allowing facilities to operate with reduced insurance premiums and simplified compliance reporting. This shift inherently lowers the barrier to entry for manufacturing these complex intermediates, enabling more flexible production scheduling and reducing the risk of shutdowns due to safety audits. Moreover, the reliance on commodity chemicals like formic acid and acetic anhydride as CO surrogates insulates the supply chain from the volatility associated with specialty gas markets. The mild reaction conditions translate directly into lower energy costs, as there is no need for extensive heating or cooling infrastructure, contributing to a greener and more sustainable manufacturing footprint. These factors collectively enhance the reliability of supply for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the synthetic route and the use of inexpensive catalysts. By consolidating multiple bond-forming events into a single step, the process drastically reduces labor hours, solvent usage, and waste disposal costs associated with multi-step syntheses. The use of palladium chloride, a relatively inexpensive palladium source compared to exotic complexes, combined with efficient ligand loading, optimizes the catalyst cost per kilogram of product. Furthermore, the high conversion rates observed under these mild conditions minimize the loss of valuable starting materials, ensuring that the theoretical yield is closely approached in practice. This efficiency allows for significant margin improvement when scaling up to commercial volumes, making the final API more cost-competitive in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures a stable and continuous supply of high-quality intermediates. Since the starting materials are commercially available and do not require custom synthesis or long lead times, procurement teams can maintain leaner inventory levels without risking production stoppages. The tolerance of the reaction to various functional groups means that a single platform technology can be adapted to produce a wide library of analogues, providing flexibility to respond quickly to changing market demands or clinical trial requirements. This adaptability reduces the dependency on single-source suppliers for niche precursors, thereby mitigating supply chain risks. The ability to scale from gram to kilogram quantities without re-optimizing the core reaction parameters further guarantees that supply can meet demand as a drug candidate progresses through development stages.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this methodology is inherently designed for expansion. The absence of high-pressure gas handling simplifies the engineering requirements for larger reactors, allowing for straightforward technology transfer from pilot plant to full-scale production. The use of THF as a solvent, which is easily recoverable and recyclable, aligns with modern green chemistry principles and facilitates waste stream management. Additionally, the low operating temperature reduces the thermal load on facility utilities, contributing to a lower carbon footprint. The process generates minimal hazardous waste compared to traditional methods, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. This environmental stewardship not only protects the ecosystem but also enhances the corporate reputation of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for potential partners and stakeholders.
Q: What is the primary safety advantage of this synthesis method compared to traditional carbonylation?
A: Unlike conventional methods that require handling toxic carbon monoxide gas cylinders, this patent utilizes a formic acid and acetic anhydride mixture to generate CO in situ, significantly reducing safety risks and infrastructure costs.
Q: What represents the key structural motif formed in this reaction?
A: The reaction constructs a complex carbonyl-bridged biheterocyclic scaffold, specifically linking an indolinone core with a trifluoromethyl-substituted imidazole ring, which is highly valued in medicinal chemistry.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates scalability to the gram level with mild reaction conditions (30°C) and commercially available starting materials, indicating strong potential for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN115353511A for accelerating drug development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to industrial manufacturing is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in palladium-catalyzed cascades allows us to optimize these reactions for maximum yield and minimal impurity profiles, guaranteeing the quality required for GMP environments.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive value and efficiency in your supply chain. Let us be your partner in turning complex chemical challenges into commercial successes.
