Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial API Manufacturing
Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial API Manufacturing
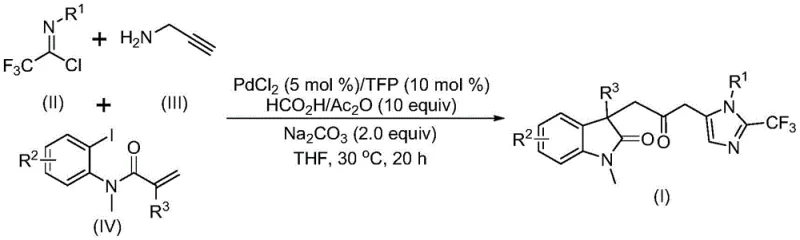
The rapidly evolving landscape of pharmaceutical research demands increasingly sophisticated molecular scaffolds capable of addressing complex biological targets. In this context, patent CN115353511A introduces a groundbreaking multi-component methodology for the efficient construction of carbonyl-bridged biheterocyclic compounds. These structures, particularly those integrating indolinone and imidazole motifs, are highly prized in medicinal chemistry for their potent biological activities and metabolic stability. The disclosed technology represents a significant leap forward in synthetic organic chemistry, replacing hazardous traditional protocols with a streamlined, one-pot catalytic system. By leveraging a palladium-catalyzed cascade reaction that utilizes inexpensive and readily available starting materials, this innovation offers a robust pathway for generating diverse libraries of fluorinated heterocycles. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediate supplier, this method provides a compelling solution that balances high purity standards with operational safety and cost-effectiveness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biheterocyclic compounds containing carbonyl bridges has been fraught with significant technical and safety challenges that hinder efficient commercial production. Traditional approaches often rely on the direct coupling of two pre-formed heterocyclic substrates, a strategy that frequently suffers from low atom economy and requires harsh reaction conditions to drive the formation of the critical carbon-carbon or carbon-nitrogen bonds. Furthermore, alternative oxidative cyclization methods involving activated methyl-substituted heterocycles often necessitate the use of stoichiometric oxidants, leading to substantial waste generation and complicated downstream purification processes. Perhaps most critically, classical carbonylation reactions typically depend on the use of pressurized carbon monoxide gas, which poses severe toxicity risks and requires specialized, expensive high-pressure equipment to handle safely. These factors collectively contribute to extended lead times for high-purity pharmaceutical intermediates and inflate the overall cost of goods, making it difficult for manufacturers to scale these processes economically.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent utilizes a transition metal palladium-catalyzed carbonylation cascade that operates under remarkably mild conditions. This innovative strategy employs a three-component coupling of trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, effectively constructing the complex biheterocyclic core in a single operational step. A defining feature of this methodology is the replacement of toxic carbon monoxide gas with a safe, in-situ CO source generated from a mixture of formic acid and acetic anhydride. This modification not only eliminates the need for high-pressure reactors but also allows the reaction to proceed efficiently at a moderate temperature of 30°C. The result is a process that offers superior substrate compatibility, enabling the incorporation of various functional groups such as halogens, alkyls, and electron-withdrawing groups without compromising yield. This level of versatility makes it an ideal candidate for cost reduction in API manufacturing, as it simplifies the supply chain by utilizing commodity chemicals.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
Understanding the mechanistic underpinnings of this transformation is essential for R&D teams aiming to optimize the synthesis of complex pharmaceutical intermediates. The reaction is believed to initiate with the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, generating a reactive organopalladium intermediate. This species subsequently undergoes an intramolecular Heck-type reaction to form a divalent alkyl-palladium complex, setting the stage for the crucial carbonylation step. The carbon monoxide required for this insertion is released gently from the formic acid and acetic anhydride mixture, reacting with the alkyl-palladium intermediate to form an acyl-palladium species. Concurrently, the trifluoroethylimidoyl chloride and propargylamine undergo a base-promoted intermolecular carbon-nitrogen bond formation to yield a trifluoroacetamidine compound, which then isomerizes. The final ring-closing event is catalyzed by the activation of this trifluoroacetamidine by the acyl-palladium intermediate, resulting in the formation of the stable carbonyl-bridged biheterocyclic framework.
From an impurity control perspective, this mechanism offers distinct advantages over stepwise syntheses. By consolidating multiple bond-forming events into a single pot, the exposure of reactive intermediates to the bulk environment is minimized, thereby reducing the formation of side products associated with intermediate isolation and handling. The use of a specific ligand system, such as trifurylphosphine (TFP), alongside palladium chloride, ensures high catalytic turnover and selectivity, which is critical for maintaining a clean impurity profile. The mild reaction temperature of 30°C further suppresses thermal degradation pathways that often plague high-temperature cyclizations. For quality assurance teams, this translates to a more predictable impurity spectrum that is easier to characterize and control during the scale-up phase, ensuring that the final high-purity OLED material or pharmaceutical intermediate meets stringent regulatory specifications.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
The practical execution of this synthesis is designed to be straightforward, minimizing the need for specialized equipment while maximizing yield and reproducibility. The process begins by charging a reaction vessel with the palladium catalyst, ligand, base, and the CO-generating mixture in an aprotic organic solvent like tetrahydrofuran. Once the catalytic system is established, the three primary substrates are introduced, and the mixture is stirred at ambient to slightly elevated temperatures. The reaction progress is monitored until completion, typically within a 12 to 20-hour window, after which standard workup procedures involving filtration and silica gel treatment are employed. Detailed standardized synthesis steps see the guide below.
- Combine palladium chloride catalyst, trifurylphosphine ligand, sodium carbonate base, and the formic acid/acetic anhydride CO source mixture in an organic solvent such as THF.
- Add the three key substrates: trifluoroethylimidoyl chloride, propargylamine, and the specific acrylamide derivative to the reaction vessel under stirring.
- Maintain the reaction mixture at 30°C for 12 to 20 hours, followed by filtration, silica gel mixing, and column chromatography purification to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The shift away from hazardous gases and high-pressure equipment fundamentally alters the risk profile of the manufacturing process, allowing for production in facilities that may not be equipped for handling toxic carbon monoxide. This flexibility significantly enhances supply chain reliability by broadening the pool of eligible contract manufacturing organizations (CMOs) capable of executing the synthesis. Furthermore, the reliance on cheap and commercially available starting materials such as propargylamine and acrylamide derivatives ensures a stable supply of raw inputs, mitigating the risk of shortages that often plague specialty chemical markets. The simplicity of the one-pot procedure also drastically reduces labor hours and utility consumption, contributing to substantial cost savings in the overall production budget.
- Cost Reduction in Manufacturing: The elimination of expensive high-pressure reactors and the associated safety infrastructure required for handling carbon monoxide gas leads to a significant decrease in capital expenditure and operational overhead. Additionally, the use of inexpensive commodity chemicals as starting materials, combined with the high atom efficiency of the multi-component reaction, minimizes raw material costs. The mild reaction conditions further reduce energy consumption compared to traditional high-temperature cyclizations, resulting in a leaner and more cost-effective manufacturing process that improves margin potential for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: By utilizing a robust catalytic system that tolerates a wide range of functional groups, manufacturers can source diverse substrates without being constrained by narrow specification requirements. The ability to perform the reaction at 30°C in common solvents like THF ensures that the process is not dependent on exotic reagents or extreme conditions that could disrupt logistics. This operational resilience translates to shorter lead times for high-purity pharmaceutical intermediates, allowing companies to respond more agilely to market demands and maintain continuous production schedules even in volatile supply environments.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the feasibility of expanding this reaction to gram-scale quantities, indicating a clear path toward kilogram and ton-scale commercial production. The avoidance of toxic gas emissions and the reduction of hazardous waste streams align with increasingly strict environmental regulations, simplifying the permitting process for new manufacturing lines. The straightforward post-treatment involving filtration and column chromatography is easily adaptable to industrial purification methods like crystallization or preparative HPLC, ensuring that the process remains scalable without sacrificing product quality or environmental compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and mechanistic proposals outlined in the patent documentation, providing clarity for technical teams evaluating this route for their own pipelines. Understanding these nuances is critical for assessing the feasibility of integrating this chemistry into existing manufacturing workflows.
Q: How does this method address the safety concerns of traditional carbonylation reactions?
A: Unlike conventional methods that require high-pressure cylinders of toxic carbon monoxide gas, this patent utilizes a formic acid and acetic anhydride mixture to generate CO in-situ under mild conditions, significantly enhancing operational safety.
Q: What is the substrate compatibility for the R1 and R3 positions in this synthesis?
A: The method demonstrates broad functional group tolerance, successfully accommodating substituents such as methyl, tert-butyl, methoxy, chloro, bromo, trifluoromethyl, and nitro groups on the aromatic rings without compromising reaction efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states that the method has been expanded to gram-scale reactions with high conversion rates, indicating strong potential for commercial scale-up due to its simple operation and inexpensive raw materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
As the demand for complex heterocyclic scaffolds continues to grow in the development of next-generation therapeutics, having a partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions with precision, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that the transition from bench-scale discovery to commercial supply requires not just chemical knowledge but also a commitment to quality and consistency that few can match.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, helping you identify the most efficient route to market. Please contact us today to request specific COA data and route feasibility assessments for your target molecules, and let us demonstrate how our expertise can accelerate your development timeline while optimizing your supply chain costs.
