Advanced Photocatalytic Synthesis of Chlorocyclopentanone Derivatives for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex cyclic scaffolds, particularly for bioactive natural products like aromatic sesquiterpenes. A groundbreaking development in this field is detailed in patent CN114920640A, which discloses a novel method for synthesizing chlorocyclopentanone derivatives via a photocatalytic [3+2] cycloaddition reaction. This technology represents a paradigm shift from traditional multi-step organometallic syntheses to a direct, atom-economical approach utilizing visible light catalysis. By employing tetrabutylammonium decatungstate (TBADT) as a photocatalyst, the process converts readily available 3-methylbutanal and p-methylphenylacetylene chloride into high-value intermediates with exceptional efficiency. For R&D directors and procurement specialists, this innovation offers a compelling solution for reducing the cost of goods sold (COGS) while enhancing the sustainability profile of complex intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
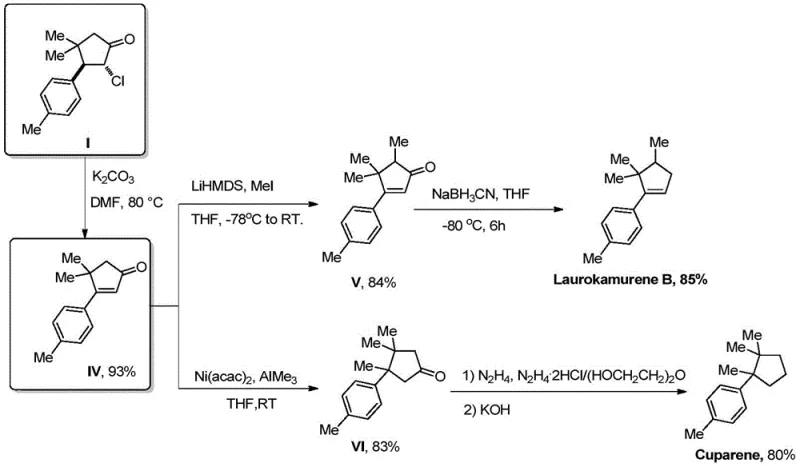
Historically, the synthesis of aromatic sesquiterpenes such as Laurokamurene B and Cuparene has been plagued by inefficient, linear synthetic routes that rely heavily on precious metal catalysis and harsh reaction conditions. Prior art, such as the work by Srikrishna et al., required up to 10 steps to achieve the target molecules, resulting in a dismal overall yield of approximately 22%. Other approaches utilized Stork–Danheiser reactions or palladium-catalyzed Heck cyclizations, which necessitate the pre-functionalization of substrates. These conventional methods often involve expensive reagents like Grignard reagents or organolithium compounds, which demand strict anhydrous conditions and cryogenic temperatures, thereby inflating operational costs and complicating safety protocols. Furthermore, the reliance on transition metals like palladium introduces significant challenges in meeting stringent residual metal specifications required for pharmaceutical applications, often necessitating costly purification steps that erode profit margins.
The Novel Approach
In stark contrast, the methodology outlined in patent CN114920640A leverages the power of hydrogen atom transfer (HAT) photocatalysis to construct the cyclopentanone core in a single step with 100% atom economy. This novel approach bypasses the need for substrate pre-functionalization and expensive transition metal catalysts, utilizing instead a robust tungsten-based photocatalyst activated by low-energy LEDs. The reaction proceeds under mild conditions—specifically at room temperature (25°C) in a benign acetonitrile-water solvent system—drastically reducing energy consumption and safety risks associated with high-pressure or high-temperature reactors. By streamlining the synthesis of the key chlorocyclopentanone intermediate, this technology enables a rapid two-step conversion to final targets like Laurokamurene B, offering a substantial reduction in lead time and a simplified supply chain for high-purity aromatic sesquiterpenes.
Mechanistic Insights into TBADT-Catalyzed [3+2] Cycloaddition
The core of this technological breakthrough lies in the unique mechanistic cycle driven by the TBADT photocatalyst under 390nm LED irradiation. The reaction initiates with the photoexcitation of the decatungstate anion, which acts as a potent hydrogen atom abstractor. It selectively activates the formyl C-H bond of the 3-methylbutanal synthon, generating a nucleophilic acyl radical. This radical species then undergoes a conjugate addition to the electron-deficient alkyne moiety of the p-methylphenylacetylene chloride. Following this addition, a critical 1,5-hydrogen atom transfer occurs, relocating the radical center to a position favorable for ring closure. The cycle culminates in an anti-Baldwin 5-endo-trig cyclization, followed by a final hydrogen abstraction to regenerate the catalyst and release the chlorocyclopentanone product. This intricate radical cascade is highly controlled, ensuring that side reactions are minimized and the desired skeletal framework is constructed with precision.
From a quality control perspective, the mechanism inherently favors the formation of a single diastereomer, achieving a diastereomeric ratio (dr) greater than 20:1. This high level of stereocontrol is crucial for R&D teams aiming to minimize impurity profiles in downstream processing. The use of a chlorine-containing alkyne not only serves as a coupling partner but also installs a functional handle that can be subsequently eliminated to form the cyclopentenone double bond. This strategic design eliminates the need for separate oxidation or dehydrogenation steps typically required in classical enone synthesis. The robustness of the TBADT catalyst ensures consistent performance over prolonged irradiation times (15 hours), providing a reliable platform for scaling up the production of these complex intermediates without the variability often seen in sensitive organometallic couplings.
![Photocatalytic [3+2] cycloaddition reaction mechanism using TBADT](/insights/img/chlorocyclopentanone-photocatalysis-pharma-supplier-20260306180912-06.webp)
How to Synthesize Chlorocyclopentanone Derivative I Efficiently
The practical implementation of this synthesis is designed for operational simplicity, making it accessible for both laboratory optimization and industrial manufacturing. The process begins by combining the aldehyde and alkyne precursors with a catalytic loading of TBADT in a mixed solvent system, followed by irradiation with standard LED arrays. The mild nature of the reaction allows for the use of standard glassware and avoids the need for specialized high-pressure equipment. After the reaction reaches completion, a straightforward aqueous workup and silica gel chromatography yield the pure chlorocyclopentanone derivative. This intermediate serves as a versatile platform; subsequent treatment with potassium carbonate facilitates an elimination reaction to generate the cyclopentenone scaffold, which can then be elaborated into various bioactive targets. The detailed standardized synthesis steps for this process are provided in the guide below.
- Combine 3-methylbutanal and p-methylphenylacetylene chloride with TBADT catalyst in a MeCN/H2O solvent system under nitrogen.
- Irradiate the reaction mixture with 390nm LEDs at room temperature for approximately 15 hours to drive the [3+2] cycloaddition.
- Quench with water, extract with ethyl acetate, and purify via silica gel column chromatography to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits that extend beyond simple yield improvements. By replacing multi-step sequences involving precious metals with a single catalytic cycle, manufacturers can significantly reduce the complexity of their supply chains. The reliance on commodity chemicals like 3-methylbutanal and simple alkynes mitigates the risk of raw material shortages that often plague specialized organometallic reagents. Furthermore, the elimination of heavy metal catalysts such as palladium or nickel removes the burden of expensive scavenging processes and rigorous metal testing, directly lowering the cost of quality assurance. This streamlined workflow enhances overall manufacturing agility, allowing for faster response times to market demands for key pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The transition to a photocatalytic protocol eliminates the need for stoichiometric amounts of expensive organolithium or Grignard reagents, which are not only costly but also hazardous to handle on a large scale. By utilizing a catalytic amount of TBADT and visible light, the process drastically reduces reagent costs and energy consumption associated with cryogenic cooling. Additionally, the high atom economy of the [3+2] cycloaddition minimizes waste generation, leading to lower disposal costs and a smaller environmental footprint. These factors combine to create a highly cost-effective manufacturing route that improves the overall margin profile for fine chemical production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis are bulk commodity chemicals with stable global supply chains, reducing the vulnerability to geopolitical disruptions or supplier bottlenecks common with specialized catalysts. The robustness of the reaction conditions—operating at ambient temperature and pressure—further ensures consistent production uptime, as the process is less susceptible to equipment failures or utility fluctuations compared to high-pressure hydrogenation or cryogenic reactions. This reliability is critical for maintaining continuous supply to downstream API manufacturers, ensuring that project timelines are met without unexpected delays caused by synthesis failures.
- Scalability and Environmental Compliance: Photochemical reactions have historically faced challenges in scale-up due to light penetration issues, but modern flow chemistry and high-surface-area LED reactors have largely solved this, making this technology ready for commercial deployment. The use of water as a co-solvent and the absence of toxic heavy metals align perfectly with increasingly stringent environmental regulations and green chemistry principles. This compliance reduces the regulatory burden on manufacturing sites and facilitates easier permitting for capacity expansion. The ability to scale from grams to tons while maintaining high selectivity ensures that this route is viable for long-term commercial supply of complex intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the primary advantages of this photocatalytic route over traditional methods?
A: This method utilizes inexpensive aldehyde synthons and achieves 100% atom economy, significantly reducing waste and raw material costs compared to multi-step organometallic routes.
Q: Is the TBADT catalyst suitable for large-scale production?
A: Yes, TBADT is a robust polyoxometalate that operates under mild conditions with visible light, making it highly scalable and safer than high-pressure or cryogenic traditional processes.
Q: What is the stereochemical outcome of the cycloaddition?
A: The reaction exhibits excellent diastereoselectivity (dr > 20:1), ensuring high purity of the chlorocyclopentanone intermediate and simplifying downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chlorocyclopentanone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic technologies in modern organic synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the TBADT-catalyzed cycloaddition are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications for complex intermediates. We are committed to delivering high-quality chlorocyclopentanone derivatives and downstream sesquiterpenes that adhere to the highest industry standards, supporting our clients' drug development pipelines with reliable and compliant supply.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your next project. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with superior chemical solutions.
