Scalable Green Synthesis of Bis-Indole Alkyl Compounds for Advanced Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign methodologies for constructing complex heterocyclic scaffolds, particularly bis-indole alkyl compounds which serve as critical precursors for bioactive molecules. Patent CN100412058C introduces a transformative approach to this synthesis by utilizing Meldrum's acid (Michaelis acid) as a highly efficient organocatalyst within an exclusively aqueous reaction medium. This innovation marks a significant departure from conventional protocols that rely heavily on volatile organic compounds (VOCs) and expensive Lewis acid catalysts, addressing both environmental compliance and economic efficiency simultaneously. By shifting the reaction paradigm to water, this technology not only mitigates the ecological footprint associated with solvent disposal but also simplifies the downstream processing through the natural precipitation of the hydrophobic product. For R&D directors and process chemists, this represents a viable pathway to streamline the manufacturing of high-value intermediates while adhering to increasingly stringent global green chemistry regulations. The versatility of this method extends across a broad substrate scope, accommodating both aliphatic and aromatic aldehydes, thereby positioning it as a universal solution for diverse synthetic challenges in the production of pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of bis-indole alkyl compounds has been predominantly achieved through the condensation of indoles with aldehydes or ketones using a variety of catalysts such as Montmorillonite K-10, lithium perchlorate, indium triflate, or iodine. While these traditional methods can achieve acceptable yields, they are fraught with significant operational and economic drawbacks that hinder their scalability and sustainability. The most critical limitation is the absolute dependence on toxic and hazardous organic solvents, which necessitates complex recovery systems, poses severe safety risks regarding flammability and toxicity, and generates substantial waste streams that are costly to treat. Furthermore, many of the effective catalysts employed in these legacy processes, particularly rare metal triflates like In(OTf)3, are prohibitively expensive and subject to supply chain volatility, making the cost of goods sold (COGS) unpredictable and often unsustainable for large-volume commercial production. The reliance on organic media also complicates the isolation of the final product, often requiring energy-intensive distillation or chromatographic purification steps that further erode profit margins and extend lead times for procurement teams.
The Novel Approach
In stark contrast to these established but inefficient practices, the methodology disclosed in CN100412058C leverages the unique properties of Meldrum's acid to catalyze the reaction efficiently in pure water. This novel approach eliminates the need for any organic co-solvents, fundamentally altering the safety and environmental profile of the manufacturing process. The reaction proceeds smoothly at mild temperatures, either under ambient stirring or with the assistance of ultrasonic radiation, which enhances mass transfer and reaction kinetics without the need for extreme thermal input. As illustrated in the reaction scheme below, the condensation of formaldehyde with indole proceeds cleanly to form bis-indolylmethane, demonstrating the efficacy of this aqueous system even for the simplest substrates. The use of water as the sole solvent not only reduces raw material costs drastically but also facilitates a 'green' workup procedure where the product precipitates out of the solution, allowing for straightforward filtration. This shift from organic to aqueous media represents a paradigm shift in process chemistry, offering a reliable pharmaceutical intermediates supplier with a distinct competitive advantage in terms of operational simplicity and regulatory compliance.
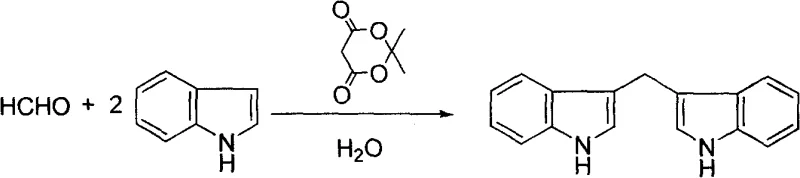
Mechanistic Insights into Meldrum's Acid-Catalyzed Electrophilic Substitution
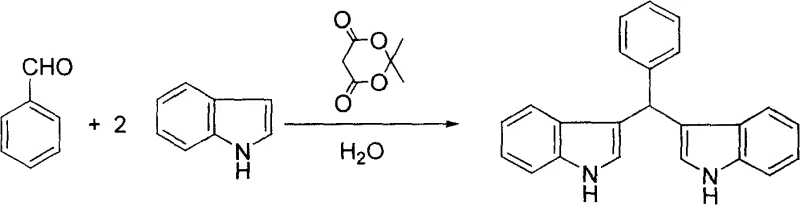
The efficacy of Meldrum's acid in this transformation stems from its ability to act as a mild yet effective Brønsted acid catalyst in an aqueous environment, facilitating the activation of the carbonyl group of the aldehyde towards nucleophilic attack by the indole ring. Unlike strong mineral acids which might lead to polymerization or decomposition of the sensitive indole nucleus, Meldrum's acid provides a controlled acidic environment that promotes the formation of the reactive electrophilic species while maintaining the integrity of the heterocyclic framework. The mechanism likely involves the protonation of the aldehyde oxygen, increasing the electrophilicity of the carbonyl carbon, which is then attacked by the electron-rich C3 position of the indole molecule. Following the initial addition and dehydration steps, a second equivalent of indole attacks the resulting intermediate to form the final bis-indole alkyl structure. This mechanistic pathway is highly tolerant of various functional groups, as evidenced by the successful synthesis of derivatives using aromatic aldehydes such as benzaldehyde. The reaction scheme for the synthesis of bis-indolephenylmethane highlights the robustness of this catalytic system when dealing with sterically and electronically diverse aromatic substrates, ensuring high purity and minimal byproduct formation which is crucial for downstream pharmaceutical applications.
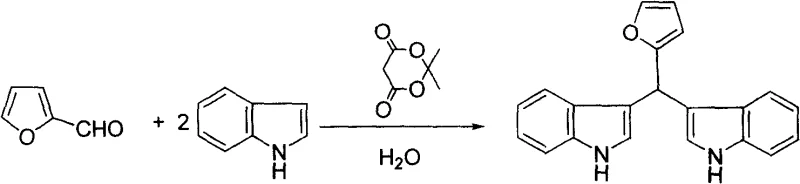
Furthermore, the impurity profile of the reactions conducted under these conditions is exceptionally clean, primarily due to the high selectivity of the catalyst and the precipitating nature of the product in water. The hydrophobic effect in the aqueous phase drives the association of the organic reactants and the subsequent product, effectively shielding the reaction center from water interference while promoting the exclusion of polar impurities that remain dissolved in the mother liquor. This self-purification mechanism is particularly advantageous when synthesizing complex heterocyclic derivatives, such as those derived from furfural, where side reactions are common in harsher acidic media. The successful application of this method to furfural, as shown in the corresponding reaction diagram, underscores the mildness of the catalytic system, preserving the furan ring which is susceptible to acid-catalyzed ring opening or polymerization under more vigorous conditions. For quality control laboratories, this translates to simpler analytical profiles and reduced burden on purification resources, allowing for the rapid release of high-purity batches that meet stringent specifications for active pharmaceutical ingredient (API) synthesis.
How to Synthesize Bis-Indole Alkyl Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of operation, requiring standard reactor equipment without the need for specialized anhydrous conditions or inert gas atmospheres. The process begins with the precise metering of the aldehyde and indole substrates into the reaction vessel, followed by the addition of water and the catalytic amount of Meldrum's acid. Detailed standardized operating procedures regarding specific stoichiometric ratios, mixing speeds, and ultrasonic parameters are critical for maximizing yield and reproducibility on a commercial scale. Operators must ensure that the reaction mixture is homogenized effectively to maximize the interfacial contact between the organic substrates and the aqueous catalyst phase, especially when dealing with liquid aldehydes that may form separate layers initially. The reaction progress is typically monitored by the appearance of the solid precipitate, which serves as a visual indicator of conversion, although HPLC or TLC analysis is recommended for precise endpoint determination to ensure complete consumption of the starting materials. Once the reaction is deemed complete, the isolation step is remarkably straightforward, involving simple suction filtration followed by washing with water to remove residual catalyst and salts, yielding a crude product that often requires minimal further purification to reach commercial grade purity standards.
- Prepare the reaction mixture by combining the selected aldehyde (C1-C10 aliphatic or aromatic) and indole in a molar ratio of 1: 2 within a reaction vessel.
- Add water as the solvent (2-6 liters per mole of aldehyde) and introduce Meldrum's acid as the catalyst at a loading of 1-5 mol% relative to the aldehyde.
- Stir the mixture at room temperature or apply ultrasonic radiation at 30-40°C for 6-10 hours until the solid product precipitates, then isolate via suction filtration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous Meldrum's acid catalyzed process offers profound strategic benefits that extend far beyond simple chemical yield improvements. The most immediate impact is seen in the drastic reduction of raw material costs, driven by the substitution of expensive organic solvents and rare metal catalysts with inexpensive, commodity-grade water and readily available Meldrum's acid. This shift not only lowers the direct material cost per kilogram of the product but also insulates the manufacturing process from the price volatility associated with petrochemical-derived solvents and specialized reagents. Additionally, the elimination of solvent recovery and distillation units significantly reduces capital expenditure (CAPEX) requirements for new production lines and lowers the operational expenditure (OPEX) related to energy consumption and maintenance. From a supply chain resilience perspective, the reliance on universally available reagents ensures continuity of supply even during global disruptions, making this a highly reliable sourcing strategy for long-term contracts.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior to traditional methods because it removes the most cost-intensive components of fine chemical manufacturing: solvent purchase, storage, recovery, and disposal. By operating in water, the facility avoids the regulatory fees and environmental taxes associated with VOC emissions, leading to substantial indirect cost savings that improve the overall margin profile. Furthermore, the catalyst loading is extremely low (1-5 mol%), and since Meldrum's acid is a bulk chemical, the cost contribution of the catalyst to the final product price is negligible compared to indium or other transition metal catalysts. This structural cost advantage allows suppliers to offer more competitive pricing to downstream API manufacturers without compromising on quality or profitability.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions—room temperature or mild heating with ultrasonic assistance—means that the process can be scaled up rapidly using existing multi-purpose reactors without requiring specialized high-pressure or cryogenic equipment. This flexibility reduces the lead time for scaling from pilot plant to commercial production, enabling suppliers to respond quickly to surges in market demand. Moreover, the safety profile of the process is significantly enhanced by the absence of flammable organic solvents, reducing insurance premiums and minimizing the risk of production stoppages due to safety incidents. This reliability is crucial for pharmaceutical customers who require guaranteed supply continuity to maintain their own production schedules and regulatory filings.
- Scalability and Environmental Compliance: As global environmental regulations become increasingly stringent, the ability to manufacture complex intermediates in water provides a future-proof advantage that aligns with corporate sustainability goals. The aqueous waste stream generated by this process is far easier and cheaper to treat than mixed organic waste, simplifying compliance with local environmental protection laws. The scalability of the precipitation-based isolation method is inherently linear; as the batch size increases, the filtration capacity can be easily adjusted without the exponential increase in complexity seen in extraction-based workflows. This makes the commercial scale-up of complex pharmaceutical intermediates not only technically feasible but also economically and environmentally sustainable for large-scale industrial operations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology, providing clarity for stakeholders evaluating its potential for integration into their supply chains. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these nuances is essential for assessing the feasibility of adopting this method for specific product portfolios and for anticipating the operational changes required for successful technology transfer.
Q: What are the primary advantages of using Meldrum's acid over traditional Lewis acids for bis-indole synthesis?
A: Meldrum's acid offers a distinct advantage by enabling the reaction to proceed in an aqueous phase, eliminating the need for toxic organic solvents required by traditional catalysts like InCl3 or Montmorillonite K-10. Furthermore, it is significantly cheaper and easier to source than rare metal triflates, leading to substantial reductions in raw material costs and simplifying the supply chain for large-scale production.
Q: How does the aqueous reaction medium impact the purification process of bis-indole derivatives?
A: The use of water as a solvent leverages the hydrophobic nature of the bis-indole products, causing them to precipitate directly from the reaction mixture as solids upon completion. This phenomenon allows for simple isolation via suction filtration, bypassing complex extraction and solvent evaporation steps typically associated with organic synthesis, thereby drastically reducing processing time and energy consumption.
Q: Is this synthesis method compatible with sensitive heterocyclic aldehydes like furfural?
A: Yes, the patent explicitly demonstrates the compatibility of this method with heterocyclic aromatic aldehydes such as furfural. The mild acidic conditions provided by Meldrum's acid in water prevent the degradation of sensitive heterocyclic rings, ensuring high purity and yield for complex intermediates required in specialized pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-Indole Alkyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the aqueous Meldrum's acid catalyzed synthesis route for producing high-quality bis-indole derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry patent are fully realized in practical, large-scale manufacturing environments. Our state-of-the-art facilities are equipped to handle aqueous phase reactions with precision, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required by the global pharmaceutical industry. We are committed to leveraging this innovative technology to deliver cost-effective, environmentally responsible solutions that empower our clients to bring life-saving medicines to market faster and more efficiently.
We invite forward-thinking procurement leaders and R&D directors to collaborate with us to explore how this advanced synthesis method can optimize your specific supply chain requirements. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume needs, alongside specific COA data and route feasibility assessments for your target molecules. Let us demonstrate how our commitment to innovation and quality can drive value for your organization, ensuring a secure and sustainable supply of critical pharmaceutical intermediates for your future projects.
