Scalable Green Synthesis of Bis-Indole Alkyl Compounds for Commercial Pharmaceutical Production
Scalable Green Synthesis of Bis-Indole Alkyl Compounds for Commercial Pharmaceutical Production
The pharmaceutical and fine chemical industries are constantly seeking robust, environmentally benign pathways to synthesize complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN100412058C introduces a groundbreaking methodology for the synthesis of bis-indole alkyl compounds, utilizing Meldrum's acid as an efficient organocatalyst in an exclusively aqueous medium. This innovation represents a significant departure from conventional protocols that rely heavily on volatile organic compounds (VOCs) and expensive transition metal catalysts. By leveraging the unique reactivity of Meldrum's acid in water, this process not only achieves high conversion rates but also drastically simplifies the workup procedure through direct precipitation of the target molecule. For R&D directors and process chemists, this patent offers a compelling route to access diverse bis-indole derivatives, ranging from simple bis-indolylmethanes to more complex heteroaryl-substituted analogs, with minimal environmental footprint.
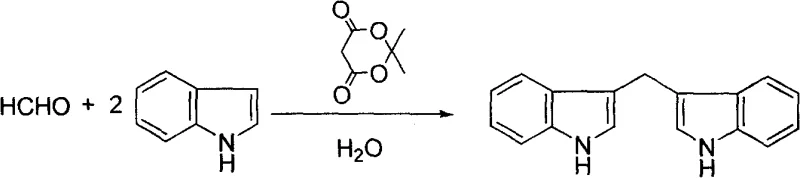
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the electrophilic substitution of indoles with aldehydes to form bis-indole alkyl compounds has been predominantly catalyzed by strong Lewis acids such as Indium(III) chloride, Lithium perchlorate, or solid supports like Montmorillonite K-10. While these traditional methods can provide acceptable yields, they are fraught with significant operational and economic drawbacks that hinder large-scale adoption. The primary limitation lies in the absolute requirement for anhydrous organic solvents such as dichloromethane, acetonitrile, or tetrahydrofuran to maintain catalyst activity and solubility. These solvents are not only costly to procure in bulk but also impose a heavy burden on waste management systems due to their toxicity and flammability. Furthermore, the removal of residual metal catalysts from the final product often necessitates complex purification steps, including column chromatography or extensive washing, which erodes overall process efficiency and increases the cost of goods sold (COGS) for high-purity pharmaceutical intermediates.
The Novel Approach
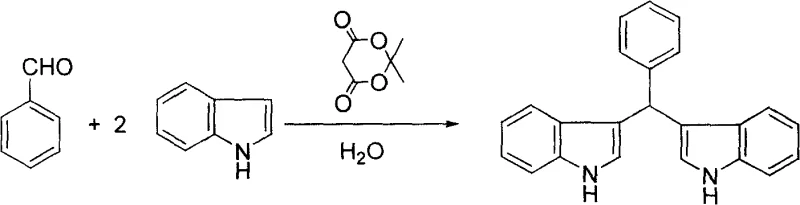
In stark contrast to the solvent-intensive legacy methods, the novel approach detailed in patent CN100412058C utilizes water as the sole reaction medium, driven by the catalytic activity of Meldrum's acid. This shift to an aqueous system fundamentally alters the thermodynamics and kinetics of the reaction, promoting the hydrophobic effect which accelerates the condensation between the aldehyde and the indole nucleophile. The use of Meldrum's acid, a cyclic diester of malonic acid, provides a mild yet effective activation of the carbonyl group without the corrosive nature of mineral acids or the moisture sensitivity of Lewis acids. As illustrated in the reaction schemes below, this method is remarkably versatile, accommodating a wide range of substrates including aromatic aldehydes like benzaldehyde. The result is a streamlined process where the product often precipitates directly from the water phase, allowing for isolation via simple filtration rather than energy-intensive distillation or extraction.
Mechanistic Insights into Meldrum's Acid-Catalyzed Electrophilic Substitution
The efficacy of Meldrum's acid in this transformation can be attributed to its ability to act as a Brønsted acid catalyst through the reversible formation of hydrogen bonds with the carbonyl oxygen of the aldehyde substrate. In the aqueous environment, the catalyst enhances the electrophilicity of the carbonyl carbon, facilitating the initial attack by the electron-rich C3 position of the indole ring. This step generates a reactive indolyl-carbinol intermediate, which subsequently undergoes dehydration to form an electrophilic iminium-like species or a stabilized carbocation. A second equivalent of indole then attacks this activated intermediate to yield the final bis-indole alkyl structure. The presence of water plays a dual role: it serves as a heat sink to manage the exothermic nature of the condensation and acts as a driving force for the reaction equilibrium by sequestering polar byproducts, although in this specific stoichiometry, water is technically a product of the condensation itself, the bulk water solvent ensures the hydrophobic product is pushed out of solution.
From an impurity control perspective, the aqueous mechanism offers distinct advantages over organic phase reactions. In traditional organic solvents, side reactions such as polymerization of the aldehyde or over-alkylation of the indole nitrogen can occur if the acidity is not perfectly tuned. However, the mild acidity of Meldrum's acid in water creates a self-buffering environment that suppresses these degradation pathways. Furthermore, because the bis-indole products are generally insoluble in water, they crystallize or precipitate out of the reaction matrix as they form. This continuous removal of product from the liquid phase prevents retro-reactions and minimizes contact time with potential degradants, resulting in a crude product profile that is significantly cleaner than those obtained from homogeneous organic catalysis. This inherent purity is critical for downstream applications where trace metal contamination or solvent residues are strictly regulated.
How to Synthesize Bis-Indole Alkyl Compounds Efficiently
The operational simplicity of this patented method makes it highly attractive for process development teams aiming to scale up production of bis-indole scaffolds. The protocol requires minimal specialized equipment, relying on standard stirred tank reactors capable of handling aqueous slurries. The key to success lies in maintaining the precise molar ratio of aldehyde to indole at 1:2 and ensuring adequate mixing to overcome mass transfer limitations inherent in heterogeneous aqueous systems. Whether utilizing ambient stirring or ultrasonic irradiation to enhance kinetics, the reaction proceeds to completion within a predictable timeframe of 6 to 10 hours. For a comprehensive understanding of the specific parameters required for different aldehyde substrates, please refer to the detailed standardized synthesis steps provided in the technical guide below.
- Prepare the reaction mixture by combining the selected aldehyde (C1-C10 aliphatic or aromatic) and indole in a molar ratio of 1: 2 within a reaction vessel.
- Add water as the sole solvent (2-6 liters per mole of aldehyde) and introduce Meldrum's acid as the catalyst at a loading of 1-5 mol% relative to the aldehyde.
- Stir the mixture at room temperature or apply ultrasonic radiation at 30-40°C for 6-10 hours until solid precipitation occurs, then filter and purify the crude product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aqueous Meldrum's acid catalyzed process translates into tangible strategic benefits that extend beyond mere technical feasibility. The elimination of hazardous organic solvents removes a major variable from the supply chain risk matrix, reducing dependency on petrochemical-derived solvent markets which are subject to volatile pricing and regulatory restrictions. Additionally, the use of water as a solvent dramatically lowers the fire hazard classification of the manufacturing facility, potentially reducing insurance premiums and easing regulatory compliance burdens associated with VOC emissions. The simplicity of the workup—often requiring only filtration and washing—reduces the demand for skilled labor and complex separation equipment, thereby lowering the overall operational expenditure (OPEX) associated with manufacturing these high-value intermediates.
- Cost Reduction in Manufacturing: The economic argument for this technology is robust, primarily driven by the substitution of expensive, anhydrous organic solvents with inexpensive tap water and the replacement of precious metal catalysts with commodity-grade Meldrum's acid. Traditional methods often incur significant costs related to solvent recovery distillation columns and the disposal of halogenated waste streams; this new method effectively eradicates those line items from the budget. Furthermore, the catalyst loading is exceptionally low, typically ranging from 1% to 5% molar equivalent, which means the raw material cost contribution of the catalyst is negligible even at multi-ton scales. By simplifying the isolation process to a filtration step, the facility also saves substantially on energy consumption that would otherwise be required for solvent evaporation and drying, leading to a leaner, more cost-effective production model.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available raw materials. Indole and various aldehydes are commodity chemicals produced by numerous global suppliers, ensuring that there is no single point of failure in the raw material sourcing strategy. Unlike processes that rely on specialized, moisture-sensitive catalysts that require cold-chain shipping and inert atmosphere storage, Meldrum's acid is a stable solid that can be stored under ambient conditions without degradation. This stability simplifies inventory management and reduces the risk of production stoppages due to reagent spoilage. Moreover, the robustness of the aqueous reaction conditions means that the process is less sensitive to minor fluctuations in raw material quality or environmental humidity, ensuring consistent batch-to-batch output and reliable delivery schedules to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often encounters bottlenecks related to heat transfer and waste management, both of which are elegantly addressed by this water-based technology. Water has a high heat capacity, which facilitates temperature control during the exothermic condensation reaction, making the scale-up from kilograms to tons safer and more predictable. From an environmental compliance standpoint, the absence of toxic organic solvents means the process generates virtually no hazardous organic waste, aligning perfectly with increasingly stringent global environmental regulations such as REACH and TSCA. This 'green' credential not only future-proofs the manufacturing site against regulatory tightening but also enhances the brand value of the final pharmaceutical products by associating them with sustainable manufacturing practices, a key differentiator in modern B2B negotiations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN100412058C, providing a factual basis for evaluating the process suitability for your specific application. Understanding these nuances is essential for making informed decisions about process integration and vendor selection.
Q: What are the primary advantages of using Meldrum's acid over traditional Lewis acid catalysts?
A: Unlike traditional catalysts such as InCl3 or Montmorillonite K-10 which often require toxic organic solvents, Meldrum's acid enables the reaction to proceed efficiently in pure water. This eliminates the need for expensive solvent recovery systems and reduces hazardous waste generation, aligning with modern green chemistry standards.
Q: Can this synthesis method accommodate both aliphatic and aromatic aldehydes?
A: Yes, the patented method demonstrates high versatility, successfully reacting with C1 to C10 aliphatic aldehydes like formaldehyde and nonanal, as well as aromatic and heterocyclic aldehydes such as benzaldehyde and furfural, yielding high-purity bis-indole derivatives in all cases.
Q: How does the aqueous reaction environment impact product isolation?
A: The use of water as a solvent facilitates a unique isolation mechanism where the hydrophobic bis-indole product precipitates directly out of the reaction mixture upon completion. This allows for simple suction filtration to obtain the crude product, significantly simplifying downstream processing compared to extraction-heavy organic protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis-Indole Alkyl Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercially viable supply chain requires deep technical expertise and rigorous quality assurance. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the green advantages of the Meldrum's acid catalyzed process are fully realized at an industrial level. Our state-of-the-art facilities are equipped to handle aqueous chemistries with precision, and our rigorous QC labs enforce stringent purity specifications to guarantee that every batch of bis-indole alkyl compound meets the exacting standards required by the global pharmaceutical industry. We are committed to delivering not just a chemical product, but a reliable, sustainable supply solution.
We invite you to leverage our technical capabilities to optimize your supply chain for these critical intermediates. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how switching to this aqueous methodology can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our current inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us help you secure a competitive advantage through superior chemistry and supply chain reliability.
