Advanced L-Menthol Manufacturing: High-Selectivity Zeolite Catalysis for Commercial Scale-Up
The global demand for high-purity L-menthol continues to surge across pharmaceutical, flavor, and fragrance sectors, driven by its critical role as an active ingredient in analgesics and a premier cooling agent. Patent CN103086845A introduces a transformative methodology for the preparation of L-menthol that addresses long-standing inefficiencies in industrial synthesis. This innovation centers on the utilization of a zinc-bromide-modified NaY molecular sieve as a heterogeneous catalyst, facilitating the ene-cyclization of d-citronellal to isopulegol with exceptional precision. Unlike traditional homogeneous systems that suffer from catalyst recovery issues and moisture sensitivity, this novel approach enables mild reaction conditions ranging from -10 to 30°C while achieving conversion rates approaching 100%. The strategic integration of zeolite support not only stabilizes the active catalytic species but also dramatically simplifies downstream processing, positioning this technology as a cornerstone for reliable pharmaceutical intermediates supplier networks seeking robust and scalable production routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of isopulegol, the key precursor to L-menthol, has relied heavily on the use of anhydrous zinc bromide (ZnBr2) as a Lewis acid catalyst. While chemically effective, this traditional homogeneous catalytic system presents severe operational bottlenecks that hinder cost reduction in fine chemical manufacturing. Anhydrous ZnBr2 is notoriously hygroscopic, absorbing significant moisture from the air during weighing and transportation, which necessitates rigorous anhydrous conditions and specialized handling equipment to maintain catalytic activity. Furthermore, conventional protocols often require stoichiometric or near-stoichiometric loadings of the catalyst, typically around 0.7 equivalents relative to the substrate, leading to substantial material costs and complex waste streams. The difficulty in recovering and recycling the dissolved metal salt from the reaction mixture further exacerbates environmental concerns and increases the overall production burden, making the process less attractive for large-scale commercial operations where efficiency and sustainability are paramount.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages a heterogeneous catalytic system where zinc bromide is chemically modified onto a NaY type molecular sieve support. This architectural shift transforms the reaction dynamics, allowing for catalytic amounts of the active species (weight ratios of 0.2 to 1.0 relative to substrate) to drive the transformation with remarkable efficiency. The solid nature of the catalyst permits simple filtration for separation, enabling the direct reuse of the catalyst in subsequent batches without significant loss of activity, thereby drastically reducing raw material consumption. Moreover, the reaction proceeds under remarkably mild conditions, tolerating temperatures between -10 and 30°C, which lowers energy requirements and enhances safety profiles. This approach effectively eliminates the need for stringent anhydrous environments associated with pure ZnBr2, streamlining the operational workflow and offering a viable pathway for the commercial scale-up of complex fragrance and pharmaceutical intermediates.
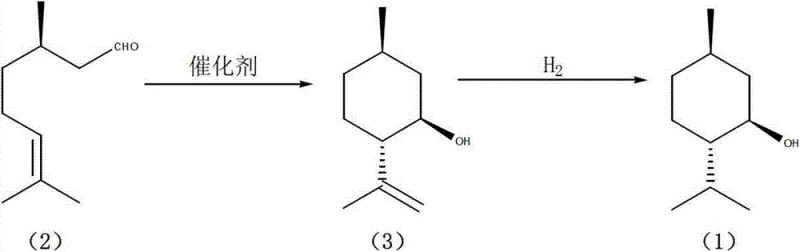
Mechanistic Insights into ZnBr2/NaY Catalyzed Ene-Cyclization
The core of this technological advancement lies in the synergistic interaction between the Lewis acidic zinc species and the porous structure of the NaY molecular sieve. During the ene-cyclization of d-citronellal, the supported zinc bromide acts as a potent electrophile, activating the aldehyde carbonyl group to initiate the intramolecular ring closure. The unique pore structure of the NaY zeolite likely imposes steric constraints that favor the formation of the desired isopulegol stereoisomer over thermodynamic byproducts. This shape-selective catalysis is critical, as it suppresses the formation of the neo-isomer and other high-boiling intermolecular condensation products that typically plague non-selective acid-catalyzed cyclizations. By anchoring the zinc species within the zeolite framework, the catalyst maintains high local concentration and stability, ensuring consistent performance over multiple cycles while preventing the leaching of metal ions that could contaminate the final product.
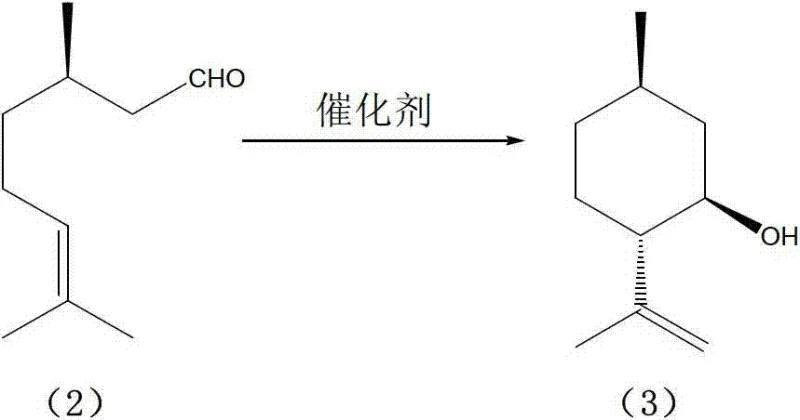
Impurity control is another pivotal aspect where this mechanistic design excels, particularly regarding the suppression of the neo-isopulegol isomer. In conventional liquid-phase acid catalysis, the lack of steric guidance often leads to a mixture of diastereomers, necessitating energy-intensive purification steps like fractional distillation to isolate the target compound. However, the modified NaY catalyst demonstrates exceptional stereoselectivity, yielding isopulegol with purity levels exceeding 99% directly from the reaction mixture. The absence of detectable optical isomers in the product stream indicates that the transition state for the cyclization is highly regulated by the catalyst surface. This high fidelity in stereochemical outcome ensures that the subsequent hydrogenation step produces L-menthol of pharmaceutical grade without the need for intermediate purification, thereby preserving the optical integrity of the molecule throughout the synthesis and minimizing yield losses associated with extensive workup procedures.
How to Synthesize L-Menthol Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this high-efficiency route in a production environment. The process begins with the in-situ or ex-situ preparation of the ZnBr2/NaY catalyst, followed by the cyclization of d-citronellal in a suitable organic solvent such as toluene or tetrahydrofuran. The reaction is conducted under inert atmosphere to prevent oxidative degradation, although the strict anhydrous requirements of traditional methods are relaxed. Upon completion, the solid catalyst is removed via filtration, and the filtrate containing crude isopulegol is subjected directly to high-pressure hydrogenation. This telescoped approach minimizes unit operations and solvent usage, aligning with green chemistry principles. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Prepare the modified catalyst by reacting NaY molecular sieve with a zinc bromide aqueous solution, followed by drying and calcination at 400-600°C.
- React d-citronellal in a solvent such as toluene with the prepared catalyst at mild temperatures (-10 to 30°C) to obtain isopulegol with high stereoselectivity.
- Subject the crude isopulegol directly to high-pressure hydrogenation without intermediate purification to yield the final L-menthol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zeolite-based catalytic system offers profound strategic benefits that extend beyond mere chemical yield. The transition from a homogeneous, moisture-sensitive catalyst to a robust, reusable heterogeneous system fundamentally alters the cost structure of L-menthol production. By eliminating the need for expensive anhydrous reagents and complex catalyst recovery units, manufacturers can achieve significant operational expenditure savings. The ability to recycle the catalyst multiple times without regeneration significantly lowers the cost of goods sold (COGS), providing a competitive edge in pricing negotiations. Furthermore, the simplified workflow reduces the dependency on specialized labor and equipment, enhancing the overall resilience of the supply chain against disruptions.
- Cost Reduction in Manufacturing: The implementation of the ZnBr2/NaY catalyst system drives down manufacturing costs through multiple mechanisms, primarily by reducing catalyst loading from stoichiometric to catalytic levels. Since the solid catalyst can be filtered and reused directly, the recurring expense of purchasing fresh zinc bromide for every batch is virtually eliminated. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which reduces the volume of waste requiring treatment and disposal. The elimination of intermediate purification steps for isopulegol further cuts down on solvent consumption and energy usage associated with distillation, resulting in a leaner and more cost-effective production process that maximizes resource utilization.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly bolstered by the robustness of the new catalytic method, which is less sensitive to environmental variables such as humidity compared to traditional anhydrous ZnBr2 processes. This tolerance simplifies raw material storage and handling logistics, reducing the risk of batch failures due to reagent degradation. The high yield and consistency of the reaction ensure predictable output volumes, allowing for more accurate inventory planning and fulfillment of large-scale orders. Moreover, the use of readily available solvents like toluene and common molecular sieves ensures that the supply of critical inputs remains stable, mitigating the risks associated with sourcing specialized or scarce chemical reagents.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this heterogeneous catalytic process aligns perfectly with modern regulatory standards and sustainable manufacturing goals. The ease of separating the solid catalyst from the liquid product stream minimizes the generation of heavy metal-contaminated wastewater, simplifying effluent treatment and ensuring compliance with strict environmental regulations. The mild reaction conditions reduce the thermal load on reactors, lowering energy consumption and carbon footprint. These factors collectively make the process highly scalable, allowing facilities to ramp up production from pilot to commercial scales with minimal engineering modifications, thereby securing long-term viability and market responsiveness.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this advanced L-menthol synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, aiming to clarify the practical benefits for industrial stakeholders. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines or developing new facilities dedicated to high-value terpene derivatives.
Q: What are the advantages of using ZnBr2/NaY molecular sieve over traditional anhydrous ZnBr2?
A: The ZnBr2/NaY system operates as a heterogeneous catalyst, allowing for simple filtration and reuse, whereas traditional anhydrous ZnBr2 is hygroscopic, difficult to handle, and requires stoichiometric amounts that complicate recovery.
Q: How does this method improve stereoselectivity compared to conventional cyclization?
A: This method achieves yields of 97-100% with negligible formation of the neo-isomer, significantly outperforming traditional methods which often struggle with byproduct formation and lower optical purity.
Q: Is purification required between the cyclization and hydrogenation steps?
A: No, the isopulegol obtained from the cyclization step is of sufficient purity (>99%) to undergo direct high-pressure hydrogenation, eliminating a costly and time-consuming purification stage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Menthol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to meet the evolving demands of the global pharmaceutical and flavor industries. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the ZnBr2/NaY catalyzed cyclization are seamlessly translated into reliable supply solutions. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of L-menthol meets the highest international standards. Our infrastructure is designed to support both custom synthesis projects and large-volume commodity production, providing our partners with the flexibility and security they need to succeed in competitive markets.
We invite you to collaborate with us to leverage these technological advancements for your specific application needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your current supply chain challenges. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can deliver superior value, consistency, and performance for your L-menthol requirements.
