Advanced Continuous Manufacturing of 2,6-Dihydroxytoluene for Global Pharmaceutical Supply Chains
The chemical industry is currently witnessing a paradigm shift towards continuous manufacturing, particularly for high-value intermediates where safety and purity are paramount. Patent CN113233962B introduces a groundbreaking method for the continuous production of 2, 6-dihydroxytoluene, a critical building block in the synthesis of pharmaceuticals, agrochemicals, and functional materials. This technology leverages microchannel reactor systems to overcome the inherent dangers and inefficiencies of traditional batch diazotization. By transitioning from intermittent processing to a seamless continuous flow, the method not only enhances operational safety by mitigating violent heat release but also significantly boosts the overall yield and stability of the final product. For R&D directors and supply chain leaders, this represents a viable pathway to secure a reliable 2,6-dihydroxytoluene supplier capable of meeting rigorous quality standards while optimizing production throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2, 6-dihydroxytoluene has been plagued by significant technical and economic hurdles that hinder large-scale adoption. Traditional routes, such as the propionyl chloride method or the resorcinol high-temperature alkylation, often involve harsh reaction conditions, toxic reagents, and complex post-treatment procedures. For instance, conventional diazotization processes using batch reactors suffer from poor heat transfer, leading to localized hot spots that can trigger runaway reactions and compromise product stability. These methods frequently result in lower yields, typically ranging between 75% and 85% for the diazotization step, and generate substantial amounts of hazardous waste that require costly disposal. Furthermore, the batch nature of these operations limits production capacity and introduces variability in product quality, making it difficult to ensure the consistency required for sensitive pharmaceutical applications.
The Novel Approach
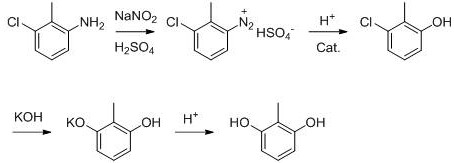
In stark contrast, the novel approach detailed in the patent utilizes a microchannel reactor to execute the diazotization of 3-chloro-2-methylaniline with unprecedented precision and control. This continuous flow technology allows for immediate heat dissipation, effectively eliminating the risk of thermal runaway and ensuring the stability of the reactive diazonium intermediate. The process achieves a remarkable improvement in the diazotization step yield, elevating it from the traditional 75-85% range to an impressive 85-95%. By integrating the reaction steps seamlessly, the method avoids the need to isolate unstable intermediates, thereby streamlining the workflow and reducing the potential for contamination. This innovation not only enhances the total yield of 2, 6-dihydroxytoluene to between 80% and 90% but also guarantees a product purity of not less than 99.0%, setting a new benchmark for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Microchannel Diazotization and Alkali Fusion
The core of this technological advancement lies in the precise control of reaction kinetics within the microchannel environment. During the diazotization phase, 3-chloro-2-methylaniline is dissolved in a 60-70% sulfuric acid solution and reacted with a sodium nitrite solution at a controlled temperature of 0-20°C. The microchannel geometry ensures a residence time of merely 6-20 seconds, which is sufficient for complete conversion while preventing the decomposition of the diazonium salt. This rapid processing is critical for maintaining the integrity of the intermediate, which is then directly hydrolyzed in a 10% sulfuric acid solution at 50-60°C. The immediate transition from diazotization to hydrolysis within the continuous flow system minimizes exposure to destabilizing conditions, thereby maximizing the formation of the key intermediate, 3-chloro-2-methylphenol.
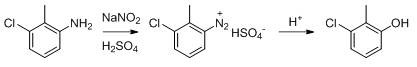
Following the formation of the phenolic intermediate, the process employs a high-pressure alkali fusion step to achieve the final substitution. The 3-chloro-2-methylphenol is reacted with a 20% potassium hydroxide solution in the presence of a catalyst system comprising cuprous chloride and sodium sulfite. This reaction is conducted in an autoclave at elevated temperatures of 170-180°C and pressures of 0.8-1.0 MPa. The catalyst plays a crucial role in facilitating the nucleophilic aromatic substitution, ensuring high conversion rates while minimizing side reactions. Subsequent acidification with hydrochloric acid precipitates the crude product, which is then purified through extraction, decolorization with activated carbon, and crystallization. This robust mechanism ensures that the final 2, 6-dihydroxytoluene meets stringent purity specifications, making it suitable for high-purity OLED material or pharmaceutical intermediate applications.
How to Synthesize 2,6-Dihydroxytoluene Efficiently
Implementing this synthesis route requires a detailed understanding of the continuous flow parameters and safety protocols associated with microchannel reactors. The process begins with the precise preparation of feed solutions, where the solubility of the amine in sulfuric acid is optimized to prevent clogging. Operators must maintain strict control over the molar ratios of reactants, specifically keeping the 3-chloro-2-methylaniline to sodium nitrite ratio between 1:1.05 and 1:1.1 to ensure complete reaction without excess reagent waste. The detailed standardized synthesis steps, including specific flow rates, pressure settings, and work-up procedures, are outlined in the technical guide below to assist process engineers in replicating this high-efficiency protocol.
- Dissolve 3-chloro-2-methylaniline in 60-70% sulfuric acid and react with sodium nitrite solution in a microchannel reactor at 0-20°C for 6-20 seconds residence time.
- Hydrolyze the diazonium salt intermediate in 10% sulfuric acid at 50-60°C for 2 hours to form 3-chloro-2-methylphenol.
- Perform high-pressure alkali fusion with potassium hydroxide and catalyst at 170-180°C, followed by acidification and crystallization to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this continuous manufacturing technology offers substantial strategic benefits beyond mere technical superiority. The shift from batch to continuous processing fundamentally alters the cost structure of production by enhancing throughput and reducing downtime. The ability to run the reaction continuously for 24 hours significantly increases asset utilization, allowing manufacturers to meet large-volume demands without the need for proportional increases in facility footprint. This efficiency translates into significant cost savings, as the streamlined process reduces labor requirements and energy consumption per unit of product. Furthermore, the improved yield directly contributes to better raw material utilization, lowering the overall cost of goods sold and providing a competitive edge in pricing negotiations.
- Cost Reduction in Manufacturing: The elimination of unstable intermediate isolation and the reduction of hazardous waste generation lead to drastically simplified post-treatment processes. By avoiding the use of expensive transition metal catalysts in the initial steps and minimizing solvent usage through efficient extraction protocols, the overall manufacturing cost is significantly reduced. The high yield of the diazotization step means less raw material is wasted, which is a critical factor in maintaining profitability when scaling up production volumes.
- Enhanced Supply Chain Reliability: Continuous manufacturing inherently provides a more stable and predictable output compared to batch methods, which are prone to variability between runs. This consistency ensures a reliable supply of high-purity 2,6-dihydroxytoluene, reducing the risk of production delays for downstream customers. The robust nature of the microchannel system also means that maintenance intervals can be optimized, further ensuring supply continuity and reducing lead time for high-purity 2,6-dihydroxytoluene deliveries to global markets.
- Scalability and Environmental Compliance: The compact design of microchannel reactors allows for easy scale-up through numbering-up rather than increasing vessel size, which mitigates the engineering challenges associated with large batch reactors. This modularity facilitates rapid capacity expansion to meet market demand. Additionally, the process generates fewer three wastes and avoids the use of highly toxic reagents found in older routes, ensuring compliance with increasingly stringent environmental regulations and supporting sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous production of 2, 6-dihydroxytoluene. These answers are derived directly from the patent data and practical implementation insights, providing clarity on safety, quality, and scalability. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their supply chains.
Q: How does microchannel technology improve the safety of diazotization?
A: Microchannel reactors provide superior heat exchange efficiency and small reaction volumes, effectively avoiding violent heat release and unstable product properties associated with traditional batch diazotization.
Q: What purity levels can be achieved with this continuous process?
A: The optimized continuous synthesis method ensures product purity of not less than 99.0%, meeting stringent requirements for pharmaceutical and fine chemical applications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the continuous nature of the microchannel reaction allows for 24-hour operation with high flux, significantly improving production efficiency and scalability compared to batch methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dihydroxytoluene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced process technology plays in securing the global supply of essential chemical intermediates. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the microchannel diazotization process can be successfully translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2, 6-dihydroxytoluene performs consistently in your downstream applications. Our capability to handle complex continuous flow chemistry positions us as a strategic partner for companies seeking to optimize their manufacturing processes.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic potential of switching to this continuous method. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to support your decision-making process. Let us help you enhance your supply chain resilience and achieve your cost reduction targets through innovative chemical manufacturing solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
