Revolutionizing Topramezone Intermediate Production with Green Electrocatalytic Carboxylation Technology
The global agrochemical industry is constantly seeking more sustainable and cost-effective pathways for producing high-value herbicides, and patent CN113862703A presents a groundbreaking solution for the synthesis of Topramezone intermediates. Topramezone, a potent HPPD inhibitor herbicide developed by BASF, has seen immense market demand due to its efficacy against resistant weeds in corn fields, yet its production has been historically constrained by hazardous synthetic routes. This patent introduces a novel electrocatalytic carboxylation method that transforms 3-(3-bromo-2-methyl-6-methylsulfonylphenyl)-4,5-dihydroisoxazole into the key benzoic acid intermediate using carbon dioxide and electricity. By replacing toxic carbon monoxide and expensive palladium catalysts with a green electrochemical system, this technology addresses critical pain points regarding safety, environmental compliance, and manufacturing costs. The structural complexity of the target molecule, as seen in the final herbicide structure below, demands precise chemical construction which this new method achieves with remarkable efficiency.
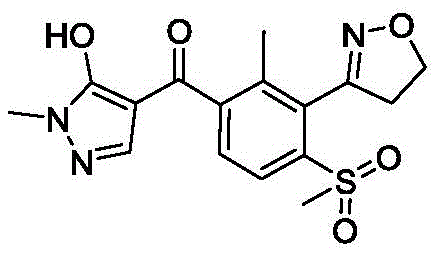
For R&D directors and process chemists, understanding the shift from traditional methodologies to this electrochemical approach is vital for future-proofing supply chains. The conventional reliance on transition metal catalysis often introduces heavy metal impurities that are difficult to remove to pharmaceutical or agrochemical grade standards, necessitating additional purification steps that erode profit margins. In contrast, the electrocatalytic strategy described in CN113862703A operates under mild conditions, typically between -10°C and 40°C, utilizing simple polar solvents like DMF or DMSO. This fundamental shift in reaction engineering not only simplifies the operational protocol but also aligns with the increasing regulatory pressure to reduce the carbon footprint and hazardous waste associated with fine chemical manufacturing. The ability to tune the reaction via current density and electrode material selection offers a level of process control that thermal catalysis simply cannot match.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Topramezone and its precursors has relied heavily on transition metal-catalyzed carbonylation reactions, which present severe logistical and safety challenges for large-scale producers. These traditional routes typically require the use of carbon monoxide, a colorless and odorless gas that poses extreme toxicity risks to plant personnel and requires specialized, high-pressure containment equipment to prevent leaks. Furthermore, the dependence on precious metal catalysts such as palladium creates a significant cost bottleneck, as these metals are subject to volatile market pricing and are often difficult to recover completely from the reaction mixture. The residual metal contamination in the final product can be detrimental to crop safety and requires extensive downstream processing, such as scavenging or recrystallization, which lowers the overall yield and increases the generation of chemical waste. Additionally, the use of active metal reagents in alternative pathways introduces pyrophoric hazards, demanding rigorous inert atmosphere controls that further complicate the manufacturing workflow and increase capital expenditure.
The Novel Approach
The innovative method disclosed in patent CN113862703A circumvents these issues by employing an electrocatalytic carboxylation reaction that utilizes carbon dioxide as a safe and abundant C1 building block. Instead of relying on thermal activation and toxic gases, this process uses electrical energy to drive the reduction of the aryl bromide substrate in the presence of CO2, effectively inserting the carboxyl group directly onto the aromatic ring. The reaction is facilitated by a phase transfer catalyst, which enhances the solubility and reactivity of the ionic species involved, allowing the transformation to proceed smoothly in common polar solvents. This approach eliminates the need for high-pressure reactors and toxic gas handling systems, drastically reducing the safety infrastructure required for production. Moreover, the electrodes used, such as magnesium and silver or nickel, are robust and recyclable, preventing the accumulation of heavy metal waste and ensuring that the process remains environmentally benign throughout its lifecycle.
Mechanistic Insights into Electrocatalytic Carboxylation
The core of this technological advancement lies in the precise electrochemical mechanism that enables the conversion of the bromo-substituted isoxazole derivative into the corresponding benzoic acid. In the electrolytic cell, the magnesium anode serves as a sacrificial electrode, providing the necessary electrons to reduce the organic substrate at the cathode surface, while simultaneously generating magnesium cations that stabilize the intermediate carboxylate species. The carbon dioxide molecule, which is typically kinetically inert, is activated at the cathode interface where it undergoes reduction to form a reactive radical anion or interacts with the electro-generated organometallic intermediate. The presence of a phase transfer catalyst, such as tetrabutylammonium iodide, plays a crucial role in shuttling ions across the solution boundary, ensuring that the electrochemical events occur efficiently within the bulk solvent rather than being limited to the electrode surface. This synergistic interaction between the electric field, the electrode materials, and the catalytic additives allows for high selectivity, minimizing side reactions such as homocoupling or dehalogenation that often plague traditional cross-coupling chemistries.
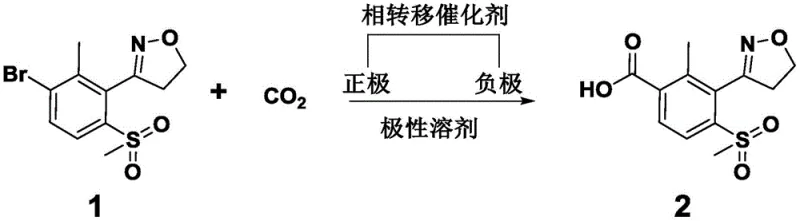
Controlling impurity profiles is a paramount concern for any intermediate intended for agrochemical use, and this electrochemical method offers distinct advantages in this regard. By operating at controlled current densities, typically optimized between 2-5 mA/cm², the reaction kinetics can be finely tuned to favor the desired carboxylation pathway over competing reduction processes. The mild temperature range prevents thermal degradation of the sensitive isoxazole ring and the sulfonyl group, preserving the structural integrity of the molecule. Furthermore, the absence of transition metal catalysts means that there is no risk of palladium or nickel leaching into the product, which simplifies the purification protocol to a straightforward solvent removal and precipitation step. The resulting product, 3-(4,5-dihydroisoxazol-3-yl)-2-methyl-4-methylsulfonylbenzoic acid, is obtained with high purity, often exceeding 90% content without the need for chromatographic separation, making it an ideal candidate for direct use in subsequent condensation reactions to form the final herbicide.
How to Synthesize Topramezone Intermediate Efficiently
To implement this synthesis effectively, manufacturers must adhere to specific operational parameters that maximize yield while maintaining safety and efficiency. The process begins by dissolving the starting bromo-compound and the phase transfer catalyst in a dry polar solvent within an undivided electrolytic cell fitted with appropriate metal electrodes. Carbon dioxide is then introduced into the system, either by bubbling or maintaining a slight positive pressure, to ensure saturation of the reaction medium. The electrolysis is conducted under galvanostatic conditions, where the current is held constant to drive the reaction to completion, typically monitored by HPLC to ensure the starting material is fully consumed. Once the reaction endpoint is reached, the workup involves simple solvent evaporation and extraction, yielding the crude acid which can be dried to specification. For a detailed breakdown of the specific molar ratios, solvent choices, and electrode configurations, please refer to the standardized synthesis guide below.
- Dissolve 3-(3-bromo-2-methyl-6-methylsulfonylphenyl)-4,5-dihydroisoxazole and a phase transfer catalyst in a polar solvent such as DMF or DMSO within an electrolytic cell equipped with magnesium and silver electrodes.
- Introduce carbon dioxide gas into the system and apply a constant current density between 1-6 mA/cm² at a controlled temperature ranging from -10°C to 40°C.
- Monitor the reaction progress via HPLC until the starting material is consumed, then remove the solvent, add ethyl acetate, filter, and dry to obtain the purified carboxylic acid intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrocatalytic technology represents a strategic opportunity to optimize costs and secure long-term supply stability for Topramezone intermediates. The elimination of expensive palladium catalysts removes a major variable cost driver, insulating the production budget from fluctuations in precious metal markets. Additionally, the substitution of toxic carbon monoxide with readily available carbon dioxide reduces the regulatory burden and insurance costs associated with handling hazardous materials, leading to substantial indirect savings. The simplified post-treatment process, which avoids complex metal scavenging steps, shortens the overall production cycle time, allowing for faster turnover and improved asset utilization within the manufacturing facility. These factors combined contribute to a more resilient supply chain that is less susceptible to disruptions caused by raw material shortages or stringent environmental audits.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the removal of high-cost inputs and the simplification of the operational workflow. By avoiding the use of noble metal catalysts, the direct material cost per kilogram of the intermediate is significantly lowered, as there is no need to purchase, recover, or dispose of palladium complexes. The use of electricity as the primary reagent offers a clean and potentially renewable energy source for the chemical transformation, aligning with corporate sustainability goals while reducing reliance on fossil-fuel-derived reagents. Furthermore, the ability to recycle the polar solvent and the durability of the metal electrodes mean that consumable costs are minimized over extended production runs, resulting in a lower total cost of ownership for the manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly improved by the use of commodity chemicals and robust equipment that are easily sourced globally. Carbon dioxide is an abundant industrial byproduct with a stable supply chain, unlike specialized gaseous reagents that may face logistics bottlenecks. The equipment required for electrochemical synthesis is relatively standard and does not require the exotic high-pressure vessels needed for carbonylation, meaning that production can be scaled up or transferred between facilities with greater ease. This flexibility ensures that manufacturers can respond quickly to market demand spikes for Topramezone without being constrained by specialized infrastructure limitations or long lead times for reactor fabrication.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is straightforward due to the modular nature of electrochemical cells and the absence of exothermic runaway risks associated with traditional coupling reactions. The environmental profile of the process is superior, as it generates minimal hazardous waste and avoids the emission of toxic gases, facilitating easier compliance with increasingly strict environmental regulations. The simplicity of the workup procedure reduces the volume of wastewater and organic waste generated, lowering disposal costs and enhancing the overall sustainability rating of the product. This makes the intermediate highly attractive for downstream formulators who are under pressure to reduce the environmental impact of their agricultural products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrocatalytic synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing production lines. The answers provided are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-making purposes.
Q: What are the primary safety advantages of this electrocatalytic method over traditional carbonylation?
A: Unlike conventional methods that require highly toxic carbon monoxide (CO) gas and expensive transition metal catalysts like palladium, this process utilizes benign carbon dioxide (CO2) and recyclable metal electrodes, significantly reducing production safety risks and equipment requirements.
Q: How does the phase transfer catalyst influence the reaction efficiency?
A: The phase transfer catalyst, such as tetrabutylammonium iodide, facilitates the transport of ions between phases, enhancing the interaction between the organic substrate and the electrochemically activated carbon dioxide, thereby improving yield and purity without needing harsh conditions.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method is designed for industrial scalability as it avoids complex post-treatment steps, allows for solvent recovery and recycling, and uses robust electrode materials that do not degrade quickly, ensuring consistent supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Topramezone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the electrocatalytic carboxylation technology described in patent CN113862703A for the agrochemical sector. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications for complex intermediates like 3-(4,5-dihydroisoxazol-3-yl)-2-methyl-4-methylsulfonylbenzoic acid. We are committed to delivering high-purity Topramezone intermediates that empower our partners to produce next-generation herbicides with superior efficacy and environmental safety profiles.
We invite global agrochemical companies and procurement teams to collaborate with us to leverage this cutting-edge synthesis method for your supply chain needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that demonstrates exactly how switching to this electrochemical route can improve your bottom line. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and competitive pricing quotes for this high-value intermediate. Let us help you secure a sustainable and cost-efficient supply of Topramezone precursors for the growing global market.
