Advanced Photocatalytic Synthesis of Alpha-Thioglycosides for Commercial Scale-up
Introduction to Novel Glycosyl Donor Technology
The landscape of carbohydrate chemistry is undergoing a significant transformation driven by the need for more efficient and stereoselective synthetic methodologies. Patent CN112279880A introduces a groundbreaking class of glycosyl donors featuring a novel allyl sulfone structure, specifically designed to overcome the longstanding limitations in thioglycoside synthesis. These compounds serve as critical building blocks for the construction of bioactive molecules, including glycopeptides and mimics of natural products that exhibit enhanced hydrolytic stability compared to their oxygen-linked counterparts. The invention provides not only the structural framework for these donors but also a robust preparation method that leverages visible-light photocatalysis to achieve high yields under exceptionally mild conditions. For pharmaceutical developers, this represents a pivotal shift towards more sustainable and controllable glycosylation strategies that can be reliably scaled for commercial production.
Thioglycosides play an indispensable role in the synthesis of complex carbohydrates and the modification of natural products, acting as stable glycosyl donors in numerous key transformations. Unlike natural O-glycosides, S-glycosides maintain similar steric conformations while offering superior resistance to enzymatic and chemical degradation, making them ideal candidates for drug development and biological probing. The ability to construct these linkages with precise stereocontrol, particularly accessing the alpha-configuration which is often thermodynamically disfavored, is of paramount importance. The technology disclosed in this patent addresses this challenge by utilizing a radical-based mechanism that bypasses the thermodynamic constraints of traditional ionic pathways, thereby opening new avenues for the synthesis of structurally diverse and biologically relevant thioglycoside intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of thioglycosides has relied heavily on Lewis acid-mediated activation of glycosyl donors, a process that is frequently plagued by stereochemical issues. Most conventional methods tend to produce the beta-configured thioglycosides due to thermodynamic control, necessitating complex separation protocols or additional inversion steps to obtain the desired alpha-anomers. Furthermore, alternative strategies developed by research groups, such as those utilizing glycosyl disulfides and phosphines for reductive condensation, are often restricted by the configuration of the anomeric carbon thiol and are generally applicable only to thermodynamically stable configurations. Another notable approach involving organolithium or Grignard reagents suffers from severe functional group incompatibility and is largely limited to 2-deoxy sugars, rendering it unsuitable for the modification of sensitive peptides or proteins. These limitations collectively hinder the efficient synthesis of complex glycoconjugates required in modern medicinal chemistry.
The Novel Approach
In stark contrast to these traditional constraints, the novel approach described in the patent utilizes a photocatalytic free radical reaction that operates under remarkably mild conditions, typically at room temperature or slightly elevated temperatures up to 45°C. By employing a specialized allyl sulfone glycosyl donor, the method facilitates the generation of glycosyl radicals that react efficiently with various thio-based acceptors to form the C-S bond with high alpha-selectivity. This radical pathway circumvents the thermodynamic preferences that dictate the outcome of ionic reactions, allowing for the direct formation of alpha-thioglycosides without the need for harsh reagents or cryogenic conditions. The simplicity of the preparation method for the donor itself, combined with the high efficiency of the coupling reaction, establishes a new standard for reliability and ease of operation in carbohydrate synthesis.
Mechanistic Insights into Photocatalytic Radical Glycosylation
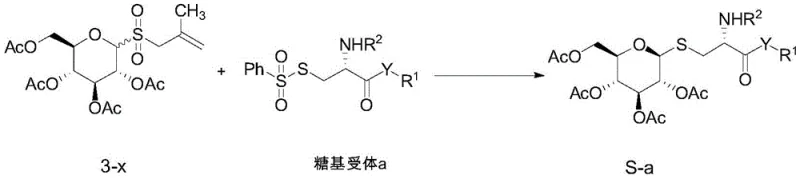
The core of this technological advancement lies in the intricate interplay between the novel allyl sulfone donor and the iridium-based photocatalyst under blue LED irradiation. The mechanism initiates with the excitation of the photocatalyst, typically Ir[dF(CF3)(ppy)2](dtbbpy)PF6, which then engages in a single-electron transfer process with the glycosyl donor. This interaction triggers the homolytic cleavage of the carbon-sulfur bond or facilitates the generation of a sulfur-centered radical that subsequently abstracts a hydrogen or interacts with the acceptor to propagate the radical chain. The use of the 2-methyl-1-allyl sulfone moiety is critical, as it provides the necessary electronic and steric environment to stabilize the transient radical intermediates while ensuring that the reaction proceeds with high fidelity. Detailed experimental optimization revealed that the nature of the leaving group on the glycosyl acceptor significantly influences the reaction efficiency, with phenylsulfone groups demonstrating superior performance compared to pyridyl or simple alkyl disulfides.
Stereocontrol in this radical manifold is achieved through a combination of kinetic factors and the inherent conformational preferences of the sugar ring during the radical recombination step. The patent data highlights that when the leaving group on the acceptor is a phenylsulfone group, the reaction yield reaches an impressive 95%, significantly outperforming other variants such as nitro-substituted pyridines which yielded 0%. This suggests that the electron-withdrawing nature and the specific redox potential of the phenylsulfone moiety are perfectly matched to the catalytic cycle, minimizing side reactions and maximizing the formation of the desired alpha-linked product. Furthermore, the solvent plays a crucial role, with acetonitrile (MeCN) proving to be the optimal medium, likely due to its ability to solvate the ionic intermediates and stabilize the excited state of the catalyst without interfering with the radical propagation steps.
How to Synthesize Allyl Sulfone Glycosyl Donors Efficiently
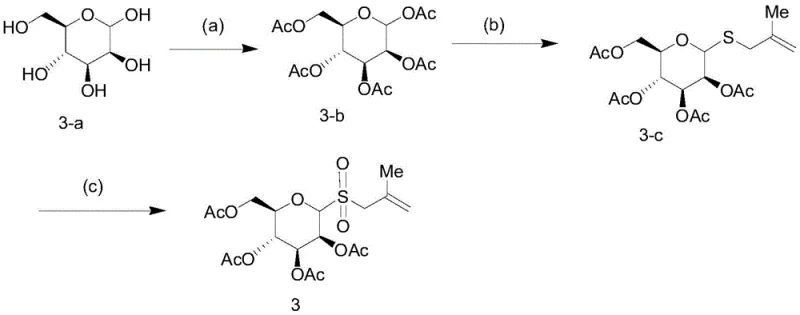
The preparation of these high-value glycosyl donors follows a streamlined three-step sequence that begins with readily available monosaccharides. The process is designed to be robust and scalable, avoiding the need for exotic reagents or extreme conditions that often complicate manufacturing. The initial step involves the peracetylation of the sugar hydroxyl groups to protect the scaffold, followed by the introduction of the allyl thio-group via a substitution reaction with thiourea and an allyl bromide derivative. The final oxidation step converts the sulfide into the reactive sulfone, completing the donor synthesis. This logical progression ensures high purity at each stage, which is essential for the subsequent photocatalytic coupling. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Protect the hydroxyl groups of the starting sugar (e.g., glucose) using acetic anhydride, triethylamine, and DMAP in dichloromethane at room temperature to obtain the peracetylated intermediate.
- React the peracetylated intermediate with thiourea and boron trifluoride etherate in acetonitrile under reflux, followed by the addition of 3-bromo-2-methylpropene and triethylamine to introduce the allyl thio-group.
- Oxidize the resulting allyl thio-sugar using mCPBA in dichloromethane at room temperature to yield the final allyl sulfone glycosyl donor.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this photocatalytic technology offers substantial strategic benefits that extend beyond mere chemical efficiency. The elimination of cryogenic conditions and highly reactive organometallic reagents translates directly into reduced operational complexity and lower safety risks during manufacturing. Traditional methods often require specialized equipment for handling pyrophoric materials or maintaining sub-zero temperatures, which increases capital expenditure and energy consumption. In contrast, the new method utilizes standard laboratory glassware equipped with blue LED lights and common organic solvents like acetonitrile, facilitating a smoother transition from bench-scale discovery to pilot plant production without the need for significant infrastructure upgrades.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the use of commercially available starting materials and catalysts that do not rely on scarce precious metals in large quantities. The high yields reported, often exceeding 90% for optimized substrates, minimize waste generation and reduce the cost per gram of the final API intermediate. Furthermore, the simplified purification protocols, which often involve standard column chromatography or crystallization rather than complex distillation or extraction sequences, contribute to significant savings in labor and solvent costs. By streamlining the synthetic route, manufacturers can achieve a more favorable cost structure that supports competitive pricing in the global pharmaceutical market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions, which are less sensitive to minor fluctuations in temperature or moisture compared to moisture-sensitive Lewis acid protocols. The broad substrate scope demonstrated in the patent, encompassing glucose, galactose, mannose, and even disaccharides like lactose and maltose, ensures that a single platform technology can be applied to produce a diverse portfolio of intermediates. This versatility reduces the dependency on multiple specialized suppliers for different glycosylation reagents and mitigates the risk of supply disruptions caused by the unavailability of niche chemicals. The ability to source raw materials from established chemical vendors further secures the continuity of production schedules.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge due to light penetration issues, but advancements in flow chemistry and LED technology have made this increasingly feasible for industrial applications. The mild reaction conditions generate fewer hazardous byproducts and reduce the overall environmental footprint of the synthesis, aligning with green chemistry principles and stringent regulatory requirements. The use of acetonitrile, a solvent with well-established recovery and recycling protocols, simplifies waste management and ensures compliance with environmental standards. This alignment with sustainability goals not only meets corporate responsibility targets but also future-proofs the manufacturing process against tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation, providing clarity on the practical aspects of adopting this method for industrial synthesis. Understanding these nuances is critical for R&D teams planning to integrate this chemistry into their existing pipelines.
Q: What is the primary advantage of this photocatalytic method over traditional Lewis acid glycosylation?
A: Traditional Lewis acid methods often favor the thermodynamically stable beta-configuration and require harsh conditions. This photocatalytic radical approach operates under mild conditions (room temperature, blue LED) and achieves high stereoselectivity for the alpha-configuration, which is often difficult to access.
Q: Which photosensitizer provides the highest reaction yield?
A: Experimental screening within the patent data indicates that the iridium-based complex Ir[dF(CF3)(ppy)2](dtbbpy)PF6 delivers superior performance, achieving yields up to 95% compared to organic dyes or other metal complexes.
Q: Can this method be applied to complex disaccharides?
A: Yes, the patent demonstrates successful application to disaccharide donors such as lactose and maltose derivatives, indicating broad substrate compatibility and potential for synthesizing complex oligosaccharide intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glycosyl Donor Supplier
As the demand for complex carbohydrate intermediates continues to rise in the pharmaceutical sector, partnering with an experienced CDMO is essential for successful project execution. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory scale to full manufacturing is seamless and efficient. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of glycosyl donor or thioglycoside intermediate meets the highest international standards, minimizing the risk of downstream processing failures. We understand the critical nature of these building blocks in drug discovery and are dedicated to providing consistent quality and reliable supply.
We invite you to engage with our technical procurement team to discuss how this advanced photocatalytic technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this novel synthetic route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your decision-making process and accelerate your development timelines. Let us be your partner in navigating the complexities of modern carbohydrate chemistry.
