Advanced Manufacturing Protocol for Everolimus: Enhancing Yield and Scalability for Global Supply Chains
Advanced Manufacturing Protocol for Everolimus: Enhancing Yield and Scalability for Global Supply Chains
The pharmaceutical landscape for immunosuppressants and oncology therapeutics is constantly evolving, driven by the need for more efficient and reliable manufacturing processes for complex molecules like Everolimus (SDZ-RAD). A significant technological advancement in this domain is detailed in patent CN102786534A, which outlines a novel preparation method that addresses critical bottlenecks in traditional synthesis routes. This proprietary approach leverages a specialized three-step sequence involving the activation of a silane reagent followed by selective coupling and deprotection, offering a pathway to significantly higher purity and yield compared to legacy methods. For R&D directors and procurement strategists, understanding this mechanism is vital, as it represents a shift from fragile, low-yield laboratory curiosities to robust, industrially scalable protocols. By optimizing the protection strategy at the 40-hydroxyl position of the rapamycin scaffold, this technology ensures better control over the impurity profile, a key metric for regulatory compliance in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Everolimus has been plagued by inefficiencies that hinder cost-effective mass production. The prior art, specifically exemplified by U.S. Patent 5665772, describes a synthetic route that, while chemically feasible, demonstrates severe practical limitations when translated to an industrial setting. The primary drawback of this conventional methodology is its notoriously poor repeatability and exceptionally low overall yield, which reportedly hovers around merely 5%. Such low efficiency not only inflates the cost of goods sold (COGS) due to massive material waste but also introduces significant variability in batch-to-batch consistency. Furthermore, the harsh conditions often required in older pathways can lead to the degradation of the sensitive macrolide ring system, generating difficult-to-remove impurities that complicate downstream purification. These factors collectively render the traditional approach economically unviable for meeting the growing global demand for high-purity Everolimus.
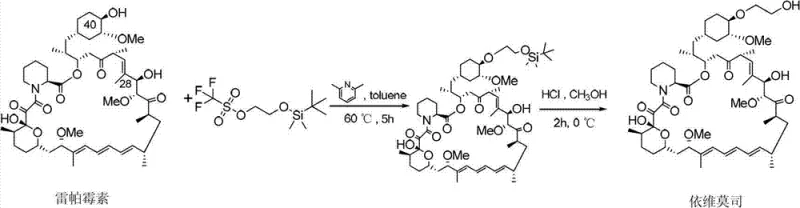
The Novel Approach
In stark contrast to the inefficiencies of the past, the method disclosed in CN102786534A introduces a streamlined and highly effective strategy centered on the use of an activated silyl ether intermediate. This novel approach begins with the precise activation of tert-butyldimethylhydroxyethoxysilane using trifluoromethanesulfonic anhydride under nitrogen protection, creating a highly reactive species capable of efficient coupling. By reacting this activated intermediate with Rapamycin (Compound A) in a mild alkaline organic environment, the process achieves selective functionalization with remarkable stability. The result is a synthesis route that is not only shorter but also operates under much gentler conditions, avoiding the extreme parameters that often degrade product quality. This methodological shift transforms the production landscape, offering a reliable everolimus supplier pathway that maximizes resource utilization and minimizes waste generation.
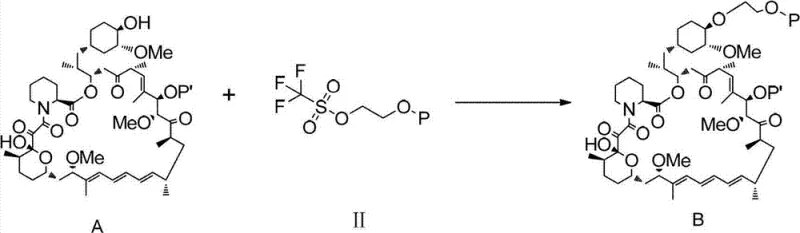
Mechanistic Insights into Silane-Mediated Selective Alkylation
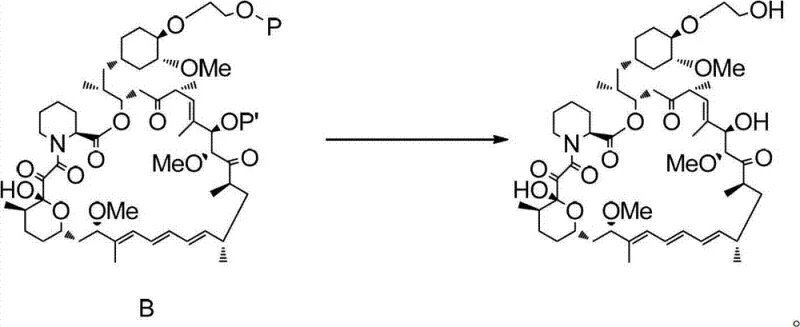
The core of this technological breakthrough lies in the mechanistic elegance of the silane activation and coupling steps. The process initiates with the conversion of a hydroxy-functionalized silane into a triflate ester (Compound II) using a strong sulfonylating agent. This transformation turns a relatively poor leaving group (hydroxyl) into an exceptional leaving group (triflate), thereby dramatically increasing the electrophilicity of the silicon-bound carbon chain. When introduced to the Rapamycin substrate in the presence of a non-nucleophilic base like 2,6-lutidine or potassium carbonate, this activated species selectively targets the 40-hydroxyl group. This selectivity is paramount, as Rapamycin contains multiple hydroxyl functionalities that could potentially react; however, the specific steric and electronic tuning of the reagent system ensures that the reaction proceeds exclusively at the desired position, preserving the integrity of the rest of the molecule.
Following the successful formation of the protected intermediate (Compound B), the final step involves a controlled acidic deprotection to reveal the final Everolimus structure. This step is critical for impurity control, as the use of mild acids like hydrochloric acid in methanol at low temperatures (0°C to 10°C) allows for the cleavage of protecting groups without inducing acid-catalyzed rearrangements or hydrolysis of the sensitive ester linkages within the macrocycle. The ability to perform this deprotection under such mild conditions is a testament to the stability of the intermediate formed in the previous step. By minimizing side reactions during the final workup, the process ensures a cleaner crude product, which significantly reduces the burden on purification teams and enhances the overall throughput of the manufacturing line.
How to Synthesize Everolimus Efficiently
Implementing this synthesis route requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and safety. The process is divided into three distinct operational phases: the preparation of the activated silane reagent, the coupling reaction with the rapamycin core, and the final acidic workup. Each stage demands precise temperature control and stoichiometric balance to maintain the delicate equilibrium between reactivity and selectivity. Operators must ensure an inert atmosphere, particularly during the activation step, to prevent moisture from quenching the reactive triflate intermediate. The following guide outlines the standardized procedural framework derived from the patent data, serving as a foundational reference for process engineers aiming to replicate this high-efficiency protocol.
- Activation of Silane Reagent: React tert-butyldimethylhydroxyethoxysilane with trifluoromethanesulfonic anhydride in the presence of a base like 2,6-lutidine at low temperatures to form the active triflate intermediate.
- Selective Coupling: Introduce the activated silane intermediate to an alkaline solution of Rapamycin (Compound A) in a ketone solvent to selectively protect the 40-hydroxyl group, yielding Compound B.
- Acidic Deprotection: Treat the protected intermediate (Compound B) with a dilute acid solution in an alcohol solvent at controlled low temperatures to remove protecting groups and isolate pure Everolimus.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method offers tangible strategic benefits that extend beyond mere chemical curiosity. The transition from a low-yield, unpredictable process to a high-yield, stable protocol directly correlates with substantial cost reduction in pharmaceutical intermediates manufacturing. By eliminating the need for excessive starting materials to compensate for poor conversion rates, manufacturers can drastically lower their raw material expenditure. Furthermore, the use of common, commodity-grade solvents such as dichloromethane, acetone, and methanol ensures that the supply chain remains resilient against fluctuations in specialty chemical availability. This reliance on standard industrial solvents simplifies logistics and reduces the risk of production stoppages due to reagent shortages.
- Cost Reduction in Manufacturing: The economic impact of improving yield from approximately 5% to over 35% cannot be overstated in terms of unit cost economics. This dramatic increase in efficiency means that less raw Rapamycin is wasted per kilogram of final Everolimus produced, leading to significant savings on one of the most expensive starting materials in the process. Additionally, the simplified operational workflow reduces labor hours and energy consumption associated with prolonged reaction times or complex purification sequences. The elimination of expensive transition metal catalysts, which often require costly removal steps to meet residual metal specifications, further contributes to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Stability is the cornerstone of a reliable supply chain, and this method delivers exactly that through its robust reaction conditions. Unlike processes that require cryogenic temperatures or ultra-dry environments that are difficult to maintain in large reactors, this protocol operates effectively at near-ambient temperatures (20°C to 30°C) for the critical coupling step. This tolerance to standard operating conditions reduces the technical risk of batch failures and ensures consistent output quality. Moreover, the reagents involved, such as trifluoromethanesulfonic anhydride and common inorganic bases, are widely available from multiple global suppliers, mitigating the risk of single-source dependency and enhancing overall supply security.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the shorter synthetic route inherently generates less waste, aligning with modern green chemistry principles. The high selectivity of the reaction minimizes the formation of byproducts, which simplifies wastewater treatment and reduces the load on effluent processing facilities. This environmental efficiency is crucial for maintaining regulatory compliance in increasingly stringent jurisdictions. Furthermore, the straightforward nature of the chemistry facilitates seamless commercial scale-up of complex immunosuppressants, allowing manufacturers to rapidly ramp up production volumes from pilot plant scales to multi-ton commercial campaigns without encountering the nonlinear challenges often associated with scaling fragile chemical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Everolimus synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear picture of the method's capabilities and constraints. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios. The answers below clarify the specific advantages over prior art and the operational parameters required for success.
Q: How does this novel synthesis method improve upon the prior art US5665772?
A: The conventional method described in US5665772 suffers from poor repeatability and extremely low yields, typically around 5%. In contrast, the method disclosed in patent CN102786534A utilizes a specific activated silane coupling strategy that significantly enhances reaction stability and selectivity, resulting in a total recovery rate reaching up to 35% in experimental embodiments, making it far more viable for industrial application.
Q: What are the critical reaction conditions for the coupling step?
A: The coupling reaction between Rapamycin and the activated silane intermediate is optimally performed in a ketone solvent, such as acetone, using a mild inorganic base like potassium carbonate. The reaction temperature is maintained between 20°C and 30°C, which balances reaction kinetics with the stability of the sensitive macrolide structure, preventing degradation while ensuring high conversion rates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for industrial suitability. It employs readily available solvents like dichloromethane, acetone, and methanol, along with common reagents such as trifluoromethanesulfonic anhydride and hydrochloric acid. The operational simplicity, combined with the elimination of complex purification steps required by lower-yield methods, supports robust commercial scale-up and consistent supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Everolimus Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative protocols like the one described in CN102786534A can be successfully translated into reality. We are committed to delivering high-purity Everolimus and related intermediates that meet the most stringent purity specifications required by international regulatory bodies. Our state-of-the-art rigorous QC labs are equipped to analyze complex impurity profiles, guaranteeing that every batch we produce adheres to the highest standards of quality and safety.
We invite potential partners to engage with our technical procurement team to discuss how we can support your specific supply chain needs. Whether you require a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this new manufacturing route or need specific COA data to verify product quality, we are ready to assist. Please contact us today to request route feasibility assessments and discover how our manufacturing capabilities can enhance your project's success and reduce your time to market.
