Advanced Synthesis of High-Purity Ibrutinib Intermediates for Commercial Scale-Up
Introduction to Next-Generation Ibrutinib Intermediate Manufacturing
The pharmaceutical landscape for oncology treatments continues to evolve, with small molecule BTK inhibitors like Ibrutinib remaining a cornerstone therapy for mantle cell lymphone. However, the commercial viability of such potent drugs relies heavily on the efficiency of their supply chain, specifically the production of high-quality intermediates. Patent CN115124536A, published in late 2022, introduces a transformative synthesis method for a critical ibrutinib intermediate salt. This innovation addresses long-standing challenges in purity and yield that have plagued previous manufacturing routes. By shifting from complex chromatographic purifications to a robust salt formation strategy, this technology offers a pathway to significantly reduce impurity profiles, particularly dimer formation, which is a critical quality attribute for regulatory approval. For global procurement teams and R&D directors, understanding this shift is vital for securing a reliable pharmaceutical intermediates supplier capable of meeting stringent GMP standards.
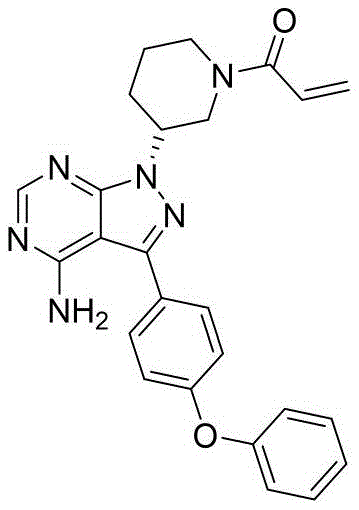
The core of this technological breakthrough lies in the preparation of the mesylate salt of 3-(4-phenoxyphenyl)-1-(piperidin-3-yl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine. Unlike traditional free-base handling, which is prone to oxidative degradation and non-selective acylation, the salt form provides enhanced stability and controlled reactivity. This allows for a cleaner final coupling step with acryloyl chloride, directly translating to higher assay values and reduced downstream processing burdens. As the demand for generic and biosimilar versions of blockbuster oncology drugs grows, the ability to produce high-purity API intermediates with consistent batch-to-batch reproducibility becomes a decisive competitive advantage in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ibrutinib has been fraught with inefficiencies that drive up costs and compromise supply security. Prior art, such as that disclosed in patent CN101610676B, outlines a route involving the reaction of 4-phenoxybenzoic acid derivatives through multiple steps including thionyl chloride activation and malononitrile condensation. While chemically feasible, this legacy approach suffers from a critical bottleneck: the reliance on flash chromatography for final purification. In an industrial setting, flash chromatography is notoriously difficult to scale, requiring vast amounts of silica gel and solvents, which creates significant waste disposal issues and operational hazards. Furthermore, the final acryloylation step in these older methods often results in low product purity due to the formation of stubborn impurities that are hard to remove without expensive preparative HPLC.
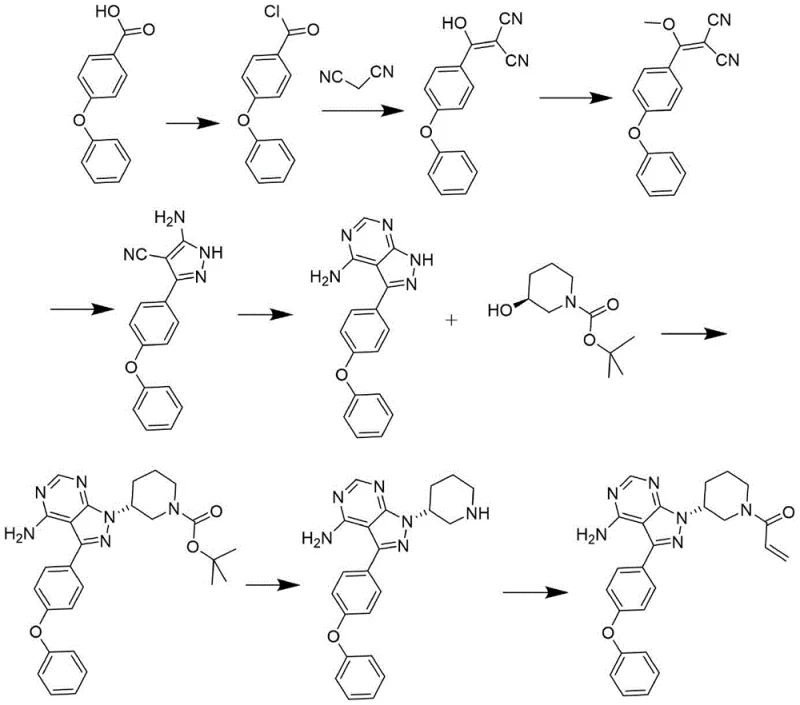
Another significant drawback identified in patent CN105985343A involves the direct reaction of the free base intermediate with acryloyl chloride. Because the intermediate molecule contains multiple nucleophilic nitrogen sites, including the piperidine nitrogen and the pyrazolo-pyrimidine ring nitrogens, non-selective acylation frequently occurs. This leads to the generation of dimer impurities and other structural analogs that are structurally similar to the target API. Removing these genotoxic or closely related impurities requires rigorous and costly purification protocols. For a procurement manager, these inefficiencies manifest as higher raw material costs, longer lead times, and a fragile supply chain that is vulnerable to yield fluctuations. The industry urgently needed a method that could bypass these purification nightmares while maintaining high stereochemical integrity.
The Novel Approach
The methodology presented in CN115124536A represents a paradigm shift towards process intensification and impurity control. Instead of struggling to purify the free base after a messy reaction, this novel approach proactively manages reactivity through salt formation. The process begins with the preparation of the key intermediate, 3-(4-phenoxyphenyl)-1-(piperidin-3-yl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine, via a streamlined sequence involving hydrazine hydrate cyclization and formamide condensation. Crucially, before the final acryloylation, the intermediate is converted into its mesylate salt form. This simple yet effective modification alters the electronic environment of the molecule, effectively blocking or deactivating unwanted reaction sites during the subsequent acylation.
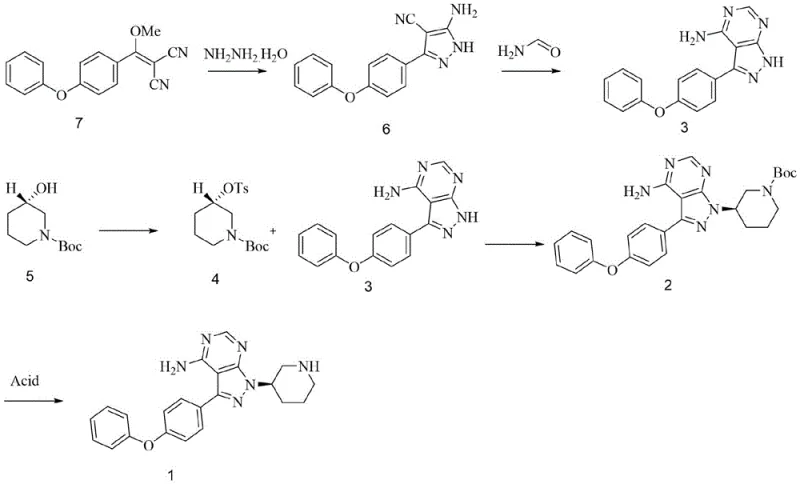
By utilizing the mesylate salt, the synthesis achieves a remarkable improvement in selectivity. The patent data indicates that reacting the mesylate intermediate with acryloyl chloride in the presence of a base yields ibrutinib with purity levels reaching 99.6% and yields as high as 98%. This is a substantial leap from the 86% purity often seen in comparative examples using free base routes. The elimination of flash chromatography in favor of crystallization-based purification (using solvents like methyl tert-butyl ether) makes this process inherently scalable. For manufacturers aiming for cost reduction in pharmaceutical manufacturing, this transition from chromatography to crystallization is the holy grail, offering drastic reductions in solvent consumption, waste generation, and processing time, thereby ensuring a more robust and economically viable supply chain for this critical oncology medication.
Mechanistic Insights into Salt-Mediated Selective Acylation
To fully appreciate the technical superiority of this method, one must delve into the mechanistic nuances of the acryloylation step. In the conventional free-base route, the piperidine nitrogen and the exocyclic amine or ring nitrogens compete for the electrophilic acryloyl chloride. This competition is driven by similar nucleophilicities, leading to a statistical mixture of products. However, by converting the intermediate into a mesylate salt, the basic nitrogen atoms are protonated or coordinated with the sulfonate anion. This protonation significantly reduces the nucleophilicity of the protected sites. When a mild base like diisopropylethylamine (DIPEA) is introduced along with the acylating agent, the deprotonation and subsequent acylation can be kinetically controlled to favor the piperidine nitrogen, which is the desired site for ibrutinib activity. This "protect-by-salt" strategy avoids the need for bulky, expensive protecting groups like Boc or Cbz, which would require additional installation and removal steps.
Furthermore, the crystallization behavior of the mesylate salt plays a pivotal role in impurity rejection. During the formation of the salt in dichloromethane followed by precipitation in methyl tert-butyl ether, the crystal lattice preferentially incorporates the target molecule while excluding structurally dissimilar impurities. This phenomenon, known as purification by crystallization, acts as a powerful filter that chromatography attempts to mimic but often fails to achieve at scale without significant loss of yield. The result is an intermediate with a purity of up to 99.9% (as seen in Example 4 of the patent) before the final coupling even occurs. Starting with such a high-purity precursor ensures that the final API meets the rigorous specifications required by global health authorities, minimizing the risk of batch rejection and ensuring supply chain reliability for downstream drug product manufacturers.
How to Synthesize Ibrutinib Intermediate Efficiently
The practical implementation of this synthesis route is designed for seamless integration into existing multipurpose chemical plants. The process leverages standard unit operations such as cooling, stirring, filtration, and drying, avoiding the need for specialized equipment like high-pressure reactors or complex distillation columns. The initial steps involve the cyclization of cyano-esters with hydrazine to form the pyrazole core, followed by condensation with formamide to build the pyrimidine ring. These are well-established reactions in heterocyclic chemistry. The innovation lies in the downstream processing: the careful control of temperature (0-5°C) during salt formation and the precise selection of anti-solvents to maximize recovery. Detailed standardized operating procedures for each reaction stage, including specific molar ratios and solvent volumes, are essential for replicating the high yields reported in the patent literature.
- Dissolve the free base intermediate 3-(4-phenoxyphenyl)-1-(piperidin-3-yl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine in dichloromethane and cool to 0-5°C.
- Add methanesulfonic acid (mesylic acid) in a stoichiometric ratio (1: 1 for mono-salt or 1:2 for di-salt) and stir to form the stable salt.
- Precipitate the product using methyl tert-butyl ether, followed by filtration and drying to obtain the high-purity mesylate salt ready for acryloylation.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and operational continuity, the implications of this patented technology extend far beyond the laboratory bench. The shift to a salt-mediated synthesis route directly addresses the three pillars of sustainable chemical manufacturing: cost, speed, and environmental impact. By eliminating the reliance on flash chromatography, manufacturers can drastically reduce the volume of organic solvents required per kilogram of product. This not only lowers the direct cost of goods sold (COGS) associated with solvent purchase and recovery but also simplifies the waste management infrastructure needed on-site. The ability to purify via crystallization means that batches can be processed faster, increasing the throughput of existing reactor trains and allowing suppliers to respond more agilely to market demand spikes without capital-intensive expansion.
- Cost Reduction in Manufacturing: The economic argument for adopting this mesylate salt route is compelling. Traditional methods that rely on chromatographic purification incur high operational expenses due to the cost of silica gel, the energy required for solvent evaporation, and the labor intensity of column packing and monitoring. By replacing this with a precipitation and filtration workflow, the process eliminates these variable costs entirely. Additionally, the higher yields (approaching quantitative levels in the final step) mean that less raw material is wasted. This efficiency translates into significant margin improvements for the supplier, which can be passed down the supply chain, offering a more competitive pricing structure for the final API intermediate without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is often compromised by processes that are sensitive to minor variations in raw material quality or reaction conditions. Chromatography, in particular, is sensitive to column performance and solvent grade. The new crystallization-based method is far more robust; slight variations in reaction conversion can often be corrected during the crystallization step, which acts as a purification buffer. This robustness ensures consistent delivery schedules and reduces the risk of batch failures that could disrupt the production of the final drug substance. For a reliable pharmaceutical intermediates supplier, this consistency is key to maintaining long-term contracts with major pharmaceutical companies who cannot afford interruptions in their oncology drug production lines.
- Scalability and Environmental Compliance: As regulatory pressure mounts to reduce the environmental footprint of pharmaceutical manufacturing, green chemistry principles are becoming mandatory rather than optional. This synthesis route aligns perfectly with these goals by minimizing solvent usage and avoiding the disposal of tons of spent silica gel. The solvents used, such as dichloromethane and methyl tert-butyl ether, are widely available and have established recovery protocols. The scalability of the process is proven by its simplicity; moving from a 100-gram lab scale to a multi-ton commercial scale involves linear scaling of reactor volumes rather than fundamental changes in the purification technology. This ease of commercial scale-up of complex pharmaceutical intermediates ensures that supply can grow in tandem with clinical and commercial demand for ibrutinib.
Frequently Asked Questions (FAQ)
Navigating the complexities of custom synthesis and technology transfer often raises specific technical and commercial questions for our partners. The following insights are derived directly from the patent data and our internal process engineering expertise to clarify how this new method impacts your sourcing strategy. We understand that switching suppliers or synthesis routes involves due diligence, and transparency regarding the technical advantages is crucial for building trust. These answers address the most common concerns regarding purity profiles, scalability, and regulatory compliance associated with the production of ibrutinib precursors.
Q: How does the mesylate salt strategy improve ibrutinib purity?
A: By forming a mesylate salt prior to the final acryloylation step, the reactivity of the amine groups is modulated. This prevents the formation of dimer impurities that typically occur when acryloyl chloride reacts with multiple nitrogen sites on the free base, thereby significantly enhancing the final API purity.
Q: What are the advantages of this method over traditional chromatography-based purification?
A: Traditional methods often rely on flash chromatography, which is difficult to scale and solvent-intensive. This novel approach utilizes crystallization and salt formation for purification, which is inherently more scalable, cost-effective, and suitable for industrial manufacturing environments.
Q: Is this synthesis route suitable for large-scale production?
A: Yes, the process avoids complex column chromatography and uses common organic solvents like dichloromethane and methyl tert-butyl ether. The high yields (up to 98%) and robust crystallization steps make it highly amenable to commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ibrutinib Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of complex oncology intermediates requires more than just chemical knowledge; it demands a commitment to excellence in process engineering and quality assurance. Our team has extensively analyzed the innovations presented in CN115124536A and has integrated similar salt-formation and crystallization strategies into our own manufacturing platforms. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that whether you need material for clinical trials or full-scale commercial launch, our capacity is ready to meet your needs. Our facilities are equipped with rigorous QC labs and state-of-the-art analytical instruments to verify stringent purity specifications, guaranteeing that every batch of ibrutinib intermediate we deliver meets the highest global standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain for ibrutinib and related BTK inhibitors. By leveraging our technical expertise in process optimization, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us be your partner in delivering life-saving medications to patients faster and more efficiently through superior chemical manufacturing solutions.
