Optimizing Entrectinib Production: A Technical Analysis of the Novel 5-Step Synthetic Route
Optimizing Entrectinib Production: A Technical Analysis of the Novel 5-Step Synthetic Route
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex kinase inhibitors, and the recent disclosure in patent CN113024521B presents a compelling advancement for the production of Entrectinib. This novel methodology addresses critical bottlenecks in existing manufacturing processes by streamlining the synthetic sequence into a concise five-step protocol. Unlike traditional routes that often suffer from excessive length and harsh reaction conditions, this approach leverages a strategic Cbz protection-deprotection sequence coupled with mild amidation conditions. The result is a robust process capable of delivering high-purity active pharmaceutical ingredients (APIs) with significantly reduced operational complexity. For R&D directors and process chemists, this represents a viable pathway to enhance yield and reduce the environmental footprint of oncology drug manufacturing.
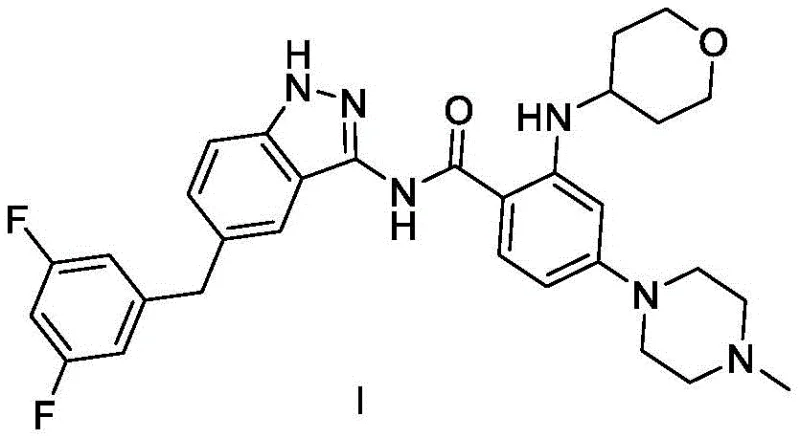
Entrectinib, a potent inhibitor of NTRK, ROS1, and ALK fusion kinases, requires precise structural assembly to maintain its therapeutic efficacy against solid tumors. The chemical architecture, as depicted in the structural formula, demands careful handling of the indazole core and the morpholine-substituted aniline fragment. The patent highlights that achieving the final Formula I structure with a purity of 99.8 percent is not merely a theoretical goal but an experimentally verified outcome of this optimized route. By focusing on reaction efficiency and impurity control, this method provides a reliable foundation for supplying high-quality pharmaceutical intermediates to the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Entrectinib and similar tyrosine kinase inhibitors has been plagued by lengthy synthetic sequences that often exceed nine distinct chemical steps. These elongated routes inevitably lead to cumulative yield losses, where the efficiency of each individual step compounds negatively, resulting in poor overall mass balance. Furthermore, many conventional protocols rely on extreme reaction conditions, such as cryogenic temperatures below minus twenty degrees Celsius, which impose significant energy costs and engineering challenges on manufacturing facilities. The reliance on multiple protection and deprotection cycles using incompatible reagents can also generate difficult-to-remove impurities, necessitating extensive and costly chromatographic purification efforts that hinder commercial scalability.
The Novel Approach
In stark contrast, the methodology outlined in CN113024521B introduces a streamlined strategy that drastically reduces the step count while maintaining high fidelity to the target molecular structure. By utilizing a Cbz (benzyloxycarbonyl) group to temporarily mask the secondary amine functionality early in the sequence, the process prevents unwanted side reactions during subsequent acylation steps. This strategic protection allows for the direct conversion of the carboxylic acid precursor into an acid chloride, which is then efficiently coupled with the indazole amine fragment. The elimination of unnecessary synthetic detours and the avoidance of extremely low-temperature requirements make this novel approach exceptionally attractive for cost reduction in pharmaceutical manufacturing, offering a clearer path from raw materials to finished API.
Mechanistic Insights into Cbz-Mediated Amidation and Deprotection
The core of this synthetic innovation lies in the precise management of nitrogen nucleophilicity and carboxylic acid activation. The initial step involves the reaction of the starting amine with benzyl chloroformate (Cbz-Cl) in the presence of triethylamine, forming a stable carbamate intermediate. This transformation is crucial as it modulates the electronic properties of the nitrogen atom, preventing it from interfering with the subsequent activation of the carboxylic acid moiety. Following the removal of the tert-butyl ester group using trifluoroacetic acid (TFA), the exposed carboxylic acid is activated using thionyl chloride (SOCl2). This generates a highly reactive acid chloride species in situ, which is immediately primed for nucleophilic attack by the amino-indazole partner. The use of DIPEA as a base in tetrahydrofuran ensures that the amidation proceeds smoothly at room temperature, minimizing the formation of racemization byproducts or hydrolysis side products.
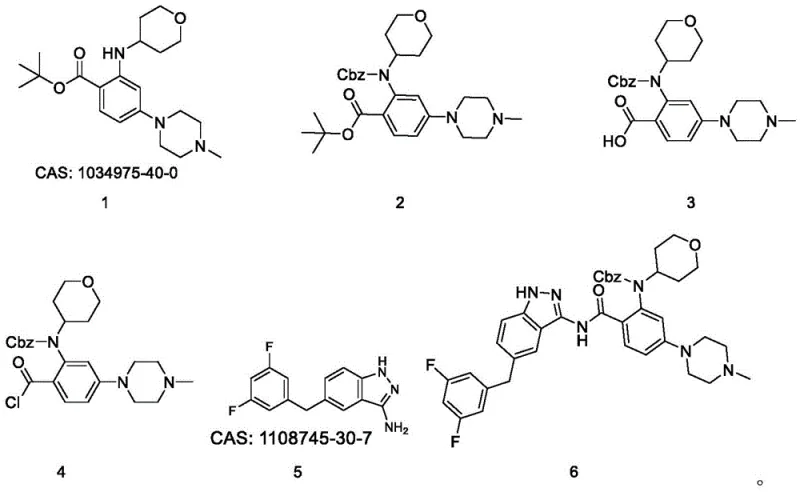
The final and perhaps most mechanistically interesting step is the removal of the Cbz protecting group to reveal the final secondary amine in Entrectinib. Instead of employing catalytic hydrogenation, which can sometimes reduce sensitive heterocycles, or strong Lewis acids that might degrade the molecule, this process utilizes trimethylsilyl iodide (Me3SiI). This reagent acts as a source of iodide ions and silyl cations, effectively cleaving the carbamate bond under mild conditions in acetonitrile. The addition of triethylamine subsequently neutralizes the generated acid and facilitates the isolation of the free amine. This specific choice of deprotection reagent demonstrates a sophisticated understanding of chemoselectivity, ensuring that the morpholine and indazole rings remain intact while achieving the desired structural unmasking with high purity.
How to Synthesize Entrectinib Efficiently
Implementing this synthetic route requires strict adherence to the specified reaction parameters to maximize yield and minimize impurity profiles. The process begins with the protection of the amine in dichloromethane, followed by acid deprotection and activation. The critical coupling step between the acid chloride and the indazole amine must be monitored to ensure complete conversion before proceeding to the final deprotection. Detailed standard operating procedures regarding stoichiometry, solvent quality, and workup protocols are essential for reproducibility. For a comprehensive breakdown of the specific molar ratios, reaction times, and purification techniques validated in the patent examples, please refer to the standardized synthesis guide below.
- Protect the secondary amine of the starting material (Compound 1) using Cbz-Cl and triethylamine in dichloromethane to form Compound 2.
- Remove the tert-butyl ester protecting group using trifluoroacetic acid (TFA) to yield the free carboxylic acid (Compound 3).
- Activate the carboxylic acid with thionyl chloride (SOCl2) to form the acid chloride (Compound 4), then couple with the indazole amine (Compound 5) using DIPEA.
- Perform final deprotection of the Cbz group using trimethylsilyl iodide (Me3SiI) and triethylamine in acetonitrile to obtain Entrectinib.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this streamlined synthesis route offers substantial strategic benefits that extend beyond simple chemical yield. The reduction in the total number of unit operations directly correlates to a decrease in manufacturing lead time and a lower consumption of solvents and reagents per kilogram of product. This efficiency gain translates into significant cost savings, as fewer processing steps mean reduced labor hours, lower utility consumption for heating and cooling, and diminished waste disposal costs. For supply chain heads, the robustness of the process, characterized by its tolerance for room temperature reactions and absence of cryogenic requirements, ensures greater production flexibility and reliability, mitigating the risks associated with equipment failure or energy supply fluctuations.
- Cost Reduction in Manufacturing: The elimination of complex, multi-step sequences inherently lowers the cost of goods sold (COGS) by reducing material throughput requirements. By avoiding the need for expensive cryogenic cooling systems and minimizing the number of isolation and purification stages, manufacturers can achieve a leaner production model. The use of commercially available reagents like thionyl chloride and trimethylsilyl iodide, rather than exotic catalysts, further stabilizes the raw material supply chain and prevents price volatility associated with specialized chemicals.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions enhances the overall reliability of the supply chain. Processes that do not require extremely low temperatures are less susceptible to disruptions caused by refrigeration failures or seasonal energy constraints. Furthermore, the high purity profile of the crude product reduces the dependency on extensive chromatographic purification, which is often a bottleneck in large-scale manufacturing. This ensures a more consistent and predictable delivery schedule for downstream API formulators and pharmaceutical partners.
- Scalability and Environmental Compliance: This route is inherently designed for scalability, utilizing standard solvents like dichloromethane, tetrahydrofuran, and acetonitrile which are well-understood in industrial settings. The reduction in step count naturally leads to a lower E-factor (environmental factor), meaning less waste is generated per unit of product. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, allowing manufacturers to operate with a smaller environmental footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Entrectinib synthesis pathway. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, aiming to clarify the feasibility and advantages of this method for potential manufacturing partners and technical stakeholders.
Q: What is the primary advantage of this Entrectinib synthesis route over conventional methods?
A: The primary advantage is the significant reduction in synthetic steps, condensing the process to just 5 key transformations compared to over 9 steps in prior art. This reduction minimizes material loss, simplifies purification, and eliminates the need for extremely low-temperature reactions, thereby enhancing overall process robustness and scalability.
Q: How does the use of trimethylsilyl iodide improve the final deprotection step?
A: Trimethylsilyl iodide (Me3SiI) allows for the mild and selective removal of the Cbz protecting group under neutral to slightly basic conditions. This avoids the harsh acidic or hydrogenation conditions often required in traditional deprotection, preserving the integrity of sensitive functional groups like the indazole ring and ensuring high product purity.
Q: Is this synthetic route suitable for large-scale industrial production?
A: Yes, the route is specifically designed for industrial viability. It utilizes common solvents like dichloromethane and acetonitrile, operates at room temperature or mild cooling (0°C), and achieves a reported purity of 99.8%, making it highly suitable for commercial scale-up without requiring specialized cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Entrectinib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert process chemists has thoroughly analyzed the methodology presented in CN113024521B and is fully equipped to translate this laboratory-scale success into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. Our state-of-the-art facilities are supported by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of Entrectinib intermediate meets the highest international standards for safety and efficacy.
We invite pharmaceutical companies and contract research organizations to collaborate with us to leverage this advanced synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can optimize your supply chain and accelerate your drug development timelines.
