Advanced Benzyl Structure Carriers for Efficient Liquid Phase Peptide Synthesis and Commercial Scale-Up
The landscape of peptide manufacturing is undergoing a significant transformation driven by the need for higher purity and more scalable processes. Patent CN110183347B introduces a groundbreaking class of compounds containing a benzyl structure, specifically designed to serve as protective carriers for the C-terminus of amino acids and peptides in liquid phase synthesis. This technology addresses critical bottlenecks in traditional methods by combining the advantages of homogeneous reaction kinetics with the ease of heterogeneous separation. The core innovation lies in the molecular architecture, which balances lipophilic long-chain alkyl groups with polar functional handles, enabling versatile solubility profiles that are essential for modern pharmaceutical intermediate production. By leveraging this chemistry, manufacturers can achieve high reaction speeds and reagent utilization rates while maintaining a universal operational flow suitable for diverse solvent systems.
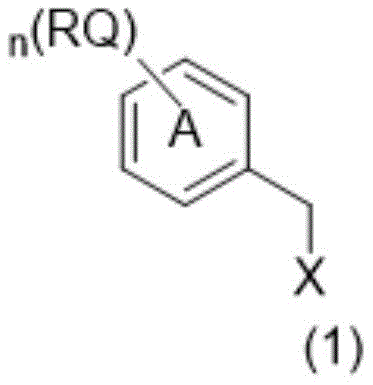
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional peptide synthesis has long been dominated by solid-phase methods, which, while universal, suffer from severe mass transfer limitations due to the poor affinity between solvents and the solid carrier. This often necessitates the use of large excesses of reactants to drive reactions to completion, inflating costs and complicating waste management. Furthermore, intermediates bound to solid supports cannot be easily analyzed or purified by standard techniques, forcing reliance on preparative chromatography for the final product. On the other hand, earlier liquid-phase carrier technologies, such as those utilizing long-chain alkoxybenzyl alcohols, faced their own set of challenges. These included low solubility in medium-to-low polarity solvents like acetate or toluene, which restricted reaction concentrations and hindered scale-up. Additionally, the purification processes often involved complex solvent switching or distillation steps, and the precipitated products were frequently amorphous solids that were difficult to filter and wash effectively, leading to poor impurity removal and extended processing times.
The Novel Approach
The compounds disclosed in CN110183347B represent a paradigm shift by engineering a molecular structure that remains soluble and reactive throughout the synthesis cycle. Unlike previous iterations that suffered from rapid solubility drops and gelation when peptide chains exceeded four amino acids, these new benzyl structures maintain consistent physical properties. The design incorporates tunable alkyl chains (ranging from C8 to C25) and specific linker groups that ensure the carrier dissolves readily in hydrocarbon solvents like heptane as well as polar solvents like DMF. This dual solubility allows for the creation of heterogeneous systems where the reaction occurs efficiently, yet the product can be isolated through simple liquid-liquid extraction. This approach eliminates the need for complex precipitation protocols or column chromatography for intermediates, drastically streamlining the workflow for cost reduction in peptide manufacturing and enabling a truly universal production method.
Mechanistic Insights into Benzyl Structure Carrier Design
The efficacy of these carriers stems from a precise balance of steric and electronic factors within the general formula (1). The molecule features a central benzyl core substituted with multiple long-chain alkoxy groups (RQ), which provide the necessary lipophilicity for solubility in non-polar organic phases. Attached to this core is a linker (L) containing heteroatoms such as oxygen or nitrogen, which connects to the reactive functional group (X), typically a hydroxyl or amino group. This architecture ensures that while the bulk of the molecule anchors it in the organic phase, the reactive end remains accessible for coupling with protected amino acids. The presence of branched alkyl groups, such as isooctyl or isononyl chains, further disrupts crystallization tendencies, preventing the gelation issues that plague linear chain analogs. This structural integrity is maintained even as the peptide chain elongates, ensuring that the intermediate remains in solution or forms a manageable phase for separation.
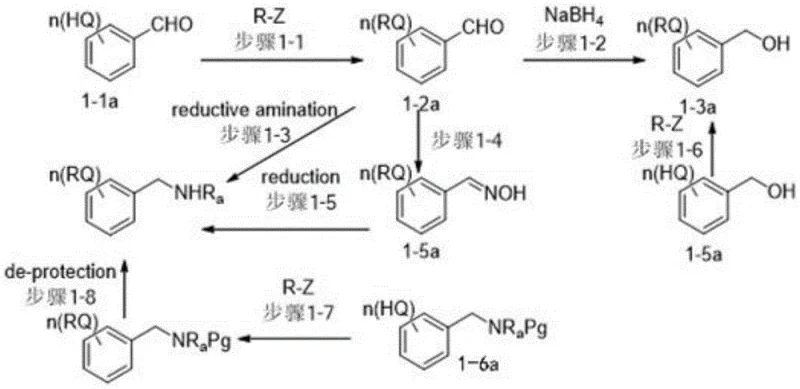
Impurity control is inherently built into the physicochemical properties of these carriers. Because the carrier-peptide conjugate exhibits distinct solubility behavior compared to unreacted reagents and by-products, purification becomes a matter of phase partitioning rather than complex chromatographic separation. In a typical heterogeneous system, the reaction mixture can be washed with high-polarity solvents like water or methanol to remove polar impurities and excess reagents, leaving the desired carrier-bound peptide in the organic layer. This mechanism significantly reduces the risk of racemization and side reactions that can occur during prolonged exposure to purification media. The robustness of the benzyl linkage also ensures stability under the acidic or basic conditions often used for N-terminal deprotection, allowing for repetitive cycles of elongation without premature cleavage of the carrier.
How to Synthesize Benzyl Structure Carriers Efficiently
The synthesis of these advanced carriers follows a logical progression of organic transformations that are well-suited for industrial scale-up. The process typically begins with the alkylation of a hydroxybenzoate precursor using long-chain alkyl halides to install the lipophilic tails, followed by hydrolysis to generate the corresponding acid. This acid is then coupled with an amino-alcohol linker via standard amidation protocols. Finally, the benzyl alcohol or amine functionality is introduced through etherification or reductive amination with substituted benzaldehydes. The detailed standardized synthesis steps for specific embodiments like BM-001 and BM-002 are outlined below to guide process development teams in replicating these high-performance materials.
- Alkylation of hydroxybenzoate derivatives with long-chain alkyl halides to introduce lipophilic tails.
- Hydrolysis of the ester group to form the corresponding benzoic acid intermediate.
- Amidation with amino-alcohol derivatives followed by etherification or reductive amination to attach the benzyl alcohol or amine functional handle.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this benzyl structure carrier technology offers tangible strategic benefits beyond mere technical performance. The primary advantage lies in the drastic simplification of the downstream processing workflow. By replacing complex chromatographic purifications and difficult filtrations with straightforward liquid-liquid extractions, manufacturers can significantly reduce the consumption of expensive silica gels and solvents. This simplification translates directly into lower operational expenditures and a reduced environmental footprint, aligning with modern green chemistry initiatives. Furthermore, the ability to operate at higher concentrations in cost-effective solvents like heptane or isopropyl acetate means that reactor volumes can be utilized more efficiently, increasing throughput without the need for capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction in reagent excesses required for solid-phase synthesis lead to substantial raw material savings. The simplified workup procedure reduces labor hours and utility costs associated with solvent recovery and waste treatment. Since the process avoids the use of specialized resins that often have limited shelf-life and high unit costs, the overall cost of goods sold (COGS) for the final peptide API is optimized. The robustness of the carrier also minimizes batch failures due to gelation or precipitation, ensuring consistent yield and reducing the financial risk associated with production campaigns.
- Enhanced Supply Chain Reliability: The starting materials for these carriers, such as gallic acid derivatives and common long-chain alcohols, are commercially available and stable, reducing the risk of supply disruptions. The synthesis route does not rely on exotic reagents or cryogenic conditions, making it easier to source inputs from multiple qualified vendors. This flexibility strengthens the supply chain against geopolitical or logistical shocks. Additionally, the high purity achievable with this method reduces the burden on quality control laboratories, accelerating the release of batches and ensuring a steady flow of materials to downstream formulation teams.
- Scalability and Environmental Compliance: The liquid-phase nature of this synthesis facilitates seamless scale-up from laboratory grams to multi-ton commercial production. The use of hydrocarbon solvents allows for easier recycling and recovery compared to the complex mixtures often generated in solid-phase synthesis. The reduction in hazardous waste generation, particularly from spent resins and chromatography fractions, simplifies compliance with increasingly stringent environmental regulations. This makes the technology not only economically attractive but also sustainable for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of benzyl structure carriers in peptide synthesis workflows. These answers are derived directly from the experimental data and specifications provided in the patent documentation, offering clarity on solubility profiles, purification strategies, and compatibility with standard amino acid protecting groups. Understanding these nuances is critical for R&D teams planning to integrate this technology into their existing manufacturing pipelines.
Q: What are the solubility characteristics of these benzyl carriers?
A: The compounds exhibit excellent solubility in both non-polar solvents like heptane and polar aprotic solvents like DMF, facilitating heterogeneous reaction systems.
Q: How does purification differ from traditional solid-phase synthesis?
A: Unlike solid-phase synthesis requiring filtration and washing of resin, these carriers allow for liquid-liquid extraction separation, significantly simplifying post-treatment and improving purity.
Q: Can these carriers prevent gelation during peptide elongation?
A: Yes, the specific structural design maintains consistent solubility throughout the synthesis process, preventing the gelation phenomena often observed in other liquid-phase methods when peptide chains exceed four amino acids.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzyl Structure Compound Supplier
As the demand for high-quality peptide therapeutics continues to surge, partnering with a knowledgeable CDMO is essential for navigating the complexities of advanced synthesis technologies. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market supply is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzyl structure carrier or peptide intermediate meets the highest industry standards. We understand the critical nature of timeline and quality in the pharmaceutical sector and are committed to delivering excellence.
We invite you to engage with our technical procurement team to discuss how these innovative carriers can optimize your specific peptide projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your target molecules, ensuring a robust and efficient supply chain for your next-generation therapeutics.
