Advanced Catalyst-Free Synthesis of Oxazepine Derivatives for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways to construct complex nitrogen-containing heterocycles, which serve as the backbone for countless therapeutic agents. In this context, the recent disclosure in patent CN113912562A presents a groundbreaking advancement in the synthesis of oxazepine derivatives, a class of compounds renowned for their presence in antidepressants, analgesics, and HIV-1 reverse transcriptase inhibitors. This novel methodology fundamentally shifts the paradigm by eliminating the reliance on expensive and environmentally burdensome transition metal or photoredox catalysts. Instead, it leverages a direct, one-step condensation between dibenzo[b,f][1,4]oxazepine derivatives and β-keto acids under remarkably mild conditions. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes, while for procurement and supply chain leaders, it signals a potential for drastic cost reductions and improved supply continuity through simplified manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of functionalized oxazepine scaffolds has often necessitated the use of sophisticated catalytic systems involving precious metals such as palladium, rhodium, or ruthenium, as well as complex photoredox setups. These conventional approaches introduce multiple layers of operational complexity and cost. The requirement for strict anhydrous conditions, elevated temperatures, or specialized light sources increases energy consumption and capital expenditure. Furthermore, the inevitable presence of residual heavy metals in the final product poses a severe regulatory hurdle, necessitating additional purification steps like scavenging or recrystallization to meet the stringent ppm limits set by global health authorities. These downstream processing requirements not only extend the overall lead time but also generate significant chemical waste, conflicting with the growing industry mandate for greener, more sustainable manufacturing practices.
The Novel Approach
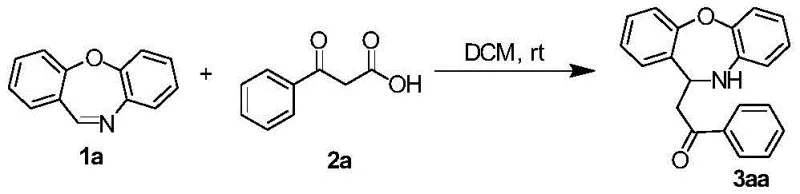
In stark contrast, the technology described in CN113912562A offers a streamlined, catalyst-free alternative that operates at room temperature in common solvents like dichloromethane. By utilizing the inherent reactivity of the dibenzo[b,f][1,4]oxazepine core and β-keto acids, this method achieves high conversion rates without external activation. The general reaction scheme illustrates the simplicity and elegance of this transformation, where diverse substituents on both the oxazepine ring and the phenyl group of the keto acid are well-tolerated. This robustness allows for the rapid generation of a wide library of derivatives, facilitating faster structure-activity relationship (SAR) studies. The elimination of catalysts not only simplifies the workup procedure—often requiring only solvent removal and standard chromatography—but also inherently produces a cleaner crude product, thereby enhancing the overall process mass intensity (PMI) and reducing the environmental footprint of the synthesis.
![General reaction scheme for catalyst-free synthesis of oxazepine derivatives from dibenzo[b,f][1,4]oxazepine and beta-keto acids](/insights/img/oxazepine-derivative-synthesis-catalyst-free-pharma-supplier-20260304181038-03.webp)
Mechanistic Insights into Catalyst-Free Condensation
The success of this catalyst-free protocol relies on the unique electronic properties of the reactants. The dibenzo[b,f][1,4]oxazepine structure possesses a nucleophilic character at the nitrogen adjacent position or the imine carbon, which can effectively attack the electrophilic carbonyl carbon of the β-keto acid. Although the exact mechanistic pathway may involve a transient zwitterionic intermediate or a direct nucleophilic addition followed by proton transfer, the absence of a catalyst suggests that the activation energy barrier is sufficiently low under ambient conditions. This intrinsic reactivity is a critical advantage for scale-up, as it removes the variability associated with catalyst loading, activation, and potential deactivation by impurities. For process chemists, understanding this mechanism allows for precise control over stoichiometry; the patent indicates that a molar ratio of 1:1.5 (oxazepine to keto acid) is optimal, ensuring complete consumption of the limiting reagent while minimizing side reactions.
Furthermore, the impurity profile of this reaction is notably clean compared to metal-catalyzed counterparts. Without transition metals, there is no risk of metal-mediated side reactions such as homocoupling or over-oxidation, which often plague complex heterocycle synthesis. The primary byproducts are likely limited to unreacted starting materials or simple hydrolysis products, which are easily separated during the purification phase. As demonstrated in specific examples, such as the synthesis of compound 3aa shown below, the reaction yields high-purity products (over 90% in some cases) with well-defined NMR spectra, confirming the structural integrity of the newly formed C-C or C-N bonds. This level of purity is essential for pharmaceutical intermediates, where downstream biological testing requires materials free from confounding contaminants.
How to Synthesize Oxazepine Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting is straightforward and requires standard equipment. The process begins with the preparation of the starting materials, which are either commercially available or easily synthesized via known literature methods, such as the condensation of 2-aminophenols with 2-fluorobenzaldehydes. Once the substrates are secured, the reaction is performed by simply mixing the components in dichloromethane at room temperature. The detailed standardized synthesis steps, including specific quantities, stirring times, and purification parameters, are outlined in the structured guide below to ensure reproducibility and safety during operation.
- Charge a reaction vessel with dibenzo[b,f][1,4]oxazepine derivative and beta-keto acid derivative in a molar ratio of approximately 1: 1.5.
- Add dichloromethane (DCM) as the solvent and stir the mixture at room temperature (25-35°C) for approximately 12 hours.
- Remove the solvent under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the target oxazepine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free technology translates into tangible strategic advantages beyond mere technical feasibility. The removal of expensive catalysts from the bill of materials (BOM) directly impacts the cost of goods sold (COGS), making the final intermediate more price-competitive in the global market. Additionally, the simplified workflow reduces the dependency on specialized reagents that may have long lead times or volatile pricing, thereby stabilizing the supply chain against market fluctuations. The mild reaction conditions also imply lower energy costs, as there is no need for heating mantles, cryogenic cooling, or high-pressure reactors, contributing to a leaner and more agile manufacturing operation.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the complete elimination of transition metal catalysts and photoredox reagents, which are often among the most expensive line items in fine chemical synthesis. By removing these costs, manufacturers can achieve substantial savings on raw material procurement. Moreover, the simplified workup procedure eliminates the need for costly metal scavenger resins or extensive washing protocols designed to reduce metal residues to trace levels. This reduction in processing steps lowers labor costs and solvent consumption, further driving down the overall production expense and improving the margin profile for high-volume API intermediate manufacturing.
- Enhanced Supply Chain Reliability: Relying on commodity chemicals like dichloromethane and readily accessible beta-keto acids significantly de-risks the supply chain. Unlike specialized catalysts that may be sourced from a single supplier or face geopolitical supply disruptions, the substrates for this reaction are widely available from multiple global vendors. This diversification of the supply base ensures continuity of supply even during market shortages. Furthermore, the robustness of the reaction means that minor variations in raw material quality are less likely to cause batch failures, leading to higher first-pass yields and more predictable delivery schedules for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: Scaling a chemical process often amplifies safety and environmental challenges, but this method mitigates many of those risks. Operating at room temperature removes the thermal hazards associated with exothermic reactions at high heat, making the process safer for large-scale reactors. From an environmental perspective, the absence of heavy metals simplifies waste stream management and treatment, reducing the burden on wastewater treatment facilities and ensuring compliance with increasingly strict environmental regulations. This "green" credential is increasingly valued by multinational corporations aiming to reduce their carbon footprint and meet sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding this synthesis method, derived directly from the experimental data and claims within the patent documentation. These insights are intended to clarify the operational parameters and potential applications for stakeholders evaluating this technology for integration into their existing portfolios.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method (CN113912562A) operates completely without photoredox or transition metal catalysts, significantly reducing raw material costs and eliminating heavy metal contamination risks.
Q: What are the optimal reaction conditions for this transformation?
A: The reaction proceeds efficiently at room temperature (25-35°C) in dichloromethane (DCM) solvent, typically reaching completion within 12 hours with high yields.
Q: Is this method suitable for large-scale pharmaceutical production?
A: Yes, the mild conditions, absence of hazardous catalysts, and simple workup procedures make this route highly scalable and compliant with stringent environmental and safety regulations for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Oxazepine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of catalyst-free synthetic methodologies in modern drug development. Our team of expert process chemists has extensively evaluated the route described in CN113912562A and confirmed its viability for commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of oxazepine derivative we supply meets the highest standards required for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced technology for your next project. Whether you require custom synthesis of specific analogs or bulk supply of the core scaffold, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
