Advanced Synthesis of 2,4,5-Trifluorophenylacetic Acid for Commercial API Production
The pharmaceutical industry continuously seeks robust and scalable pathways for critical API intermediates, and the synthesis of 2,4,5-trifluorophenylacetic acid stands as a pivotal challenge in the production of Sitagliptin, a leading DPP-IV inhibitor for type II diabetes. Patent CN111187154A introduces a groundbreaking methodology that addresses the longstanding inefficiencies of conventional routes by leveraging a novel metal oxide catalytic system for decarboxylation. This technical advancement is not merely a laboratory curiosity but represents a significant leap towards industrial viability, offering a pathway that combines high yield with exceptional operational safety. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent purity specifications. The strategic implementation of this synthesis route allows for the production of high-purity Sitagliptin intermediate while simultaneously mitigating the environmental and safety risks associated with older technologies. By focusing on the core chemical transformations disclosed, manufacturers can achieve a competitive edge in cost reduction in API intermediate manufacturing without compromising on quality or regulatory compliance.
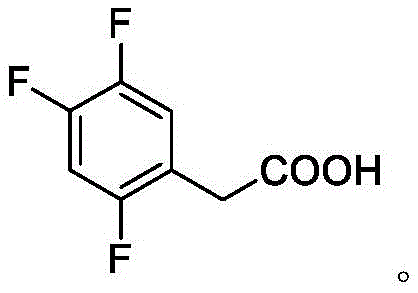
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2,4,5-trifluorophenylacetic acid has been plagued by synthetic routes that impose severe constraints on industrial scalability and safety protocols. Traditional methods often rely on the use of Grignard reagents, which necessitate strictly anhydrous conditions and present significant explosion hazards due to the high reactivity of the intermediates involved in the process. Furthermore, alternative pathways utilizing sodium cyanide for cyanidation reactions introduce toxic hazards that complicate waste management and increase the overall environmental footprint of the manufacturing facility. The reliance on precious metal catalysts such as ruthenium in oxidation steps further exacerbates the cost structure, making the final product less economically viable in a competitive global market. These conventional approaches also suffer from poor atom economy and generate substantial amounts of industrial wastewater, requiring complex and expensive treatment systems to meet environmental regulations. Consequently, the lead time for high-purity pharmaceutical intermediates produced via these legacy methods is often extended due to the need for rigorous safety checks and extensive purification steps.
The Novel Approach
In stark contrast to the hazardous and costly legacy methods, the novel approach disclosed in the patent utilizes a streamlined three-step sequence involving substitution, hydrolysis, and a unique metal oxide catalyzed decarboxylation. This innovative route eliminates the need for dangerous reagents like liquid bromine or sodium hydride, thereby drastically simplifying the safety protocols required for commercial scale-up of complex fluorinated intermediates. The use of common protonic acids for hydrolysis and inexpensive metal oxides for decarboxylation ensures that the raw material costs are significantly reduced while maintaining high reaction selectivity. This method allows for the direct conversion of readily available starting materials into the target acid with minimal byproduct formation, enhancing the overall efficiency of the production line. By avoiding the use of transition metals that require complex removal steps, the process ensures a cleaner final product profile, which is critical for meeting the stringent purity specifications demanded by regulatory bodies. This strategic shift in synthetic design provides a robust foundation for reducing lead time for high-purity pharmaceutical intermediates and ensures a more stable supply chain for downstream API manufacturers.
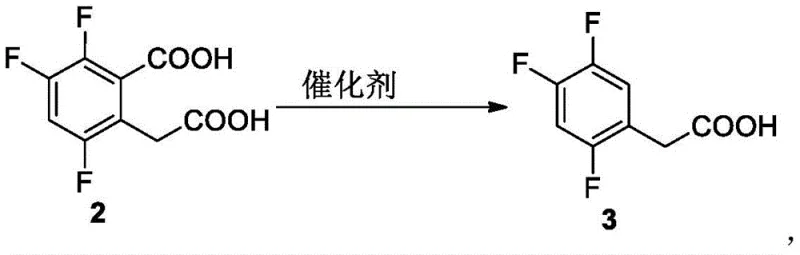
Mechanistic Insights into Metal Oxide Catalyzed Decarboxylation
The core innovation of this synthesis lies in the decarboxylation step, where a metal oxide catalyst facilitates the removal of a carboxyl group under relatively mild thermal conditions. Unlike traditional thermal decarboxylation which often requires extreme temperatures and pressures, the presence of catalysts such as silver oxide or cuprous oxide lowers the activation energy required for the reaction to proceed efficiently. This catalytic mechanism ensures that the reaction selectivity is maintained, preventing the degradation of the sensitive fluorinated aromatic ring which is crucial for the biological activity of the final API. The catalyst interacts with the dicarboxylic acid precursor to stabilize the transition state, allowing for the selective release of carbon dioxide while preserving the integrity of the adjacent functional groups. This level of control is essential for minimizing the formation of impurities that could complicate downstream purification and affect the quality of the Sitagliptin intermediate. The ability to operate at temperatures between 120-220°C without the need for high-pressure equipment further enhances the safety and scalability of the process for industrial applications.
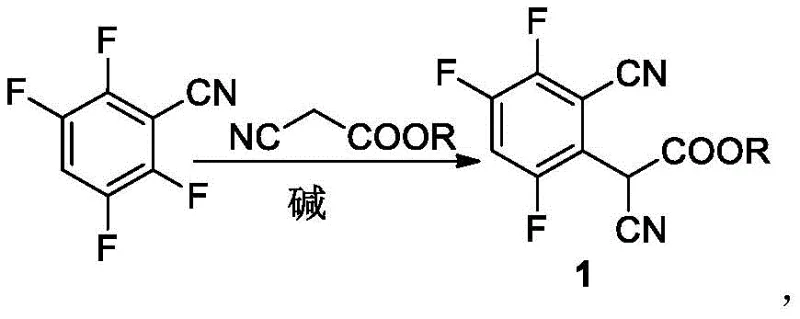
Complementing the decarboxylation step is the initial nucleophilic substitution reaction, which sets the foundation for the entire synthetic sequence by establishing the carbon framework of the molecule. The reaction between 2,3,5,6-tetrafluorobenzonitrile and cyanoalkanoic acid esters proceeds with high efficiency in the presence of common organic or inorganic bases, ensuring a robust start to the synthesis. This step is critical for introducing the side chain that will eventually become the acetic acid moiety, and the high yield reported in the patent examples demonstrates the reliability of this transformation. The subsequent hydrolysis step converts the nitrile and ester groups into carboxylic acids using protonic acid, a process that is both cost-effective and environmentally friendlier than alkaline hydrolysis methods. The combination of these mechanistic steps creates a cohesive pathway that maximizes atom economy and minimizes waste generation, aligning with modern green chemistry principles. For technical teams, understanding these mechanistic details is key to optimizing the process parameters for commercial scale-up of complex fluorinated intermediates.
How to Synthesize 2,4,5-Trifluorophenylacetic Acid Efficiently
The practical implementation of this synthesis route involves a carefully controlled sequence of reactions that can be adapted for large-scale manufacturing environments with minimal modification. The process begins with the substitution reaction in an organic solvent, followed by a hydrolysis step under acidic conditions, and concludes with the catalytic decarboxylation to yield the final product. Each step has been optimized to ensure high conversion rates and purity, making it an ideal candidate for facilities looking to enhance their production capabilities for fluorinated building blocks. The detailed standardized synthesis steps see the guide below provide a comprehensive framework for technical teams to replicate the success reported in the patent data. By adhering to the specified temperature ranges and catalyst loading ratios, manufacturers can achieve consistent results that meet the rigorous quality standards required for pharmaceutical applications. This structured approach ensures that the transition from laboratory scale to industrial production is smooth and efficient, minimizing the risks associated with process scaling.
- Perform nucleophilic substitution on 2,3,5,6-tetrafluorobenzonitrile with cyanoalkanoic acid ester using a base.
- Hydrolyze the resulting intermediate under protonic acid conditions at 100-220°C to form the dicarboxylic acid precursor.
- Execute decarboxylation using a metal oxide catalyst such as silver oxide or cuprous oxide to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this novel synthesis route offers substantial benefits that directly address the pain points of procurement managers and supply chain heads in the fine chemical industry. The elimination of expensive and hazardous reagents translates into a significant reduction in raw material costs, allowing for more competitive pricing structures without sacrificing margin. Furthermore, the simplified process flow reduces the complexity of the manufacturing operation, which enhances supply chain reliability by minimizing the potential for production delays caused by safety incidents or equipment failures. The use of readily available starting materials ensures that the supply of key inputs remains stable, reducing the risk of bottlenecks that can disrupt the delivery of critical intermediates to API manufacturers. This stability is crucial for maintaining continuous production schedules and meeting the demanding timelines of the pharmaceutical sector. Additionally, the environmental benefits of the process, such as reduced waste generation, align with corporate sustainability goals and can lead to lower compliance costs associated with waste disposal and environmental regulations.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with inexpensive metal oxides drastically lowers the cost of goods sold, while the high yield of the reaction minimizes material waste and maximizes output per batch. The simplified purification process further reduces operational expenses by cutting down on solvent usage and energy consumption during downstream processing. This economic efficiency makes the route highly attractive for cost-sensitive projects where margin optimization is a primary objective for the business.
- Enhanced Supply Chain Reliability: By utilizing common and commercially available reagents, the process mitigates the risk of supply disruptions that are often associated with specialized or hazardous chemicals. The robust nature of the reaction conditions ensures that production can be maintained consistently, providing a dependable source of high-purity intermediates for downstream customers. This reliability is essential for building long-term partnerships and securing contracts with major pharmaceutical companies that require guaranteed supply continuity.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic reagents make the process highly scalable, allowing for easy expansion of production capacity to meet growing market demand. The reduced environmental footprint facilitates easier compliance with increasingly strict environmental regulations, reducing the administrative burden and potential fines associated with non-compliance. This alignment with green chemistry principles enhances the corporate image and opens up opportunities in markets that prioritize sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, providing clarity for stakeholders evaluating its potential for their supply chains. These answers are derived directly from the technical data and beneficial effects disclosed in the patent, ensuring accuracy and relevance for decision-makers. Understanding these aspects is crucial for assessing the feasibility of adopting this technology for commercial production of fluorinated intermediates. The insights provided here aim to bridge the gap between technical specifications and business strategy, enabling informed decisions regarding procurement and process development.
Q: What are the safety advantages of this new synthesis route?
A: The new method eliminates the need for hazardous Grignard reagents and toxic sodium cyanide, significantly reducing explosion risks and environmental hazards associated with traditional pathways.
Q: How does the metal oxide catalyst improve cost efficiency?
A: By using inexpensive metal oxides like cuprous oxide instead of precious metal catalysts, the process drastically lowers raw material costs and simplifies catalyst recovery.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the mild reaction conditions and high selectivity make it highly adaptable for commercial scale-up, ensuring consistent quality and supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trifluorophenylacetic Acid Supplier
As a leading CDMO expert, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like this one can be successfully translated into industrial reality. Our team is equipped with rigorous QC labs and the capability to meet stringent purity specifications, guaranteeing that every batch of 2,4,5-Trifluorophenylacetic Acid meets the highest quality standards required for pharmaceutical applications. We understand the critical importance of supply continuity and cost efficiency, and we are committed to delivering solutions that optimize your manufacturing process while maintaining full regulatory compliance. Our technical expertise allows us to troubleshoot potential scale-up issues proactively, ensuring a smooth transition from development to full-scale production.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. By initiating a dialogue with us, you can obtain a Customized Cost-Saving Analysis that demonstrates how implementing this novel synthesis route can enhance your competitive position in the market. Our dedicated support team is ready to assist you in evaluating the technical and commercial viability of this intermediate for your specific API synthesis needs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
