Advanced Sodium-Mediated Enaminone Synthesis for Scalable Pharmaceutical Manufacturing
Advanced Sodium-Mediated Enaminone Synthesis for Scalable Pharmaceutical Manufacturing
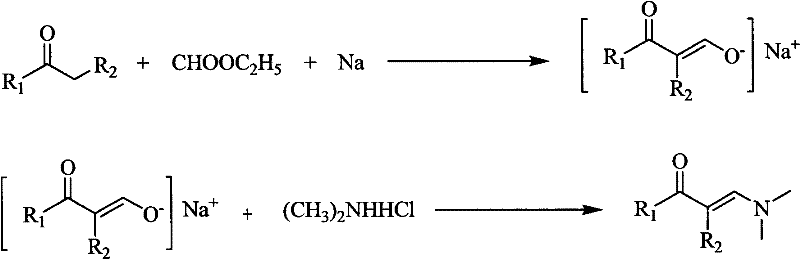
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for more sustainable and cost-effective synthetic routes for critical intermediates. Patent CN101781222A introduces a transformative methodology for the preparation of enaminone compounds, specifically targeting those with two substituents on the nitrogen atom. This innovation leverages a robust alkali metal-mediated formylation strategy, bypassing the complexities associated with traditional transition metal catalysis. By utilizing common ketones with alpha-methyl or alpha-methylene groups as substrates, the process generates alkali metal enolates which are subsequently condensed with dimethylamine hydrochloride. This approach not only streamlines the synthetic pathway but also aligns perfectly with modern green chemistry principles by operating under mild reaction conditions. For global procurement teams and R&D directors, this patent represents a significant opportunity to optimize supply chains for nitrogen-containing heterocyclic precursors. The technical depth of this method ensures high selectivity and yield, addressing the perennial challenges of impurity control in complex molecule synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of enaminone compounds has relied heavily on condensation reactions between beta-diketones and substituted amines, often necessitating harsh conditions that complicate industrial scalability. Prior art, such as USP 4579675, frequently requires the use of toluene or benzene for azeotropic water removal, introducing significant safety hazards and environmental burdens due to the toxicity of these solvents. Furthermore, these conventional routes often suffer from prolonged reaction times and yields that are unpredictably sensitive to the steric and electronic nature of the substituents involved. The reliance on Lewis acids, while sometimes improving conversion rates, creates a downstream bottleneck regarding catalyst recovery and reuse, which inflates the overall cost of goods sold. Additionally, methods involving terminal alkynes and palladium catalysts, as noted in Synthesis 2003, are prohibitively expensive for large-scale applications due to the high cost of precious metals and the stringent requirement for inert atmospheres. These legacy processes collectively impose a heavy burden on manufacturing efficiency, limiting the ability of suppliers to respond rapidly to market demand fluctuations.
The Novel Approach
In stark contrast, the methodology disclosed in CN101781222A offers a streamlined alternative that fundamentally reshapes the economic and operational parameters of enaminone production. By shifting the paradigm to an alkali metal-mediated formylation followed by dehydration condensation, the process eliminates the need for expensive transition metal catalysts and toxic azeotropic solvents. The reaction proceeds efficiently at mild temperatures ranging from 0°C to 50°C, with a preferred window of 20°C to 30°C, drastically reducing energy consumption and thermal stress on the equipment. The use of dimethylamine hydrochloride as the amine source in an aqueous two-phase system further enhances safety and simplifies the workup procedure compared to anhydrous amine handling. This novel route demonstrates exceptional versatility, accommodating a wide array of aliphatic and aromatic ketones without compromising on yield or purity. Consequently, this approach provides a robust foundation for reliable agrochemical intermediate supplier networks and pharmaceutical manufacturers seeking to de-risk their production pipelines.
Mechanistic Insights into Alkali Metal-Mediated Formylation
The core of this synthetic breakthrough lies in the precise generation and stabilization of the alkali metal enolate intermediate, which serves as the nucleophilic engine for the subsequent transformation. In the first stage, an alkali metal, typically sodium, is suspended in an organic solvent such as diethyl ether, creating a highly reactive surface for the formylation event. Upon the dropwise addition of the ketone substrate and ethyl formate, the alpha-proton of the ketone is abstracted, leading to the formation of a resonance-stabilized enolate anion coordinated with the sodium cation. This step is critical, as the molar ratio of ketone to ethyl formate is carefully maintained between 1:1 and 1:10, with a preferred ratio of 1:3, to ensure complete conversion while minimizing side reactions. The presence of the organic solvent plays a dual role, acting both as a dispersant for the solid metal and a solubilizer for the resulting enolate salt, thereby maintaining a homogeneous reaction environment that facilitates smooth kinetics.
Following the formation of the enolate, the mechanism progresses to a dehydration condensation phase upon the introduction of dimethylamine hydrochloride. The aqueous nature of the amine salt solution creates a biphasic system that moderates the reaction rate, preventing runaway exotherms and ensuring high selectivity for the desired enaminone product. The enolate oxygen acts as a leaving group in a concerted elimination process, driven by the nucleophilic attack of the dimethylamine nitrogen on the carbonyl carbon. This mechanistic pathway effectively suppresses the formation of polymeric byproducts often seen in acid-catalyzed condensations, resulting in a cleaner crude profile that is easier to purify. The final isolation involves standard separation techniques such as extraction and drying, followed by purification via recrystallization or vacuum distillation, yielding products with high structural integrity.
How to Synthesize Enaminone Compounds Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and physical state of the reagents to maximize the efficiency of the enolate formation. The process begins with the preparation of a suspension of the alkali metal in a dry, aprotic solvent, followed by the controlled addition of the ketone and formylating agent under inert conditions. Once the enolate is fully generated, indicated by the cessation of gas evolution or color change, the amine salt solution is introduced to trigger the cyclization and dehydration sequence. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Suspend alkali metal (e.g., Sodium) in an organic solvent like diethyl ether and react with a ketone having alpha-methyl/methylene groups and ethyl formate at 0-50°C to form an alkali metal enolate.
- Add an aqueous solution of dimethylamine hydrochloride to the enolate suspension at 0-60°C to facilitate dehydration and condensation, yielding the crude enaminone.
- Separate the organic layer, dry over anhydrous magnesium sulfate, and purify the final product via vacuum distillation or recrystallization using petroleum ether and acetone.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this sodium-mediated synthesis route offers profound advantages that directly impact the bottom line and supply chain resilience for chemical buyers. The elimination of precious metal catalysts and the use of commodity chemicals like sodium metal and ethyl formate drastically reduce the raw material cost base, making the final enaminone intermediates significantly more price-competitive in the global market. Furthermore, the mild reaction conditions negate the need for specialized high-pressure or high-temperature reactors, allowing production to be shifted to multipurpose facilities with lower capital expenditure requirements. This flexibility enhances supply chain reliability by diversifying the potential manufacturing base and reducing dependency on single-source technology providers. The simplified purification process also translates to shorter cycle times and reduced solvent waste, contributing to a more sustainable and cost-efficient manufacturing footprint.
- Cost Reduction in Manufacturing: The substitution of expensive palladium or Lewis acid catalysts with inexpensive alkali metals results in substantial cost savings on reagent procurement and waste disposal. By avoiding the complex recovery protocols associated with transition metals, manufacturers can streamline their operational workflows and reduce the overhead costs linked to catalyst regeneration. The use of common solvents like diethyl ether and water further minimizes solvent purchasing costs compared to specialized anhydrous or toxic solvents required by legacy methods. This economic efficiency allows for more aggressive pricing strategies while maintaining healthy margins, providing a distinct competitive advantage in cost reduction in pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals such as sodium, ethyl formate, and dimethylamine hydrochloride ensures a stable supply of raw materials that is less susceptible to geopolitical disruptions or market volatility. Unlike processes dependent on niche ligands or custom-synthesized catalysts, this method leverages a supply chain that is robust and well-established globally. The simplicity of the equipment requirements means that production can be easily scaled or transferred between different sites without significant retooling investments. This adaptability is crucial for reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream drug manufacturers receive their materials on schedule.
- Scalability and Environmental Compliance: The mild thermal profile of the reaction, operating effectively at ambient temperatures, significantly lowers the energy intensity of the process, aligning with corporate sustainability goals and regulatory pressure to reduce carbon footprints. The absence of toxic benzene or toluene azeotropes simplifies wastewater treatment and VOC management, reducing the environmental compliance burden on the manufacturing site. The high selectivity of the reaction minimizes the generation of hazardous byproducts, facilitating easier waste stream management and lowering disposal costs. These factors combined make the commercial scale-up of complex enaminone derivatives not only technically feasible but also environmentally responsible and economically viable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enaminone synthesis technology, derived directly from the patent specifications and experimental data. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing process portfolios. The answers provided reflect the specific advantages and operational parameters detailed in the intellectual property, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of this sodium-mediated method over traditional Lewis acid catalysis?
A: Unlike traditional methods requiring expensive Lewis acids or azeotropic water removal with benzene, this method uses common alkali metals and mild temperatures (20-30°C), significantly simplifying equipment requirements and reducing catalyst recovery costs.
Q: Is this process suitable for large-scale industrial production of enaminones?
A: Yes, the patent explicitly states the process avoids large expensive equipment and uses readily available raw materials like ethyl formate and dimethylamine hydrochloride, making it highly adaptable for commercial scale-up from kilograms to metric tons.
Q: What types of ketone substrates are compatible with this synthesis route?
A: The method is versatile and accommodates various aliphatic and aromatic ketones possessing alpha-methyl or alpha-methylene groups, including acetone, acetophenone, and substituted acetophenones, ensuring broad applicability for diverse intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enaminone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic methodologies in securing a stable supply of high-value intermediates for the global pharmaceutical industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify identity and assay. By leveraging the efficiencies of the sodium-mediated formylation process, we can offer our partners a reliable enaminone supplier relationship that balances cost, quality, and delivery performance.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. Contact us today to request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener, more efficient manufacturing platform. Our specialists are ready to provide specific COA data and route feasibility assessments to support your next development milestone, ensuring your supply chain remains resilient and competitive in a dynamic market.
