Revolutionizing Benzofuran Production: Iron-Catalyzed C-H Activation for Commercial Scale
The landscape of heterocyclic chemistry is undergoing a significant transformation with the disclosure of patent CN112812084B, which details a novel synthetic methodology for benzofuran compounds. This intellectual property introduces a groundbreaking approach utilizing economical iron phthalocyanine as a catalyst, effectively replacing traditional noble metal systems. Benzofuran scaffolds are ubiquitous in medicinal chemistry, serving as critical pharmacophores in candidates targeting HIV, cancer, and cardiovascular diseases. The shift towards base-metal catalysis represents a paradigm shift for the industry, addressing long-standing concerns regarding the cost and environmental impact of precious metal residues in active pharmaceutical ingredients. By leveraging the unique electronic properties of iron complexes, this technology enables the efficient construction of complex oxygen-containing heterocycles from readily available salicylaldehydes and sulfur ylides.
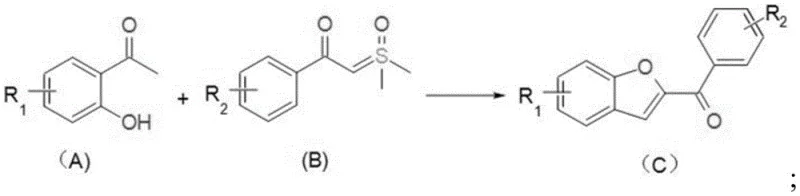
Historically, the construction of benzofuran cores via C-H activation has been dominated by expensive transition metals such as Rhodium(III) and Ruthenium(II). As illustrated in prior art literature, these conventional methods often necessitate harsh conditions, including high temperatures up to 120°C, inert atmospheres, and stoichiometric amounts of silver salts as oxidants. For instance, Rhodium-catalyzed relay carbenoid functionalization requires complex catalytic systems involving pentamethylcyclopentadienyl rhodium dimers and anhydrous copper acetate additives. These reliance on scarce resources not only inflates the bill of materials but also introduces significant supply chain vulnerabilities. Furthermore, the removal of trace rhodium or ruthenium to meet stringent regulatory limits for drug substances adds costly purification steps, such as scavenger resin treatments or extensive chromatography, which severely impact overall process throughput and yield.
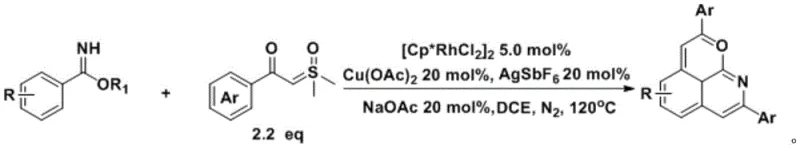
The novel approach described in the patent circumvents these limitations by employing iron phthalocyanine, an earth-abundant and non-toxic catalyst. This system operates efficiently in tetrahydrofuran (THF) at moderate temperatures around 100°C, remarkably proceeding under air atmosphere rather than requiring rigorous nitrogen protection. The reaction utilizes potassium carbonate as a benign base, avoiding the need for expensive silver oxidants or specialized ligands. This simplification of the reaction matrix is a major technical breakthrough, as it reduces the number of unit operations required during workup. The compatibility of this catalytic system with a broad spectrum of substrates, including those bearing sensitive halogen and methoxy substituents, underscores its robustness. By eliminating the dependency on precious metals, manufacturers can achieve substantial cost reductions while simultaneously improving the sustainability profile of their synthetic routes.
Mechanistic Insights into FePc-Catalyzed Cyclization
The mechanistic pathway of this iron-catalyzed transformation involves a sophisticated interplay between the metal center and the sulfur ylide species. Unlike noble metals that often rely on high-valent organometallic intermediates, the iron phthalocyanine catalyst likely facilitates the generation of a reactive carbene-like species from the sulfoxonium ylide through a single-electron transfer or coordination mechanism. This activated intermediate then undergoes nucleophilic attack by the phenolic hydroxyl group of the salicylaldehyde, followed by intramolecular cyclization to form the furan ring. The planar structure of the phthalocyanine ligand provides a stable environment for the iron center, preventing catalyst deactivation and ensuring turnover numbers sufficient for practical application. Understanding this mechanism is crucial for process chemists aiming to optimize reaction parameters, as it highlights the importance of maintaining the oxidation state of the iron throughout the catalytic cycle to prevent precipitation of inactive iron oxides.
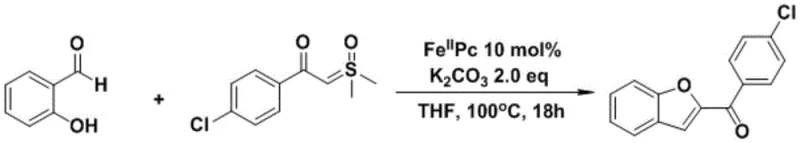
Impurity control is another critical aspect where this iron-catalyzed method excels compared to traditional routes. In noble metal catalysis, side reactions such as homocoupling of the ylide or over-oxidation of the aldehyde can lead to complex impurity profiles that are difficult to separate. The specific coordination geometry of the iron phthalocyanine appears to direct the reaction selectively towards the desired annulation product, minimizing the formation of byproducts. Additionally, the use of potassium carbonate as a mild base reduces the risk of base-mediated degradation of sensitive functional groups on the aromatic rings. This selectivity translates directly to higher crude purity, which simplifies the final crystallization or chromatography steps. For R&D teams, this means a more predictable impurity fate and purge profile, facilitating faster regulatory filing and approval timelines for new drug candidates containing the benzofuran motif.
How to Synthesize Acylated Benzofurans Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the sulfur ylide precursor, followed by the one-pot coupling with the salicylaldehyde derivative. The reaction conditions are forgiving, tolerating slight variations in stoichiometry and temperature without significant loss of yield. Detailed standardized operating procedures for scaling this reaction from gram to kilogram scale are essential for ensuring reproducibility and safety. The following guide outlines the critical steps for executing this transformation effectively, ensuring that technical teams can replicate the high yields reported in the patent data while maintaining strict quality control standards throughout the production batch.
- Preparation of sulfur ylide precursors via reaction of acyl chlorides with trimethyl sulfoxide iodide.
- Combine salicylaldehyde derivative, sulfur ylide, Fe(II)Pc catalyst (10 mol%), and K2CO3 (2.0 eq) in THF solvent.
- Heat the reaction mixture at 100°C for 18 hours under air atmosphere, followed by standard workup and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iron-catalyzed technology offers transformative benefits for procurement strategies and supply chain resilience. The primary driver for value creation is the elimination of precious metal catalysts, which are subject to volatile market pricing and geopolitical supply risks. By switching to iron phthalocyanine, manufacturers can decouple their production costs from the fluctuating prices of rhodium and ruthenium. This stability allows for more accurate long-term budgeting and pricing contracts with downstream pharmaceutical clients. Moreover, the simplified reaction conditions reduce the demand for specialized infrastructure, such as high-pressure reactors or advanced inert gas handling systems, thereby lowering capital expenditure requirements for new production lines.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with inexpensive iron salts results in a direct and significant decrease in raw material costs. Beyond the catalyst itself, the removal of silver oxidants and complex ligands further reduces the bill of materials. This cost efficiency is compounded by the simplified downstream processing; without the need for extensive heavy metal scavenging, the consumption of purification resins and solvents is drastically reduced. Consequently, the overall cost of goods sold (COGS) for the benzofuran intermediate is lowered, enhancing profit margins and allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Iron is one of the most abundant elements on earth, ensuring a secure and continuous supply of the catalyst compared to scarce platinum group metals. The starting materials, salicylaldehydes and sulfur ylides, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source bottlenecks. This diversification of the supply base enhances the resilience of the manufacturing network against disruptions. Furthermore, the ability to run the reaction in air eliminates the need for bulk nitrogen or argon supplies, simplifying logistics and reducing utility dependencies at the manufacturing site, which is critical for maintaining uninterrupted production schedules.
- Scalability and Environmental Compliance: The mild reaction conditions and use of common solvents like THF make this process highly scalable from laboratory to commercial tonnage. The absence of toxic heavy metals simplifies waste stream management, as effluent treatment does not require specialized protocols for removing trace rhodium or ruthenium. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The reduced environmental footprint not only mitigates regulatory risk but also appeals to eco-conscious partners in the pharmaceutical value chain. The robustness of the process ensures consistent quality at scale, supporting the reliable delivery of high-purity intermediates required for clinical and commercial drug manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed benzofuran synthesis. These answers are derived directly from the experimental data and comparative studies presented in the patent documentation. They provide clarity on the operational flexibility, substrate compatibility, and economic rationale behind adopting this new methodology. Understanding these details is vital for technical decision-makers evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is Iron Phthalocyanine preferred over Rhodium or Ruthenium catalysts for this synthesis?
A: Iron phthalocyanine offers a drastic reduction in raw material costs compared to precious metals like Rhodium and Ruthenium. Furthermore, it eliminates the need for rigorous heavy metal removal steps required for pharmaceutical grade intermediates, simplifying downstream processing and ensuring compliance with strict impurity profiles.
Q: What is the substrate scope of this iron-catalyzed benzofuran synthesis?
A: The methodology demonstrates exceptional versatility, accommodating a wide array of functional groups on both the salicylaldehyde and sulfur ylide components. Electron-donating groups like methyl and methoxy, as well as electron-withdrawing halogens such as fluorine, chlorine, and bromine, are all well-tolerated, yielding products with high efficiency.
Q: Are the reaction conditions suitable for large-scale industrial manufacturing?
A: Yes, the process operates under relatively mild thermal conditions (100°C) in common solvents like THF and does not require inert gas protection, reacting successfully in air. These factors significantly lower the barrier for commercial scale-up, enhancing operational safety and reducing energy consumption compared to high-pressure or cryogenic alternatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of adopting innovative catalytic technologies to maintain a competitive edge in the pharmaceutical intermediate market. Our team of expert process chemists has extensively evaluated the iron phthalocyanine catalyzed route described in CN112812084B and confirmed its potential for robust commercial manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of benzofuran derivative delivered meets the highest international standards for drug substance manufacturing.
We invite potential partners to engage with our technical procurement team to discuss how this cost-effective synthesis route can be tailored to your specific project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this iron-catalyzed process for your specific molecule. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our manufactured intermediates against your internal benchmarks. Let us help you optimize your supply chain and accelerate your drug development timeline with our reliable, high-quality benzofuran solutions.
