Advanced Organocatalytic Synthesis of Chiral Nitrogen Heterocycles for Pharmaceutical Applications
Introduction to Breakthrough Organocatalytic Technology
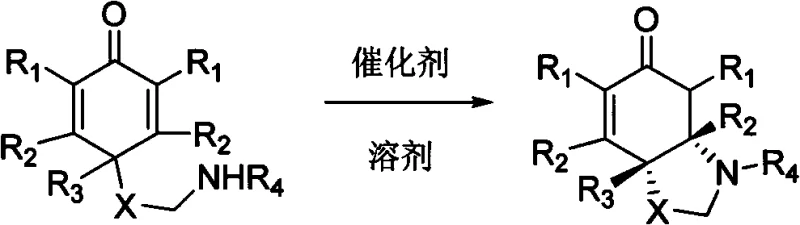
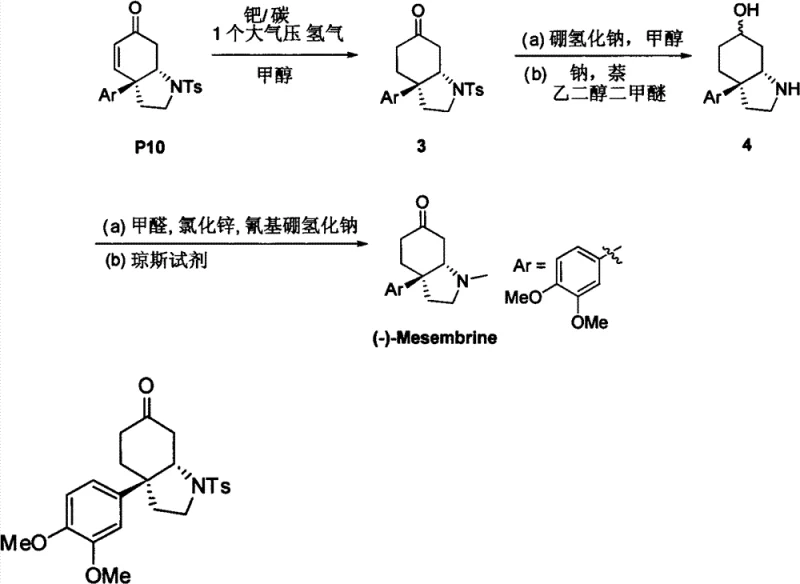
The pharmaceutical industry is constantly seeking more efficient and environmentally benign methods for constructing complex chiral scaffolds, particularly nitrogen-containing heterocycles which serve as the backbone for countless bioactive molecules. Patent CN102153501B introduces a transformative approach to synthesizing chiral pyrrolidine, piperidine, and morpholine compounds through an intramolecular aza-Michael reaction. Unlike traditional methods that rely heavily on stoichiometric chiral auxiliaries or expensive transition metal complexes, this innovation utilizes chiral phosphoric acids or chiral thioureas as organocatalysts. This shift represents a paradigm change in fine chemical manufacturing, offering a pathway to high-purity intermediates without the burden of heavy metal residue removal. The technology is particularly notable for its application in the total synthesis of the biologically active alkaloid (-)-Mesembrine, demonstrating its practical utility in producing high-value pharmaceutical ingredients.
For R&D directors and process chemists, the significance of this patent lies in its operational simplicity and robustness. The reaction proceeds under mild conditions, typically ranging from -60°C to 25°C, and tolerates a wide variety of functional groups on the cyclohexadienone substrate. The absence of metal salts not only aligns with green chemistry principles but also drastically reduces the complexity of the workup procedure. By leveraging hydrogen-bonding activation modes, these organocatalysts achieve remarkable levels of stereocontrol, with reported enantiomeric excess values consistently falling between 89% and 99%. This level of precision is critical for ensuring the biological efficacy and safety of the final drug product, making this technology a cornerstone for modern asymmetric synthesis strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of chiral quaternary carbon centers in nitrogen heterocycles has been a formidable challenge in organic synthesis. Conventional strategies often depend on the use of chiral auxiliaries, which require stoichiometric amounts of expensive chiral starting materials and additional synthetic steps for attachment and removal. This inherently lowers the overall atom economy and increases the cost of goods sold (COGS). Furthermore, transition metal-catalyzed asymmetric reactions, while effective, introduce significant regulatory hurdles. The presence of residual metals such as palladium, rhodium, or iridium in the final Active Pharmaceutical Ingredient (API) is strictly regulated by agencies like the FDA and EMA. Removing these traces to parts-per-million levels often necessitates specialized scavenging resins or complex crystallization protocols, adding time and cost to the manufacturing timeline. Additionally, many metal-catalyzed processes require stringent anhydrous conditions or cryogenic temperatures, complicating scale-up efforts.
The Novel Approach
The methodology described in CN102153501B circumvents these issues by employing metal-free organocatalysis. The core transformation involves the cyclization of cyclohexadienone derivatives bearing a pendant amine group. As illustrated in the reaction scheme, the chiral catalyst activates the substrate through a network of hydrogen bonds, facilitating the nucleophilic attack of the nitrogen atom onto the enone system with high facial selectivity. This approach eliminates the need for stoichiometric chiral reagents, as the catalyst is used in sub-stoichiometric amounts (typically 5-20 mol%). The reaction is operationally simple, often requiring only mixing of reagents in common solvents like dichloromethane at room temperature or mild reflux. The resulting products, such as the chiral pyrrolidine derivatives, are obtained in excellent yields ranging from 73% to 97%. This efficiency, combined with the absence of toxic metals, positions this technology as a superior alternative for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Organocatalytic Asymmetric Aza-Michael Reaction
The success of this synthesis relies on the precise molecular recognition between the chiral catalyst and the substrate. In the case of chiral thiourea catalysts, such as the cinchona alkaloid-derived structures exemplified in the patent, the dual hydrogen-bond donor motif interacts with the carbonyl oxygen of the cyclohexadienone. This interaction lowers the LUMO energy of the electrophile, making it more susceptible to nucleophilic attack. Simultaneously, the basic tertiary amine moiety of the catalyst may assist in deprotonating or organizing the nucleophilic amine chain. For chiral phosphoric acids, the mechanism involves Brønsted acid activation, where the acidic proton activates the enone while the phosphoryl oxygen stabilizes the developing charge in the transition state. This bifunctional activation creates a rigid chiral environment that dictates the trajectory of the incoming nucleophile, thereby controlling the absolute configuration of the newly formed stereocenter.
Understanding this mechanism is vital for impurity control. The high enantioselectivity (up to 99% ee) implies that the formation of the undesired enantiomer is kinetically suppressed. In a manufacturing context, this means fewer impurities to separate during purification, leading to higher overall purity of the final API. The patent demonstrates the versatility of this mechanism by successfully synthesizing a library of derivatives (P1-P20) with varying substituents on the aromatic rings and the nitrogen protecting groups. Whether the substrate contains electron-withdrawing groups like nitro or halogens, or electron-donating groups like methoxy, the catalytic system maintains high performance. This robustness suggests that the transition state is well-insulated from steric and electronic perturbations, a desirable trait for a reliable pharmaceutical intermediate supplier aiming to produce diverse analogues.
How to Synthesize Chiral Nitrogen-Containing Heterocycles Efficiently
The practical implementation of this technology involves a straightforward protocol suitable for both laboratory discovery and pilot plant operations. The process begins with the preparation of the cyclohexadienone precursor, which is then subjected to the cyclization conditions. Detailed standard operating procedures regarding reagent grades, solvent drying, and specific workup techniques are essential for reproducing the high enantiomeric excess reported in the literature. The following guide outlines the generalized steps derived from the patent examples to ensure consistent quality and yield.
- Prepare the reaction system by dissolving cyclohexadienone derivatives in an organic solvent such as dichloromethane under inert atmosphere.
- Add chiral phosphoric acid or chiral thiourea catalyst (5-20 mol%) to the solution and stir at temperatures ranging from -60°C to 25°C.
- Monitor reaction progress via TLC, then purify the resulting chiral heterocyclic product using column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic route offers tangible economic and logistical benefits. The most significant advantage is the elimination of heavy metal catalysts. In traditional manufacturing, the cost associated with metal scavengers, specialized filtration equipment, and extensive analytical testing for metal residues can be substantial. By switching to a metal-free process, companies can achieve significant cost reduction in API manufacturing. The simplified purification process also translates to shorter cycle times, allowing for faster turnaround from raw material to finished intermediate. This efficiency is crucial for maintaining lean inventory levels and responding quickly to market demand fluctuations.
- Cost Reduction in Manufacturing: The use of organocatalysts removes the dependency on precious metals like palladium or rhodium, whose prices are volatile and often high. Furthermore, the high yields (73%-97%) minimize raw material waste, directly improving the mass balance of the process. The ability to run reactions at ambient pressure and near-room temperature reduces energy consumption compared to high-pressure hydrogenation or cryogenic reactions. These factors collectively contribute to a lower cost of goods, enhancing the competitiveness of the final pharmaceutical product in the global market.
- Enhanced Supply Chain Reliability: Organocatalysts are typically small organic molecules that are stable and easy to store, unlike sensitive metal complexes that may require inert atmosphere storage or cold chain logistics. This stability ensures a consistent supply of the catalyst, reducing the risk of production stoppages due to reagent degradation. Additionally, the broad substrate scope means that a single catalytic platform can be used to generate a wide range of intermediates, simplifying the supply chain by reducing the number of unique reagents that need to be sourced and qualified.
- Scalability and Environmental Compliance: The reaction conditions are inherently safer, avoiding the use of pyrophoric reagents or high-pressure hydrogen gas often found in alternative reduction methods. This safety profile facilitates easier scale-up from gram to kilogram scales without requiring major engineering changes. From an environmental perspective, the metal-free nature of the process reduces the generation of hazardous waste streams, aligning with increasingly strict environmental regulations and corporate sustainability goals. This makes the technology attractive for companies aiming to reduce their carbon footprint and improve their E-factor (environmental factor).
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this organocatalytic technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making.
Q: What are the primary advantages of using organocatalysis over transition metal catalysis for chiral amines?
A: Organocatalysis eliminates the risk of heavy metal contamination, which is critical for pharmaceutical regulatory compliance (ICH Q3D). It also simplifies downstream purification processes, reducing production costs and environmental waste.
Q: What level of enantioselectivity can be achieved with this method?
A: The patented method demonstrates high stereocontrol, consistently achieving enantiomeric excess (ee) values between 89% and 99% across various substrates, including pyrrolidines and piperidines.
Q: Can this synthesis route be scaled for commercial production of (-)-Mesembrine?
A: Yes, the reaction conditions are mild (room temperature to reflux) and utilize stable organic catalysts, making the process highly amenable to scale-up from laboratory to multi-kilogram commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Nitrogen Heterocycles Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the one described in CN102153501B. As a leading CDMO partner, we possess the technical expertise to translate these academic breakthroughs into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from benchtop to market. We understand that consistency is key, which is why our rigorous QC labs enforce stringent purity specifications for every batch of chiral intermediates we produce, guaranteeing that the high enantiomeric excess achieved in the lab is maintained at scale.
We invite you to collaborate with us to leverage this metal-free technology for your next drug development program. Whether you require custom synthesis of specific pyrrolidine derivatives or full-scale production of (-)-Mesembrine precursors, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and accelerate your time to market.
