Advanced Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly those bearing trifluoromethyl groups which are pivotal for enhancing metabolic stability and lipophilicity in drug candidates. Patent CN110467579B discloses a groundbreaking preparation method for 5-trifluoromethyl substituted 1,2,4-triazole compounds, addressing critical bottlenecks in current synthetic strategies. This innovation leverages a non-metallic iodine-promoted cyclization that operates under remarkably mild conditions, eliminating the stringent requirement for anhydrous or oxygen-free environments often associated with traditional heterocycle synthesis. The significance of this technology extends beyond academic interest, offering a tangible pathway for the commercial scale-up of complex pharmaceutical intermediates with improved safety profiles and operational simplicity.
The structural versatility of the 1,2,4-triazole scaffold is well-documented, serving as a core motif in numerous bioactive molecules ranging from antifungal agents to kinase inhibitors. As illustrated in the following visual representation, the incorporation of the trifluoromethyl group at the 5-position can dramatically alter the electronic properties of the molecule, thereby optimizing its interaction with biological targets.
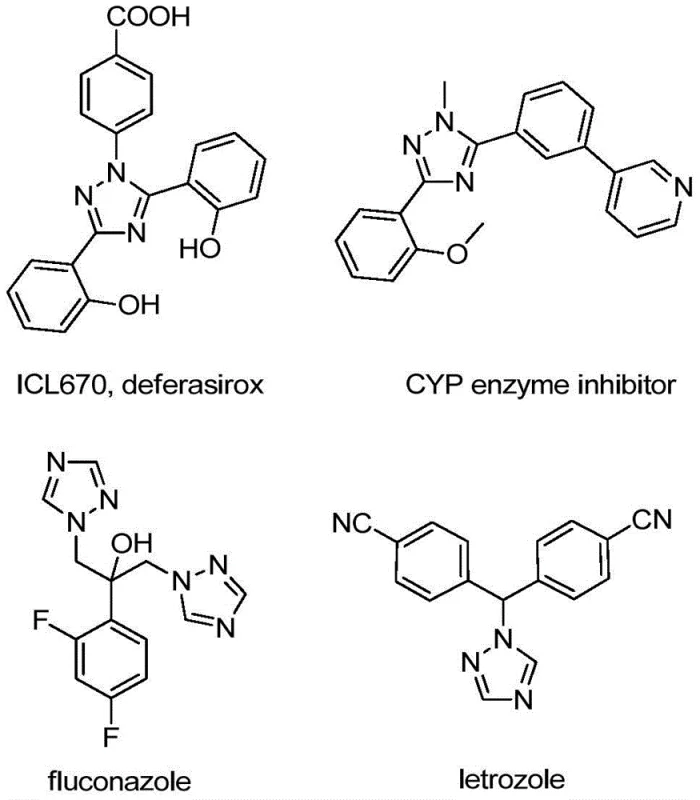
For R&D directors evaluating new synthetic routes, the ability to access diverse substitution patterns at the 4 and 5 positions of the triazole ring is paramount. The disclosed method utilizes readily available hydrazones and trifluoroethylimidoyl chlorides as building blocks, allowing for extensive substrate design. This flexibility ensures that medicinal chemists can rapidly generate libraries of analogues for structure-activity relationship (SAR) studies without being hindered by complex protecting group strategies or incompatible reaction conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of trifluoromethylated nitrogen heterocycles has relied heavily on two primary strategies, both of which present significant drawbacks for large-scale manufacturing. The first approach involves the direct trifluoromethylation of pre-synthesized heterocyclic cores, which necessitates the use of specialized and often hazardous trifluoromethylating reagents that are costly and difficult to source in bulk quantities. The second mainstream method employs synthons like trifluorodiazoethane, which, while effective, poses severe safety risks due to its explosive nature and instability, requiring specialized equipment and rigorous safety protocols that drive up operational expenditures.
Furthermore, many traditional cyclization protocols depend on transition metal catalysts, such as copper or palladium complexes, which introduce the risk of heavy metal contamination in the final active pharmaceutical ingredient (API). Removing these trace metals to meet regulatory standards often requires additional purification steps, such as scavenging or extensive chromatography, which drastically reduces overall yield and increases waste generation. These factors collectively contribute to higher production costs and longer lead times, creating a substantial barrier for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast, the methodology described in patent CN110467579B introduces a paradigm shift by utilizing trifluoroethylimidoyl chloride and hydrazones in an iodine-promoted system. This novel route bypasses the need for dangerous diazo compounds and expensive metal catalysts entirely. The reaction proceeds through a base-promoted condensation followed by an oxidative cyclization mediated by elemental iodine, a reagent that is inexpensive, stable, and easy to handle. The general reaction scheme below highlights the simplicity of transforming these accessible starting materials into the desired triazole core.
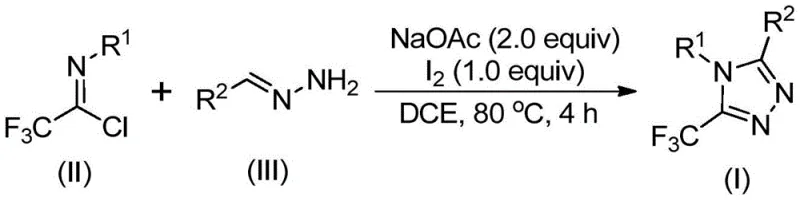
The scope of this reaction is exceptionally broad, accommodating a wide range of substituents on both the hydrazone and the imidoyl chloride components. As demonstrated by the specific examples provided in the patent, various aryl, heteroaryl, and alkenyl groups can be successfully incorporated, yielding products with high purity and excellent yields. This adaptability makes the process an ideal candidate for the production of high-purity pharmaceutical intermediates required for late-stage drug development, where structural diversity is key to optimizing therapeutic efficacy.
Mechanistic Insights into Iodine-Promoted Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for process optimization and troubleshooting. The reaction is believed to initiate with a base-promoted intermolecular carbon-nitrogen bond formation between the hydrazone and the trifluoroethylimidoyl chloride, generating a trifluoroacetamidine intermediate. This species subsequently undergoes isomerization to align the reactive centers for cyclization. The addition of elemental iodine then triggers an oxidative iodination step, creating an electrophilic iodine species that facilitates the subsequent ring closure.
Following the formation of the iodinated intermediate, an intramolecular electrophilic substitution occurs, leading to the closure of the five-membered triazole ring. The final step involves aromatization, driven by the elimination of hydrogen iodide, to furnish the stable 5-trifluoromethyl-1,2,4-triazole product. This mechanism avoids the formation of radical species that often lead to polymerization or side reactions in metal-catalyzed processes, thereby ensuring a cleaner impurity profile. The absence of transition metals also means that the final product is free from metal residues, simplifying the regulatory approval process for downstream applications.
From an impurity control perspective, the mild reaction conditions (80°C to 100°C) minimize thermal degradation of sensitive functional groups. The use of sodium acetate as a mild base further prevents the hydrolysis of the imidoyl chloride or the decomposition of the hydrazone, which are common side reactions in harsher alkaline environments. This precise control over the reaction environment allows for the synthesis of complex molecules with multiple functional groups, such as halogens or nitro groups, without compromising the integrity of the final structure.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazoles Efficiently
The operational simplicity of this protocol makes it highly attractive for both laboratory-scale discovery and pilot-plant production. The procedure involves a straightforward one-pot sequence where reagents are mixed in a common organic solvent like dichloroethane, heated, and then treated with iodine. Detailed standardized synthesis steps for implementing this technology in your facility are outlined in the guide below.
- Mix sodium acetate, trifluoroethylimidoyl chloride, and hydrazone in an organic solvent such as dichloroethane.
- Heat the reaction mixture to 80°C and maintain for 2 to 4 hours to allow initial condensation.
- Add elemental iodine to the system and continue heating for an additional 1 to 2 hours to complete the oxidative cyclization.
- Perform post-treatment including filtration and silica gel purification to isolate the final triazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits. By shifting away from proprietary or hazardous reagents towards commodity chemicals, companies can significantly de-risk their supply chains and reduce dependency on single-source suppliers. The elimination of heavy metal catalysts not only lowers raw material costs but also reduces the environmental burden associated with waste disposal and metal recovery, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts and hazardous diazo reagents with inexpensive elemental iodine and stable imidoyl chlorides results in a drastic reduction in raw material expenditure. Furthermore, the simplified workup procedure, which often requires only filtration and standard chromatography rather than complex metal scavenging, leads to substantial savings in processing time and consumables. This streamlined process directly translates to a lower cost of goods sold (COGS) for the final intermediate.
- Enhanced Supply Chain Reliability: The starting materials, including aromatic amines, aldehydes, and hydrazine hydrate, are widely available commodities with established global supply chains. This abundance ensures consistent availability and protects against market volatility that often affects specialized fluorinating agents. The robustness of the reaction, which tolerates ambient moisture and oxygen to a degree, further enhances reliability by reducing the need for specialized inert atmosphere equipment that can be prone to failure.
- Scalability and Environmental Compliance: The method has been demonstrated to be easily scalable, with the patent noting successful expansion to gram-level synthesis without loss of efficiency. The absence of toxic heavy metals simplifies wastewater treatment and solid waste disposal, facilitating compliance with increasingly stringent environmental regulations. This eco-friendly profile makes the process sustainable for long-term commercial production, reducing the risk of regulatory shutdowns or fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iodine-promoted triazole synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities and limitations for potential adopters.
Q: Does this synthesis require expensive heavy metal catalysts?
A: No, the patented method utilizes elemental iodine as a promoter, completely avoiding the need for toxic or expensive transition metal catalysts, which simplifies purification and reduces environmental impact.
Q: What are the advantages of using trifluoroethylimidoyl chloride over other synthons?
A: Trifluoroethylimidoyl chloride is more stable and easier to handle compared to alternatives like trifluorodiazoethane, offering a safer and more operationally simple route for industrial scale-up.
Q: Is the reaction sensitive to moisture or oxygen?
A: The process is robust and does not require strict anhydrous or anaerobic conditions, making it highly suitable for standard manufacturing environments without specialized inert atmosphere equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into industrial reality. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 5-trifluoromethyl-1,2,4-triazole intermediate meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you optimize your supply chain and achieve your production goals efficiently.
