Revolutionizing Monofluoroolefin Production with Cost-Effective Copper Catalysis for Commercial Scale-Up
The landscape of organic synthesis for fluorinated compounds is undergoing a significant transformation driven by the urgent need for cost-effective and highly selective methodologies in the pharmaceutical and agrochemical sectors. Patent CN114957170A, published on August 30, 2022, introduces a groundbreaking direct monofluoroolefination reaction targeting the ortho-position carbon-hydrogen bonds of ether compounds. This innovation addresses a critical bottleneck in the production of monofluoroolefins, which serve as vital structural motifs in modern drug design due to their ability to act as amide bond isosteres, thereby enhancing metabolic stability and conformational rigidity. The introduction of fluorine atoms into organic molecules is known to profoundly influence lipophilicity and water solubility, with statistics indicating that approximately 25% of pharmaceutical molecules and 40% of pesticide molecules currently contain at least one fluorine atom. 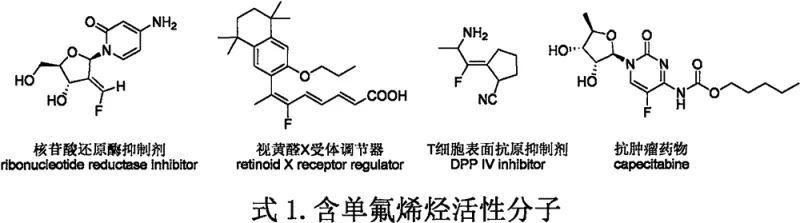 As illustrated in the structural diversity of bioactive molecules shown above, the monofluoroolefin fragment is a recurring feature in high-value active pharmaceutical ingredients, underscoring the immense commercial potential of mastering its synthesis. This patent specifically discloses a method that bypasses the limitations of traditional sp2 carbon-hydrogen bond functionalization, focusing instead on the more challenging yet valuable sp3 carbon-hydrogen bond activation adjacent to ether oxygens. By utilizing fluoroacrylic acid and ether compounds as primary feedstocks, this technology offers a streamlined pathway to Z-configured monofluoroolefins, positioning itself as a game-changer for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
As illustrated in the structural diversity of bioactive molecules shown above, the monofluoroolefin fragment is a recurring feature in high-value active pharmaceutical ingredients, underscoring the immense commercial potential of mastering its synthesis. This patent specifically discloses a method that bypasses the limitations of traditional sp2 carbon-hydrogen bond functionalization, focusing instead on the more challenging yet valuable sp3 carbon-hydrogen bond activation adjacent to ether oxygens. By utilizing fluoroacrylic acid and ether compounds as primary feedstocks, this technology offers a streamlined pathway to Z-configured monofluoroolefins, positioning itself as a game-changer for reliable pharmaceutical intermediate suppliers seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct monofluoroalkenylation of carbon-hydrogen bonds has been dominated by methods targeting sp2 centers, leaving the functionalization of sp3 centers largely underdeveloped and reliant on prohibitively expensive catalytic systems. Prominent prior art, such as the work by Hashmi et al. in 2016, relied heavily on photocatalytic systems employing pre-prepared iridium complexes, specifically bis[2-(2,4-difluorophenyl)-5-trifluoromethylpyridine][2-2'-bipyridine]iridium bis(hexafluorophosphate) salts. 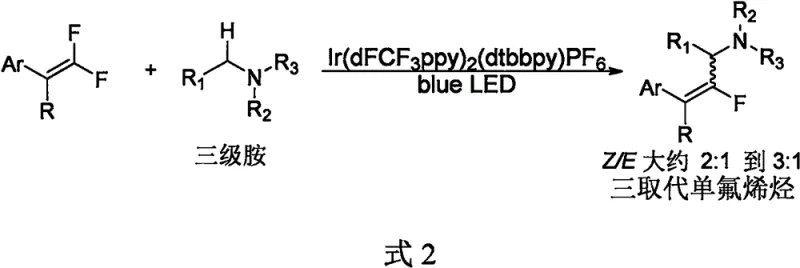 As depicted in the reaction scheme above, these legacy methods suffer from two major commercial drawbacks: the exorbitant cost of the iridium catalyst, cited at approximately 2000 yuan per gram, and mediocre stereoselectivity, yielding Z/E ratios of merely 2:1 to 3:1 for trisubstituted monofluoroolefins. Subsequent developments by Wang et al. and Deng et al. attempted to expand the substrate scope to include ethers and other C-H bonds but remained tethered to the same expensive iridium photocatalysts and failed to improve the Z/E selectivity beyond a range of 1:1 to 3:1. This lack of stereocontrol creates significant downstream processing challenges, necessitating complex and yield-eroding purification steps to isolate the desired Z-isomer, which is often the biologically active configuration. For procurement managers and supply chain heads, these factors translate into volatile raw material costs, extended lead times due to difficult separations, and a fragile supply chain dependent on scarce precious metals.
As depicted in the reaction scheme above, these legacy methods suffer from two major commercial drawbacks: the exorbitant cost of the iridium catalyst, cited at approximately 2000 yuan per gram, and mediocre stereoselectivity, yielding Z/E ratios of merely 2:1 to 3:1 for trisubstituted monofluoroolefins. Subsequent developments by Wang et al. and Deng et al. attempted to expand the substrate scope to include ethers and other C-H bonds but remained tethered to the same expensive iridium photocatalysts and failed to improve the Z/E selectivity beyond a range of 1:1 to 3:1. This lack of stereocontrol creates significant downstream processing challenges, necessitating complex and yield-eroding purification steps to isolate the desired Z-isomer, which is often the biologically active configuration. For procurement managers and supply chain heads, these factors translate into volatile raw material costs, extended lead times due to difficult separations, and a fragile supply chain dependent on scarce precious metals.
The Novel Approach
In stark contrast to the precious metal-dependent status quo, the methodology disclosed in CN114957170A leverages inexpensive cuprous bromide (CuBr) as the primary catalyst, which is available at a fraction of the cost, approximately 0.25 yuan per gram. 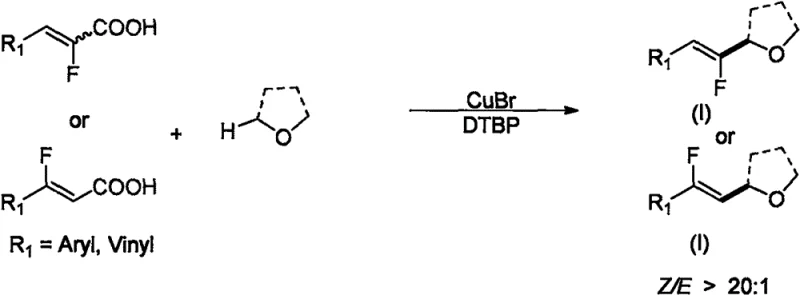 The general reaction scheme presented above highlights the elegance of this new approach, where fluoroacrylic acids react with ether compounds in the presence of di-tert-butyl peroxide (DTBP) as a radical initiator to exclusively form Z-configured monofluoroolefins. This shift from photocatalysis to a thermal radical process not only eliminates the need for specialized LED equipment but also dramatically simplifies the reactor setup, allowing for standard heating mantles or oil baths to be used effectively. The most striking improvement lies in the stereoselectivity; where previous methods struggled to exceed a 3:1 ratio, this copper-catalyzed system consistently achieves Z/E selectivity greater than 20:1, with many specific examples demonstrating ratios exceeding 30:1. This exceptional selectivity minimizes the formation of unwanted E-isomers, drastically reducing the burden on purification teams and increasing the overall throughput of the manufacturing process. For a reliable pharmaceutical intermediate supplier, this represents a paradigm shift towards high-efficiency, low-cost manufacturing that does not compromise on the stringent purity specifications required by global regulatory bodies.
The general reaction scheme presented above highlights the elegance of this new approach, where fluoroacrylic acids react with ether compounds in the presence of di-tert-butyl peroxide (DTBP) as a radical initiator to exclusively form Z-configured monofluoroolefins. This shift from photocatalysis to a thermal radical process not only eliminates the need for specialized LED equipment but also dramatically simplifies the reactor setup, allowing for standard heating mantles or oil baths to be used effectively. The most striking improvement lies in the stereoselectivity; where previous methods struggled to exceed a 3:1 ratio, this copper-catalyzed system consistently achieves Z/E selectivity greater than 20:1, with many specific examples demonstrating ratios exceeding 30:1. This exceptional selectivity minimizes the formation of unwanted E-isomers, drastically reducing the burden on purification teams and increasing the overall throughput of the manufacturing process. For a reliable pharmaceutical intermediate supplier, this represents a paradigm shift towards high-efficiency, low-cost manufacturing that does not compromise on the stringent purity specifications required by global regulatory bodies.
Mechanistic Insights into CuBr-Catalyzed Radical Monofluoroolefination
The mechanistic underpinning of this transformation involves a radical-mediated pathway initiated by the thermal decomposition of di-tert-butyl peroxide (DTBP) at elevated temperatures, typically around 110°C. Upon heating, DTBP undergoes homolytic cleavage to generate tert-butoxy radicals, which are potent hydrogen atom abstractors capable of activating the relatively inert alpha-C-H bonds adjacent to the oxygen atom in ether substrates. The resulting alpha-oxy radical is then intercepted by the copper catalyst species, facilitating the subsequent addition to the electron-deficient double bond of the fluoroacrylic acid derivative. This radical addition step is crucial for establishing the carbon-carbon bond and setting the stereochemistry of the final olefin. The high Z-selectivity observed is hypothesized to arise from the specific transition state geometry favored by the copper coordination and the steric interactions between the bulky ether substituent and the carboxylic acid group during the elimination or decarboxylation sequence that follows. Unlike the photoredox cycles which often involve long-lived excited states prone to non-selective pathways, this thermal radical process is kinetically controlled, favoring the formation of the thermodynamically more stable or kinetically accessible Z-isomer through a tightly organized transition state.
From an impurity control perspective, the use of CuBr offers distinct advantages over iridium complexes regarding the removal of residual metals from the final API intermediate. Copper is a ubiquitous element in biological systems and generally faces less stringent limits in pharmaceutical products compared to heavy precious metals like iridium, simplifying the metal scavenging process. Furthermore, the reaction demonstrates remarkable functional group tolerance, accommodating various substituents on the aromatic ring of the fluoroacrylic acid, including electron-withdrawing groups like fluorine, chlorine, and bromine, as well as electron-donating groups. 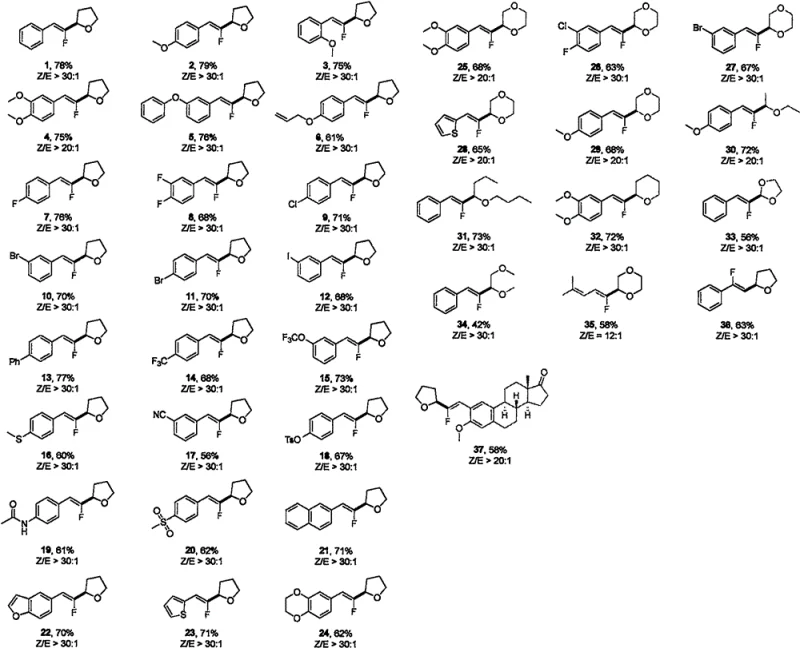 The extensive substrate scope displayed in the grid above confirms that the reaction is robust across a wide array of structurally diverse ethers and fluoroacrylic acids, maintaining high yields and selectivity even with sterically hindered or electronically deactivated substrates. This breadth of compatibility ensures that the process can be adapted for the synthesis of complex drug candidates without requiring extensive re-optimization for each new analog, providing R&D directors with a versatile tool for rapid library generation and process development.
The extensive substrate scope displayed in the grid above confirms that the reaction is robust across a wide array of structurally diverse ethers and fluoroacrylic acids, maintaining high yields and selectivity even with sterically hindered or electronically deactivated substrates. This breadth of compatibility ensures that the process can be adapted for the synthesis of complex drug candidates without requiring extensive re-optimization for each new analog, providing R&D directors with a versatile tool for rapid library generation and process development.
How to Synthesize Z-Monofluoroolefin Efficiently
The practical implementation of this synthesis route is designed for operational simplicity, requiring standard laboratory glassware and common reagents that are readily available from bulk chemical suppliers. The protocol avoids the need for rigorous exclusion of moisture or oxygen beyond standard inert atmosphere techniques, and the reaction proceeds efficiently in common solvents such as tetrahydrofuran or 1,4-dioxane. Detailed standardized synthesis steps for the preparation of these high-purity intermediates are provided in the guide below, outlining the precise stoichiometry and workup procedures necessary to achieve the reported yields and selectivity.
- Combine cuprous bromide (10 mol%) and alpha-fluorocinnamic acid (0.2 mmol) in a sealed reaction tube under air, then flush with argon three times.
- Add tetrahydrofuran (1.5 mL) and di-tert-butyl peroxide (3 equiv) under argon protection, seal the vessel, and stir in an oil bath at 110°C for 18 hours.
- Filter the reaction mixture through a short silica gel column with ethyl acetate, concentrate the organic phase, and purify the crude product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this copper-catalyzed monofluoroolefination technology offers profound strategic advantages for procurement managers and supply chain leaders focused on cost reduction in pharmaceutical intermediate manufacturing and supply continuity. By transitioning away from iridium-based photocatalysis, companies can eliminate the volatility associated with precious metal markets and the supply chain risks linked to sourcing specialized, high-cost catalysts that may have long lead times. The substitution of a catalyst costing approximately 2000 yuan/g with one costing roughly 0.25 yuan/g represents a reduction in catalyst material cost by several orders of magnitude, directly impacting the gross margin of the final product. This drastic cost saving is compounded by the simplified reaction infrastructure, as the process does not require expensive blue LED arrays or specialized photoreactors, allowing production to occur in standard stainless steel or glass-lined reactors found in most multipurpose chemical plants.
- Cost Reduction in Manufacturing: The economic impact of replacing expensive iridium catalysts with commodity-grade cuprous bromide cannot be overstated, as it fundamentally alters the cost structure of the synthesis. Beyond the raw material savings, the high Z/E selectivity (>20:1) significantly reduces the solvent and silica consumption associated with chromatographic purification, as there is far less E-isomer byproduct to separate. This efficiency translates to lower waste disposal costs and higher overall process mass intensity (PMI), aligning with green chemistry principles while simultaneously driving down the cost of goods sold (COGS). Furthermore, the use of DTBP as a cheap and effective initiator avoids the need for complex ligand synthesis or handling of air-sensitive organometallic reagents, streamlining the operational expenditure.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved because the key reagents—fluoroacrylic acids, simple ethers, CuBr, and DTBP—are all commodity chemicals produced at a global scale with stable supply chains. Unlike custom-synthesized iridium complexes which may rely on single-source suppliers, these bulk chemicals can be procured from multiple vendors, mitigating the risk of supply disruption. The robustness of the reaction conditions, which tolerate a wide range of functional groups and do not require cryogenic temperatures or ultra-high vacuum, ensures that the process can be transferred between different manufacturing sites with minimal tech transfer friction. This flexibility allows supply chain heads to diversify their manufacturing base, ensuring continuous availability of critical intermediates even in the face of regional logistical challenges.
- Scalability and Environmental Compliance: The thermal nature of this reaction makes it inherently easier to scale from gram-scale laboratory optimization to multi-ton commercial production compared to photochemical processes, which often suffer from light penetration issues in large vessels. The absence of precious heavy metals simplifies the environmental compliance profile, as copper residues are easier to manage and treat in wastewater streams compared to persistent organoiridium species. The high atom economy and selectivity reduce the generation of hazardous chemical waste, supporting corporate sustainability goals and reducing the regulatory burden associated with waste disposal. This scalability ensures that the technology can meet the growing demand for fluorinated intermediates in the pharmaceutical and agrochemical industries without requiring massive capital investment in new specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel monofluoroolefination technology, derived directly from the experimental data and comparative analysis provided in the patent documentation. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this process for integration into their existing manufacturing workflows. Understanding these details is crucial for making informed decisions about process adoption and supplier qualification.
Q: What is the primary economic advantage of this new monofluoroolefination method?
A: The method replaces expensive iridium photocatalysts, which can cost approximately 2000 yuan/g, with inexpensive cuprous bromide costing around 0.25 yuan/g, resulting in drastic raw material cost reductions.
Q: How does the Z/E selectivity compare to previous iridium-catalyzed methods?
A: While prior art methods using iridium catalysts typically achieve Z/E ratios of only 1:1 to 3:1, this copper-catalyzed protocol consistently delivers exceptional Z-selectivity with ratios exceeding 20:1, often reaching >30:1.
Q: Is this reaction suitable for large-scale pharmaceutical intermediate production?
A: Yes, the reaction utilizes readily available raw materials, operates under relatively simple thermal conditions without specialized light equipment, and demonstrates broad functional group tolerance, making it highly scalable for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monofluoroolefin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed technology in delivering high-value fluorinated building blocks to the global market. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped to handle the thermal radical reactions described in this patent, supported by rigorous QC labs that guarantee stringent purity specifications for every batch of monofluoroolefin intermediates we produce. We are committed to leveraging this cost-effective methodology to provide our clients with a competitive edge in the development of next-generation therapeutics and agrochemicals.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this copper-catalyzed process for your specific target molecules. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in advancing complex fluorine chemistry from bench to benchtop to plant.
