Advanced Rhodium-Catalyzed Synthesis of Chiral 3,3-Disubstituted Isoindolinones for Commercial Scale-Up
Introduction to Next-Generation Isoindolinone Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex chiral scaffolds, particularly those containing quaternary carbon centers which are prevalent in bioactive molecules. Patent CN113735756A introduces a groundbreaking methodology for the synthesis of chiral 3,3-disubstituted isoindolinone compounds, utilizing a highly sophisticated rhodium-catalyzed C-H activation strategy. This technology represents a significant leap forward in synthetic organic chemistry, enabling the direct construction of these valuable heterocyclic skeletons from readily available N-methoxybenzamides and 1,3-enynes. By leveraging a chiral cyclopentadienyl rhodium catalyst, the process achieves remarkable levels of stereocontrol and chemical efficiency under exceptionally mild reaction conditions. For R&D directors and process chemists, this patent offers a robust solution to the longstanding challenges associated with building congested chiral centers, providing a reliable foundation for the development of new drug candidates and advanced material intermediates.
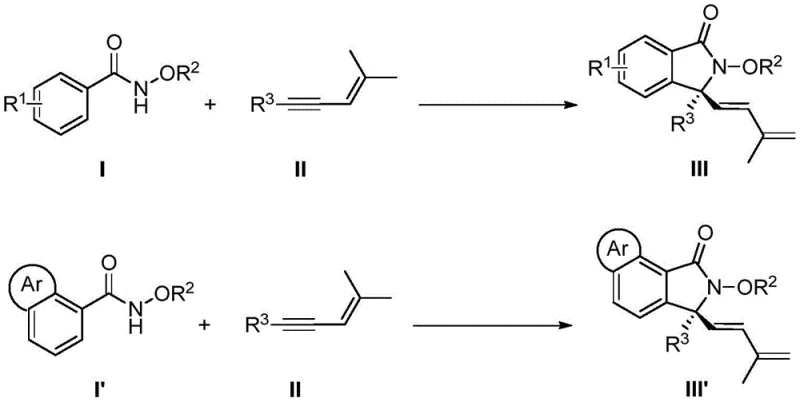
The core innovation lies in the seamless integration of C-H bond activation, enyne migration insertion, 1,4-rhodium migration, and nucleophilic cyclization into a single operational sequence. This tandem reaction pathway not only streamlines the synthetic route but also drastically improves the overall atom economy compared to classical approaches. The ability to operate at temperatures as low as 5°C to 15°C further underscores the practicality of this method for large-scale applications, where thermal stability and energy consumption are critical factors. As a leading entity in the chemical sector, understanding the nuances of such transformative technologies is essential for maintaining a competitive edge in the supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of 3,3-disubstituted chiral isoindolinone skeletons has been a formidable challenge for organic synthesizers, often relying on strategies that suffer from inherent inefficiencies. Conventional routes typically involve the enantioselective functionalization of pre-existing isoindolinone cores, a approach that necessitates the prior synthesis of the heterocyclic ring system itself. This multi-step requirement inevitably leads to poor atom and step economy, resulting in increased waste generation and higher production costs. Furthermore, the substrates required for these traditional methods are often difficult to prepare and possess limited stability, restricting the diversity of derivatives that can be accessed. The harsh reaction conditions frequently employed in older methodologies can also lead to decomposition of sensitive functional groups, thereby limiting the scope of applicable starting materials and complicating the purification process. These cumulative drawbacks create significant bottlenecks in the supply chain for complex chiral intermediates, driving up lead times and reducing the overall feasibility of commercial manufacturing.
The Novel Approach
In stark contrast, the novel rhodium-catalyzed [4+1] cyclization described in the patent data offers a paradigm shift by utilizing simple, stable, and commercially accessible starting materials. The direct use of N-methoxybenzamides and 1,3-enynes eliminates the need for pre-activated substrates, allowing for a convergent synthesis that builds complexity rapidly from simple precursors. This approach capitalizes on the unique reactivity of alkynes acting as one-carbon synthons, a transformation that is rare in literature but highly effective in this specific catalytic system. The reaction proceeds through a well-defined cascade involving C-H activation and migratory insertion, facilitated by the chiral rhodium catalyst, to construct the quaternary carbon center with high precision. The mild conditions, utilizing alcohol solvents and moderate temperatures, ensure compatibility with a wide array of functional groups, including halogens, esters, and ethers, which might be intolerant of harsher traditional reagents. This versatility makes the new method an ideal candidate for the cost reduction in pharmaceutical intermediate manufacturing, as it simplifies process development and enhances the robustness of the production line.
Mechanistic Insights into Rhodium-Catalyzed C-H Activation and Cyclization
The success of this synthetic transformation is fundamentally rooted in the precise design of the chiral cyclopentadienyl rhodium catalyst, which orchestrates the stereochemical outcome of the reaction. The catalytic cycle initiates with the coordination of the rhodium species to the directing group of the N-methoxybenzamide, facilitating the cleavage of the ortho C-H bond to form a rhodacycle intermediate. This step is crucial as it sets the stage for the subsequent insertion of the 1,3-enyne substrate. The chiral environment provided by the pentamethylcyclopentadienyl ligand, often substituted with alkoxy groups such as isopropoxy or methoxy, exerts steric control over the approach of the enyne, ensuring that the migratory insertion occurs with high facial selectivity. Following the insertion, a 1,4-rhodium migration takes place, shifting the metal center to a position that enables the final nucleophilic attack by the amide oxygen or nitrogen onto the activated carbonyl or imine species, depending on the specific mechanistic pathway proposed. This intricate dance of bond breaking and forming is managed with exceptional fidelity, resulting in the formation of the isoindolinone ring with the simultaneous establishment of the chiral quaternary center.
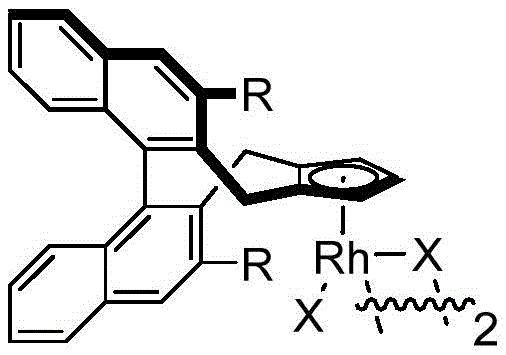
Impurity control in this system is inherently managed by the high selectivity of the catalyst and the mild reaction conditions. The use of silver difluoride as an oxidant ensures the regeneration of the active Rh(III) species without introducing aggressive oxidative side reactions that could degrade the product or starting materials. Furthermore, the specific choice of carboxylic acid additives, such as acetic acid or pivalic acid, plays a vital role in protonating intermediates and facilitating the turnover of the catalytic cycle, thereby minimizing the accumulation of off-cycle species that could lead to byproduct formation. The broad substrate scope demonstrated in the patent examples, ranging from electron-rich to electron-deficient benzamides and various enyne structures, indicates that the catalytic system is robust against electronic variations. This resilience suggests that the transition states are well-stabilized by the ligand framework, preventing non-selective background reactions. For quality assurance teams, this mechanistic robustness translates to a cleaner reaction profile, simplifying downstream purification and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications with minimal effort.
How to Synthesize Chiral 3,3-Disubstituted Isoindolinones Efficiently
Implementing this advanced synthetic route requires careful attention to the stoichiometry and reaction parameters outlined in the patent to maximize yield and enantiomeric excess. The process begins with the preparation of the reaction mixture in an inert atmosphere, combining the N-methoxybenzamide derivative and the 1,3-enyne coupling partner in a suitable alcohol solvent such as 3-pentanol or ethanol. The addition of the chiral cyclopentadienyl rhodium catalyst, typically at a loading of 3-5 mol%, along with the silver difluoride oxidant and a carboxylic acid additive, initiates the catalytic cycle. Maintaining the temperature within the narrow window of 5°C to 15°C is critical for preserving the stereochemical integrity of the product, as higher temperatures may erode enantioselectivity. After the reaction period of 60 to 80 hours, the mixture is quenched and subjected to standard workup procedures. The detailed standardized synthesis steps for this process are provided in the guide below.
- Combine N-methoxybenzamide substrate, 1,3-enyne coupling partner, chiral cyclopentadienyl rhodium catalyst, and silver difluoride oxidant in an alcohol solvent.
- Add a carboxylic acid additive such as acetic acid and maintain the reaction mixture at mild temperatures between 5°C and 15°C for 60 to 80 hours.
- Quench the reaction with ethylenediamine, concentrate the mixture, and purify the crude product via silica gel column chromatography to isolate the target chiral isoindolinone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this rhodium-catalyzed technology offers substantial strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials; since the method utilizes stable and easily prepared N-methoxybenzamides and 1,3-enynes, sourcing becomes more reliable and less prone to the volatility associated with exotic or unstable reagents. This stability ensures a consistent flow of materials, reducing the risk of production stoppages due to raw material shortages. Furthermore, the high atom economy and step efficiency of the [4+1] cyclization mean that fewer unit operations are required to reach the final target, which directly correlates to reduced capital expenditure on equipment and lower operational overheads. The ability to run the reaction at near-ambient temperatures also contributes to significant energy savings, aligning with modern sustainability goals and reducing the carbon footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The elimination of multi-step sequences and the use of catalytic amounts of rhodium rather than stoichiometric chiral auxiliaries leads to a profound reduction in material costs. By avoiding the need for pre-functionalized substrates and harsh reagents, the process minimizes waste disposal costs and reduces the consumption of solvents and energy. Although rhodium is a precious metal, its low loading and the potential for recovery or the use of cost-effective ligand systems ensure that the overall cost per kilogram of the final product is optimized. This efficiency allows for competitive pricing structures when supplying high-purity pharmaceutical intermediates to global markets, making the technology economically viable for large-scale production.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against a wide variety of functional groups means that a single platform technology can be adapted to produce a diverse library of isoindolinone derivatives. This flexibility is invaluable for supply chain managers who need to respond quickly to changing market demands or custom synthesis requests. The use of common solvents like alcohols and standard oxidants further simplifies logistics, as these materials are widely available and easy to handle. Consequently, lead times for high-purity pharmaceutical intermediates can be significantly reduced, as the process is less susceptible to delays caused by specialized reagent procurement or complex purification bottlenecks.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of hazardous byproducts make this process highly scalable from gram to ton quantities without significant re-engineering. The simplified workup procedure, often involving straightforward column chromatography or crystallization, reduces the generation of chemical waste, facilitating compliance with increasingly stringent environmental regulations. The high selectivity of the reaction ensures that the final product requires less intensive purification, further lowering the environmental impact. This alignment with green chemistry principles not only mitigates regulatory risks but also enhances the corporate social responsibility profile of the manufacturing operation, appealing to eco-conscious partners and stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this rhodium-catalyzed synthesis. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this method into their existing production workflows.
Q: What are the primary advantages of this Rh-catalyzed method over traditional isoindolinone synthesis?
A: This method utilizes direct C-H bond activation and a [4+1] cyclization strategy, eliminating the need for pre-functionalized substrates and multi-step sequences. It operates under mild conditions with high atom economy and achieves excellent enantioselectivity (up to 96% ee), significantly simplifying the production of complex chiral scaffolds.
Q: What is the scope of substrates compatible with this catalytic system?
A: The process demonstrates broad substrate tolerance, accommodating N-methoxybenzamides with various electronic properties including electron-donating groups like methyl and methoxy, as well as electron-withdrawing groups such as halogens, nitro, and trifluoromethyl. Additionally, diverse 1,3-enyne partners with alkyl, cycloalkyl, and aryl substituents are effectively converted.
Q: How does the choice of catalyst influence the stereochemical outcome?
A: The use of specifically designed chiral cyclopentadienyl rhodium catalysts is critical for inducing asymmetry. The steric environment provided by ligands such as isopropoxy or methoxy-substituted Cp rings directs the enantioselective migratory insertion and nucleophilic cyclization, ensuring the formation of the desired 3,3-disubstituted quaternary carbon center with high optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3,3-Disubstituted Isoindolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN113735756A for the future of pharmaceutical manufacturing. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We are committed to delivering chiral 3,3-disubstituted isoindolinones and related intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify identity and enantiomeric excess. Our infrastructure is designed to handle complex synthetic challenges, providing a secure and reliable source for your critical drug development projects.
We invite you to collaborate with us to leverage this cutting-edge synthesis for your specific application needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. We encourage you to contact us today to obtain specific COA data for our available inventory and to discuss route feasibility assessments for your target molecules, ensuring a seamless supply of high-quality intermediates for your pipeline.
