Advanced Decarboxylative Halogenation for Scalable Production of High-Purity Aryl Halides
The landscape of aromatic halogenation is undergoing a significant transformation driven by the urgent need for greener, more atom-economical processes in the fine chemical industry. Patent CN107325002B introduces a groundbreaking methodology for synthesizing aryl halides by utilizing aryl carboxylic acids as the primary starting material. This innovative approach leverages a synergistic catalytic system comprising a silver catalyst, a copper additive, and a bidentate nitrogen ligand under an oxygen atmosphere. By replacing hazardous elemental halogens or unstable diazonium salts with inexpensive and stable alkali metal halide salts (MX), this technology addresses critical pain points in the supply chain of pharmaceutical and agrochemical intermediates. The process not only enhances safety profiles by eliminating volatile halogen gases but also significantly broadens the substrate scope to include electron-deficient aromatics that are traditionally difficult to functionalize via electrophilic pathways.
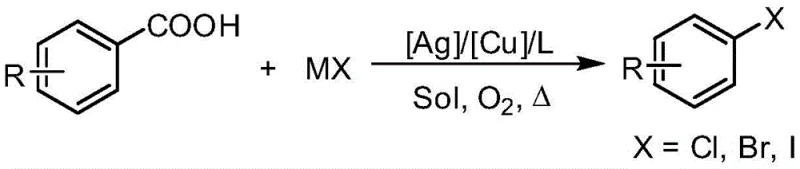
For R&D directors and process chemists, the ability to access diverse aryl halides from stable carboxylic acid precursors represents a paradigm shift in retrosynthetic planning. The method described in the patent operates effectively across a wide temperature range, typically between 100°C and 200°C, and demonstrates remarkable tolerance towards sensitive functional groups such as nitro, fluoro, chloro, and methoxy substituents. This robustness ensures that complex molecular architectures can be constructed without the need for extensive protecting group strategies, thereby streamlining the overall synthetic route. Furthermore, the use of molecular oxygen as the oxidant aligns perfectly with modern sustainability goals, minimizing the generation of toxic waste streams associated with stoichiometric oxidants often found in legacy protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of halogen atoms onto aromatic rings has relied heavily on classical electrophilic aromatic substitution or diazonium salt chemistry, both of which suffer from inherent limitations that hinder industrial scalability and efficiency. The traditional electrophilic substitution method, typically employing Lewis acids like iron(III) halides and elemental halogens, strictly requires the presence of electron-donating groups on the aromatic ring to activate the substrate for attack. 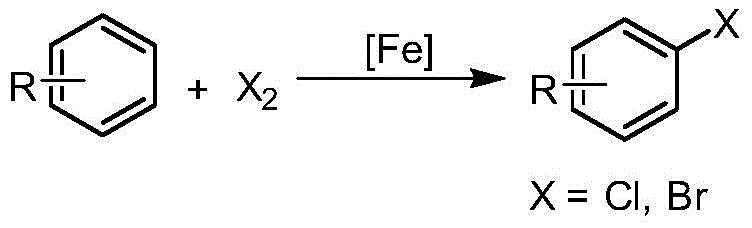 This dependency severely restricts the scope of applicable substrates, rendering it ineffective for electron-deficient systems which are prevalent in many bioactive molecules. Moreover, reactions involving elemental fluorine are notoriously violent and uncontrollable, posing severe safety risks, while iodination via electrophilic attack is often thermodynamically unfavorable, leading to poor yields and reversibility issues.
This dependency severely restricts the scope of applicable substrates, rendering it ineffective for electron-deficient systems which are prevalent in many bioactive molecules. Moreover, reactions involving elemental fluorine are notoriously violent and uncontrollable, posing severe safety risks, while iodination via electrophilic attack is often thermodynamically unfavorable, leading to poor yields and reversibility issues.
Alternatively, the Sandmeyer reaction utilizing aryl diazonium salts offers a pathway to aryl halides but introduces significant operational hazards and stability concerns. 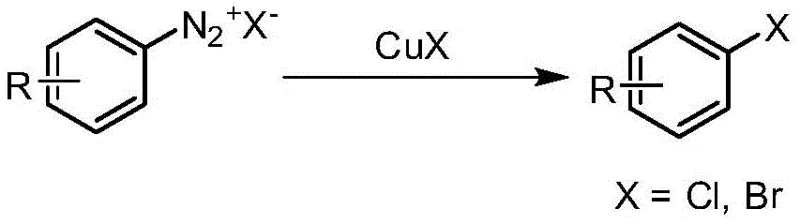 Diazonium salts are thermally unstable and prone to explosive decomposition, necessitating strict temperature control and often requiring in-situ generation which complicates process engineering. Additionally, this method struggles with the synthesis of aryl iodides when using cuprous iodide due to competing side reactions and low conversion rates. Another historical approach involves the decarboxylation of aryl carboxylic acids using stoichiometric amounts of cuprous halides.
Diazonium salts are thermally unstable and prone to explosive decomposition, necessitating strict temperature control and often requiring in-situ generation which complicates process engineering. Additionally, this method struggles with the synthesis of aryl iodides when using cuprous iodide due to competing side reactions and low conversion rates. Another historical approach involves the decarboxylation of aryl carboxylic acids using stoichiometric amounts of cuprous halides. 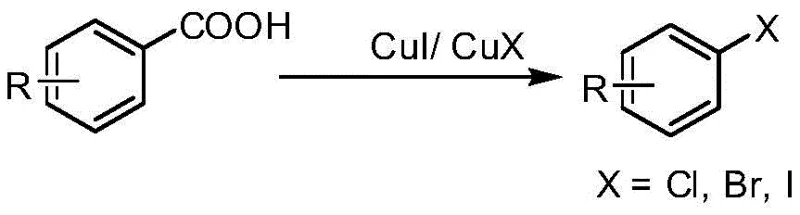 While this avoids diazonium instability, it consumes excessive amounts of expensive copper salts, generating substantial heavy metal waste and driving up raw material costs, which is economically unsustainable for large-scale manufacturing.
While this avoids diazonium instability, it consumes excessive amounts of expensive copper salts, generating substantial heavy metal waste and driving up raw material costs, which is economically unsustainable for large-scale manufacturing.
The Novel Approach
The methodology disclosed in CN107325002B elegantly circumvents these drawbacks by employing a catalytic amount of silver coupled with a copper promoter and a bidentate nitrogen ligand. This system activates the carboxylic acid moiety for decarboxylation under mild oxidative conditions, allowing for the direct replacement of the carboxyl group with a halogen atom. Unlike the stoichiometric copper methods, this catalytic variant drastically reduces metal consumption, lowering both the cost of goods sold and the environmental burden of metal waste disposal. The reaction proceeds smoothly with simple alkali metal halides like NaI, NaBr, or NaCl, which are orders of magnitude cheaper and safer to handle than elemental halogens or specialized organometallic reagents. This shift from stoichiometric to catalytic processes, combined with the use of benign oxidants like oxygen, establishes a new standard for sustainable aryl halide production that is both economically viable and environmentally responsible.
Mechanistic Insights into Ag/Cu-Catalyzed Decarboxylative Halogenation
The core of this technological advancement lies in the intricate interplay between the silver catalyst, the copper additive, and the bidentate nitrogen ligand within an oxidative environment. The proposed mechanism involves the initial coordination of the silver species to the carboxylate anion, facilitating the formation of a silver-carboxylate intermediate. Under the influence of heat and oxygen, this intermediate undergoes decarboxylation to generate an aryl-silver species. The copper additive plays a crucial role as a transmetallation partner or a redox mediator, assisting in the transfer of the aryl group to the halogen source. The bidentate nitrogen ligand, such as 1,10-phenanthroline or 2,9-dimethyl-1,10-phenanthroline, stabilizes the metal centers and modulates their electronic properties, ensuring high catalytic turnover and preventing the aggregation of metal particles which would lead to catalyst deactivation.
Crucially, the presence of molecular oxygen is non-negotiable for the success of this transformation, as evidenced by comparative experiments where inert atmospheres resulted in complete reaction failure. Oxygen likely serves to re-oxidize the reduced metal species back to their active oxidation states, closing the catalytic cycle and sustaining the reaction momentum. This oxidative regeneration mechanism allows for the use of catalytic rather than stoichiometric quantities of metals. From an impurity control perspective, the high selectivity of this system minimizes the formation of homocoupling byproducts (biaryls) which are common in radical-based decarboxylation processes. The specific choice of ligand and solvent, typically polar aprotic solvents like DMSO, further suppresses side reactions, ensuring that the halogenation occurs exclusively at the ipso-position of the carboxyl group, delivering high-purity products suitable for downstream pharmaceutical applications without extensive purification.
How to Synthesize Aryl Halides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the ratio of catalytic components and the maintenance of an oxygen-rich environment. The general protocol involves charging a reaction vessel with the aryl carboxylic acid substrate, the appropriate alkali metal halide salt, the silver catalyst (e.g., silver sulfate or silver iodide), the copper promoter (e.g., copper acetate), and the nitrogen ligand. The mixture is then suspended in a suitable organic solvent and heated to temperatures ranging from 150°C to 170°C under an oxygen balloon or flow. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by combining the aryl carboxylic acid substrate, alkali metal halide salt (MX), silver catalyst, copper additive, and bidentate nitrogen ligand in an organic solvent such as DMSO.
- Heat the reaction mixture to a temperature between 100°C and 200°C, preferably 150°C to 170°C, while maintaining an oxygen atmosphere to facilitate the oxidative decarboxylation process.
- Upon completion, quench the reaction with water, extract the organic phase with ethyl acetate, wash with base, and purify to isolate the target aryl halide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this decarboxylative halogenation technology offers profound strategic benefits that extend beyond mere technical feasibility. The primary driver for cost optimization is the substitution of expensive and hazardous halogenating agents with commodity halide salts. Sodium iodide, sodium bromide, and sodium chloride are globally available in bulk quantities at negligible costs compared to elemental iodine or bromine, which require specialized storage and handling infrastructure. This raw material switch directly translates to a significant reduction in the variable cost of production, enhancing the margin profile for high-volume aryl halide intermediates. Furthermore, the elimination of stoichiometric copper waste reduces the financial liability associated with hazardous waste treatment and disposal, contributing to a leaner and more compliant operational model.
Supply chain reliability is markedly improved due to the stability and shelf-life of the starting materials. Aryl carboxylic acids are generally stable solids that are easy to transport and store, unlike diazonium salts which must be generated immediately prior to use, creating bottlenecks in continuous manufacturing workflows. The robustness of the catalytic system against various functional groups means that a single standardized protocol can be applied to a wide library of substrates, simplifying inventory management and reducing the need for multiple specialized reagent lines. This flexibility allows manufacturers to respond rapidly to changing market demands for different API intermediates without retooling entire production suites. Additionally, the use of oxygen as the oxidant removes the dependency on specialized chemical oxidants that may have volatile pricing or supply constraints, further securing the supply chain against external market shocks.
Scalability and environmental compliance are inherently built into this process design. The reaction conditions, while requiring elevated temperatures, operate at atmospheric pressure, removing the need for expensive high-pressure autoclaves often required for carbonylation or hydrogenation reactions. The absence of toxic halogen gas emissions aligns with increasingly stringent environmental regulations, reducing the risk of regulatory fines and community opposition. The catalytic nature of the silver and copper system ensures that metal residues in the final product are minimized, easing the burden on downstream purification units and ensuring that the final API intermediates meet rigorous heavy metal specifications required by global health authorities. This combination of safety, cost-efficiency, and regulatory alignment makes the technology an ideal candidate for long-term commercial scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method. These answers are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for process development teams evaluating this technology for potential licensing or adoption.
Q: What are the primary advantages of this decarboxylative halogenation method over traditional electrophilic substitution?
A: Unlike traditional electrophilic substitution which requires specific electron-donating groups and suffers from poor regioselectivity, this silver-catalyzed method utilizes readily available aryl carboxylic acids and tolerates a wide range of functional groups including electron-withdrawing nitro and halo groups, offering superior versatility for complex molecule synthesis.
Q: Why is oxygen required in this catalytic system?
A: Oxygen acts as the terminal oxidant in this catalytic cycle. Experimental data indicates that inert gas shielding or vacuum conditions completely inhibit the reaction, suggesting that molecular oxygen is essential for regenerating the active catalytic species and driving the decarboxylation forward efficiently.
Q: Can this method be applied to heterocyclic substrates?
A: Yes, the methodology demonstrates excellent functional group tolerance and substrate scope, successfully converting heterocyclic carboxylic acids such as benzothiophene and benzofuran derivatives into their corresponding halides with good yields, making it highly suitable for medicinal chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Halides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the silver-mediated decarboxylative halogenation described in CN107325002B. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial realities. Our state-of-the-art facilities are equipped to handle the specific thermal and oxidative requirements of this chemistry, while our stringent purity specifications and rigorous QC labs guarantee that every batch of aryl halide intermediate meets the exacting standards of the global pharmaceutical industry.
We invite forward-thinking partners to collaborate with us to leverage this cost-effective and green synthesis route for your next project. By integrating this technology into your supply chain, you can achieve substantial cost savings and enhance the sustainability profile of your product portfolio. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can accelerate your development timelines and optimize your manufacturing economics.
