Advanced Metal-Free Synthesis of Coumarin Aryl Ethers for Commercial Functional Materials
The landscape of organic synthesis is undergoing a significant transformation driven by the demand for greener, more efficient manufacturing processes. Patent CN110862367A introduces a groundbreaking methodology for the preparation of coumarin aryl ether compounds, leveraging intramolecular aryl migration reactions mediated by hypervalent iodine reagents. This technology represents a paradigm shift away from traditional transition metal-catalyzed cross-coupling, offering a pathway that is not only environmentally friendlier but also economically superior for large-scale production. By utilizing mild alkali conditions and eliminating the need for expensive noble metals, this invention addresses critical pain points in the supply chain for high-purity organic functional materials. The resulting compounds possess unique blue fluorescence properties, positioning them as valuable assets for advanced applications in optoelectronics and specialized pharmaceutical intermediates. As a reliable coumarin aryl ether supplier, understanding the depth of this technological advancement is crucial for strategic sourcing decisions.
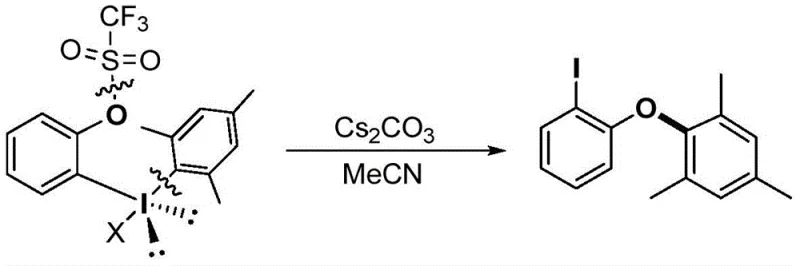
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aryl ether bonds, particularly in complex heterocyclic systems like coumarins, has relied heavily on transition metal catalysis, such as palladium or copper-mediated Ullmann or Buchwald-Hartwig type reactions. These conventional methodologies, while effective in laboratory settings, introduce substantial complications when translated to commercial manufacturing environments. The primary drawback lies in the residual metal contamination, which necessitates rigorous and costly purification steps to meet the stringent purity specifications required by the pharmaceutical and electronic materials industries. Furthermore, transition metal catalysts are often sensitive to air and moisture, requiring inert atmosphere conditions that increase operational complexity and energy consumption. The atom economy of these traditional routes is frequently suboptimal due to the generation of stoichiometric metal waste, leading to higher environmental disposal costs and a larger carbon footprint for the overall manufacturing process.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes hypervalent iodine chemistry to facilitate an intramolecular aryl migration, effectively bypassing the need for external transition metal catalysts. This method operates under remarkably mild conditions, typically employing cesium carbonate as a base in acetonitrile solvent at moderate temperatures ranging from 40°C to 60°C. The elimination of heavy metals not only simplifies the workup procedure but also drastically reduces the risk of product contamination, thereby enhancing the overall quality of the final coumarin aryl ether derivatives. The reaction demonstrates high atom utilization, aligning with modern principles of green chemistry and sustainable manufacturing. This technological leap allows for cost reduction in organic functional material manufacturing by streamlining the synthesis workflow and minimizing the reliance on volatile and expensive metal reagents, making it an attractive option for procurement teams focused on long-term supply stability.
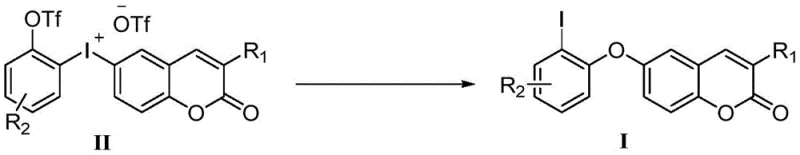
Mechanistic Insights into Hypervalent Iodine-Mediated Aryl Migration
The core of this innovative synthesis lies in the unique reactivity of the coumarin iodonium salt intermediate, which serves as the precursor for the aryl migration event. Under the influence of a mild base, the hypervalent iodine center undergoes a reductive elimination process that triggers the migration of the aryl group to the oxygen atom of the coumarin scaffold. This intramolecular mechanism is highly selective, minimizing the formation of side products that are commonly associated with intermolecular coupling reactions. The electronic properties of the substituents on the aromatic rings can be finely tuned to modulate the reaction rate and yield, offering chemists a versatile toolkit for generating a diverse library of derivatives. Understanding this mechanistic pathway is essential for R&D directors aiming to optimize the process for specific target molecules, as it provides a clear rationale for the observed high efficiency and selectivity without the need for ligand optimization typically required in metal catalysis.
Furthermore, the impurity profile generated by this metal-free route is significantly cleaner compared to traditional methods, which often suffer from homocoupling byproducts or dehalogenated species. The absence of metal catalysts removes a major source of variability in the reaction outcome, leading to more consistent batch-to-batch reproducibility. This consistency is paramount for commercial scale-up of complex polymer additives or pharmaceutical intermediates where regulatory compliance demands rigorous control over impurity spectra. The mechanism also allows for the tolerance of various functional groups, including halogens and electron-withdrawing groups, which might otherwise interfere with transition metal catalysts. This robustness ensures that the synthesis can be adapted to produce high-purity OLED material precursors or other specialty chemicals with minimal process redevelopment, securing a competitive advantage in time-to-market for new product introductions.
How to Synthesize Coumarin Aryl Ether Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these valuable compounds, starting from readily available substituted o-iodophenols and coumarin derivatives. The process involves the initial formation of a hypervalent iodine species, followed by coupling with the coumarin substrate to generate the key iodonium salt intermediate. This intermediate is then subjected to mild basic conditions to induce the critical aryl migration step, yielding the final fluorescent product after simple purification. Detailed standardized synthesis steps see the guide below, which breaks down the precise molar ratios, temperature controls, and solvent choices required to maximize yield and purity. Adhering to these parameters ensures that the beneficial effects of the invention, such as high atom utilization and mild reaction conditions, are fully realized in a practical setting.
- Preparation of substituted o-trifluoromethanesulfonate iodobenzene acetate by reacting substituted o-iodophenol with trifluoromethanesulfonic anhydride and Selectfluor.
- Formation of coumarin iodonium salt intermediate by coupling the acetate precursor with substituted coumarin under acidic conditions.
- Final cyclization via intramolecular aryl migration using cesium carbonate in acetonitrile at mild temperatures to yield the target fluorescent compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis technology translates into tangible strategic benefits that extend beyond mere chemical novelty. The elimination of transition metal catalysts directly addresses one of the most significant cost drivers in fine chemical manufacturing: the removal of trace metals to meet regulatory standards. By removing this step, the overall processing time is shortened, and the consumption of expensive scavenging resins or additional chromatography media is drastically reduced. This leads to substantial cost savings in the overall production budget, allowing for more competitive pricing structures without compromising on the quality of the high-purity coumarin derivatives supplied to downstream customers. Additionally, the mild reaction conditions reduce the energy load required for heating and cooling, contributing to a more sustainable and cost-effective operation.
- Cost Reduction in Manufacturing: The removal of palladium or copper catalysts eliminates the need for specialized metal scavengers and extensive purification protocols, which are often bottlenecks in production schedules. This simplification of the downstream processing workflow results in significantly reduced operational expenditures and lowers the barrier for entry for scaling these reactions to multi-ton quantities. The use of common reagents like cesium carbonate and acetonitrile further ensures that raw material costs remain stable and predictable, shielding the supply chain from the volatility often associated with precious metal markets. Consequently, the overall cost structure for producing these organic functional materials becomes more resilient and efficient.
- Enhanced Supply Chain Reliability: Relying on transition metals often exposes manufacturers to supply risks due to geopolitical factors and mining constraints affecting elements like palladium. By shifting to a hypervalent iodine-based system, the dependency on these critical raw materials is severed, enhancing the security of the supply chain. The reagents used in this process are commercially available in bulk quantities from multiple sources, ensuring reducing lead time for high-purity intermediates and preventing production delays caused by material shortages. This diversification of the supply base provides a robust buffer against market disruptions, guaranteeing continuous availability of key intermediates for clients in the pharmaceutical and electronics sectors.
- Scalability and Environmental Compliance: The mild nature of the reaction conditions, operating at temperatures between 40°C and 60°C, makes this process inherently safer and easier to scale compared to high-temperature or high-pressure alternatives. The reduced generation of heavy metal waste simplifies wastewater treatment and disposal, ensuring compliance with increasingly stringent environmental regulations globally. This environmental compatibility not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing entity. The ability to scale this process from laboratory grams to commercial tons with minimal modification demonstrates its readiness for immediate industrial adoption, supporting the growing demand for advanced functional materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational details outlined in the patent documentation, providing clarity for stakeholders evaluating this method for their own production needs. These insights help bridge the gap between theoretical chemical potential and practical industrial application, ensuring that all parties have a clear understanding of the capabilities and limitations of the process.
Q: What is the primary advantage of this synthesis method over traditional cross-coupling?
A: The primary advantage is the complete elimination of transition metal catalysts. Traditional methods often require palladium or copper, which necessitate expensive removal steps to meet pharmaceutical purity standards. This metal-free approach utilizing hypervalent iodine significantly simplifies downstream purification.
Q: Can this process be scaled for industrial production of organic functional materials?
A: Yes, the reaction conditions are notably mild, operating at temperatures between 40°C and 60°C with common bases like cesium carbonate. The absence of sensitive metal catalysts and the use of stable solvents like acetonitrile enhance the safety profile and feasibility for large-scale commercial manufacturing.
Q: What are the potential applications for these coumarin aryl ether compounds?
A: These compounds exhibit strong blue fluorescence, making them highly suitable as organic functional materials. Potential applications include use in OLED displays, chemical sensors, and as specialized intermediates in the synthesis of complex pharmaceutical agents requiring specific optical properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin Aryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of metal-free synthesis technologies like the one described in CN110862367A for the future of fine chemical manufacturing. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial realities. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We understand that the transition to new synthetic routes requires a partner who can navigate the complexities of process optimization while maintaining supply continuity and cost efficiency for your critical projects.
We invite you to collaborate with us to leverage this advanced chemistry for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this metal-free route can optimize your bottom line. Please contact us to request specific COA data for our available coumarin derivatives and to discuss route feasibility assessments for your target molecules. Together, we can drive innovation and efficiency in the production of next-generation organic functional materials.
