Advanced Metal-Free Synthesis of Coumarin Aryl Ethers for Functional Materials
The landscape of organic functional material synthesis is undergoing a significant transformation, driven by the urgent need for greener, more efficient, and cost-effective manufacturing protocols. A pivotal advancement in this domain is documented in patent CN110862367A, which introduces a novel class of coumarin aryl ether compounds characterized by their unique structural motifs and impressive blue fluorescence properties. This technology represents a paradigm shift away from traditional transition-metal-catalyzed cross-coupling reactions, which have long been the industry standard but suffer from inherent drawbacks related to metal contamination and harsh reaction environments. By leveraging hypervalent iodine chemistry, this invention enables an intramolecular aryl migration reaction that drastically improves atom utilization while maintaining exceptionally mild reaction conditions. For R&D directors and procurement specialists seeking reliable suppliers of high-purity electronic chemical intermediates, this metal-free approach offers a compelling value proposition that aligns with modern sustainability goals and stringent purity specifications required for optoelectronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aryl ether linkages, particularly within complex heterocyclic systems like coumarins, has relied heavily on transition-metal-catalyzed processes such as the Ullmann condensation or Buchwald-Hartwig coupling. While effective, these conventional methodologies impose significant burdens on the supply chain and production economics due to their dependence on expensive noble metal catalysts like palladium or copper. The presence of these metals necessitates rigorous and costly downstream purification steps to meet the stringent ppm-level residue limits mandated by the electronics and pharmaceutical industries. Furthermore, these reactions often require elevated temperatures, strong bases, and inert atmospheres, which increase energy consumption and operational complexity. The generation of heavy metal waste also presents substantial environmental compliance challenges, complicating waste treatment protocols and increasing the overall carbon footprint of the manufacturing process. These factors collectively hinder the scalability and cost-efficiency of producing high-value functional materials using traditional routes.
The Novel Approach
In stark contrast, the methodology disclosed in CN110862367A utilizes a hypervalent iodine-mediated intramolecular aryl migration that completely circumvents the need for transition metal catalysts. This innovative route operates under remarkably mild conditions, typically employing cesium carbonate as a base in acetonitrile solvent at moderate temperatures ranging from 40°C to 60°C. The elimination of heavy metals not only simplifies the purification workflow but also inherently enhances the purity profile of the final product, which is critical for applications in display and optoelectronic materials where trace impurities can quench fluorescence or degrade device performance. 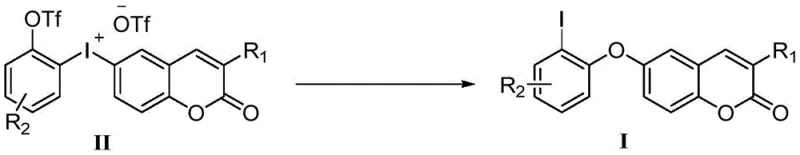 The process demonstrates excellent atom economy, as the hypervalent iodine reagent facilitates the migration efficiently without generating excessive stoichiometric byproducts. This technological leap allows for cost reduction in electronic chemical manufacturing by removing the expense of catalyst recovery and metal scavenging resins, thereby streamlining the entire production lifecycle from raw material input to final isolation.
The process demonstrates excellent atom economy, as the hypervalent iodine reagent facilitates the migration efficiently without generating excessive stoichiometric byproducts. This technological leap allows for cost reduction in electronic chemical manufacturing by removing the expense of catalyst recovery and metal scavenging resins, thereby streamlining the entire production lifecycle from raw material input to final isolation.
Mechanistic Insights into Hypervalent Iodine-Mediated Aryl Migration
The core of this synthetic breakthrough lies in the sophisticated manipulation of hypervalent iodine species to drive an intramolecular aryl migration. The reaction initiates with the formation of a pseudo-cyclic coumarin iodonium salt intermediate, which serves as a highly reactive electrophilic species. Upon exposure to a mild base, the electron-rich aromatic ring within the coumarin system attacks the iodine center, triggering a rearrangement that transfers the aryl group to the oxygen atom. This mechanism avoids the high-energy oxidative addition and reductive elimination steps typical of palladium cycles, instead proceeding through a concerted or stepwise migration pathway that is both kinetically favorable and thermodynamically stable. 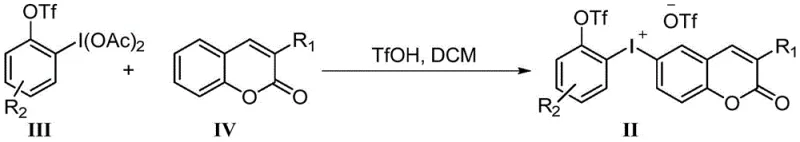 The use of triflic acid and specific iodine precursors ensures that the leaving group ability is optimized, facilitating the migration at lower thermal energies. For technical teams evaluating route feasibility, understanding this mechanism is crucial as it highlights the robustness of the chemistry against sensitive functional groups that might otherwise decompose under harsher transition-metal conditions.
The use of triflic acid and specific iodine precursors ensures that the leaving group ability is optimized, facilitating the migration at lower thermal energies. For technical teams evaluating route feasibility, understanding this mechanism is crucial as it highlights the robustness of the chemistry against sensitive functional groups that might otherwise decompose under harsher transition-metal conditions.
Impurity control is another critical aspect where this mechanism excels, particularly for stakeholders focused on the quality of high-purity OLED material precursors. Traditional methods often generate biaryl homocoupling byproducts or dehalogenated impurities that are structurally similar to the target and difficult to separate. The intramolecular nature of this aryl migration inherently suppresses intermolecular side reactions, leading to a cleaner crude reaction profile. The mild basic conditions prevent the hydrolysis of the coumarin lactone ring, a common degradation pathway in alkaline environments, ensuring the structural integrity of the fluorophore. This high level of chemoselectivity reduces the burden on chromatographic purification, allowing for higher overall yields and more consistent batch-to-batch quality, which is essential for maintaining supply chain reliability in the production of complex polymer additives or specialty chemicals.
How to Synthesize Coumarin Aryl Ether Efficiently
The practical implementation of this synthesis involves a sequential two-stage process beginning with the preparation of the hypervalent iodine precursor followed by the migration step. The initial phase requires the precise coupling of substituted ortho-iodophenols with coumarin derivatives under acidic conditions to generate the iodonium salt intermediate. This step demands careful temperature control and stoichiometry to ensure complete conversion without decomposing the sensitive iodine species. Once the intermediate is isolated, the subsequent migration is achieved by dissolving the salt in acetonitrile and treating it with cesium carbonate at moderate heat. Detailed standardized synthesis steps see the guide below.
- Preparation of hypervalent iodine precursor via triflation and oxidation of ortho-iodophenols.
- Coupling of the iodine precursor with substituted coumarins using triflic acid to form the iodonium salt intermediate.
- Intramolecular aryl migration induced by mild base (cesium carbonate) in acetonitrile to yield the final aryl ether product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free synthesis route translates into tangible strategic advantages that extend beyond simple unit cost metrics. The primary benefit lies in the drastic simplification of the supply chain for raw materials, as the process eliminates the dependency on volatile and expensive transition metal catalysts that are subject to geopolitical supply risks and price fluctuations. By removing the need for specialized metal scavengers and extensive filtration equipment, capital expenditure for production facilities is significantly reduced, and operational throughput is enhanced. This streamlined workflow facilitates faster turnaround times from order to delivery, addressing the critical need for reducing lead time for high-purity electronic chemical intermediates in a fast-paced market environment.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts removes a major cost center associated with both the purchase of noble metals and the disposal of hazardous metal waste. Without the need for expensive palladium or copper salts, the raw material bill of materials is optimized, and the costs linked to regulatory compliance for heavy metal discharge are substantially lowered. Furthermore, the simplified purification process reduces solvent consumption and labor hours dedicated to chromatography, resulting in comprehensive operational savings. These qualitative efficiencies allow for a more competitive pricing structure while maintaining healthy margins, making the commercial scale-up of complex organic functional materials more economically viable.
- Enhanced Supply Chain Reliability: Relying on readily available organic reagents and common inorganic bases like cesium carbonate ensures a robust and resilient supply chain that is less susceptible to disruptions. Unlike specialized catalysts that may have long lead times or single-source dependencies, the reagents for this hypervalent iodine chemistry are commoditized and accessible from multiple global vendors. This diversity in sourcing mitigates the risk of production stoppages and ensures continuous availability of key intermediates for downstream customers. The stability of the reagents also simplifies inventory management and storage requirements, further enhancing the reliability of supply for long-term contracts.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metals make this process inherently safer and easier to scale from laboratory benchtop to industrial tonnage. The reduced hazard profile lowers insurance premiums and safety infrastructure costs, while the green chemistry attributes align with increasingly strict environmental regulations globally. This compliance advantage future-proofs the manufacturing asset against tightening emission standards and facilitates smoother audits from environmentally conscious clients. The ability to scale without proportionally increasing waste treatment complexity is a decisive factor for sustainable long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel coumarin derivatives. The answers are derived directly from the patented technical specifications and process data to ensure accuracy and relevance for potential partners. Understanding these details is vital for assessing the fit of this technology within your existing R&D pipeline or procurement strategy.
Q: Does this synthesis require transition metal catalysts?
A: No, the patented process explicitly avoids the use of transition metal catalysts such as palladium or copper, relying instead on hypervalent iodine chemistry which eliminates heavy metal residue concerns.
Q: What are the reaction conditions for the key migration step?
A: The critical intramolecular aryl migration proceeds under mild conditions, typically between 40°C to 60°C in acetonitrile solvent using cesium carbonate as the base.
Q: What is the primary application of these compounds?
A: These coumarin aryl ether compounds exhibit strong blue fluorescence, making them highly suitable for use as organic functional materials in optoelectronic applications and potential pharmaceutical research.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin Aryl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the next generation of organic functional materials. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready product is seamless and efficient. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of coumarin aryl ether meets the exacting standards required for high-performance optoelectronic applications. We are committed to delivering not just chemicals, but reliable solutions that enhance your product's performance and market competitiveness.
We invite you to engage with our technical procurement team to discuss how this innovative route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a partnership built on transparency, technical excellence, and mutual growth.
