Optimizing Cariprazine Production: A Technical Analysis of Patent CN112533908B
The pharmaceutical landscape for antipsychotic medications continues to evolve, driven by the need for more efficient and purer active pharmaceutical ingredients (APIs). A significant advancement in this domain is detailed in Chinese Patent CN112533908B, which outlines a robust synthetic method for Cariprazine, a potent dopamine D3 and D2 receptor partial antagonist. This patent addresses critical bottlenecks in the manufacturing of this complex molecule, specifically targeting the final acylation step that converts the key intermediate (Compound I) into the final drug substance (Compound II). For R&D directors and procurement specialists alike, understanding the nuances of this technology is vital, as it promises not only superior chemical purity exceeding 99.0% but also a dramatic reduction in processing time. The innovation lies in the strategic use of aqueous inorganic bases during the acylation with dimethylcarbamoyl chloride, a departure from the prolonged and impurity-prone methods of the past. This report delves deep into the technical merits and commercial implications of adopting this refined synthesis pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN112533908B, the synthesis of Cariprazine was plagued by inefficiencies that posed significant challenges for large-scale manufacturing. As illustrated in earlier patent literature such as WO 2005012266, the conventional condensation of Compound I with dimethylcarbamoyl chloride typically required stirring at room temperature for an exhaustive 48 hours. 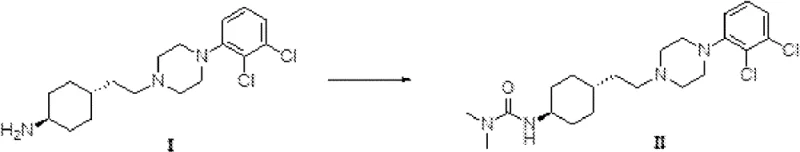 Furthermore, alternative methods disclosed in documents like CN105330616 necessitated low-temperature dropwise addition followed by extended reaction periods of up to 36 hours to achieve completion. These protracted timelines are not merely inconvenient; they represent a substantial opportunity cost in a commercial setting, tying up reactor capacity and increasing energy consumption. More critically, these older methodologies suffered from poor selectivity, leading to the gradual accumulation of difficult-to-remove double-condensation impurities. As the reaction time extended beyond 18 hours, the levels of these specific byproducts would rise from negligible amounts to as high as 0.6%, complicating downstream purification and negatively impacting overall yield, which often hovered around a mediocre 63% to 65%.
Furthermore, alternative methods disclosed in documents like CN105330616 necessitated low-temperature dropwise addition followed by extended reaction periods of up to 36 hours to achieve completion. These protracted timelines are not merely inconvenient; they represent a substantial opportunity cost in a commercial setting, tying up reactor capacity and increasing energy consumption. More critically, these older methodologies suffered from poor selectivity, leading to the gradual accumulation of difficult-to-remove double-condensation impurities. As the reaction time extended beyond 18 hours, the levels of these specific byproducts would rise from negligible amounts to as high as 0.6%, complicating downstream purification and negatively impacting overall yield, which often hovered around a mediocre 63% to 65%.
The Novel Approach
The methodology presented in the subject patent offers a transformative solution to these longstanding issues by introducing an aqueous inorganic base system into the reaction matrix. Instead of relying solely on organic bases or prolonged thermal exposure, this novel approach facilitates the acylation in the presence of bases such as sodium carbonate, potassium carbonate, or sodium hydroxide within a biphasic solvent system. 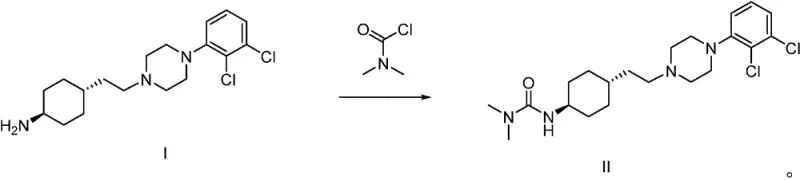 This shift in reaction engineering allows the process to reach completion in a fraction of the time, typically between 12 to 15 hours, representing a massive gain in throughput efficiency. The result is a process that is not only faster but also cleaner, yielding Cariprazine with purity levels consistently above 99.0% and isolated yields soaring to over 90% in optimized examples. By fundamentally altering the reaction environment, this method effectively mitigates the formation of the problematic double-condensation side products, ensuring a much smoother path to the final API.
This shift in reaction engineering allows the process to reach completion in a fraction of the time, typically between 12 to 15 hours, representing a massive gain in throughput efficiency. The result is a process that is not only faster but also cleaner, yielding Cariprazine with purity levels consistently above 99.0% and isolated yields soaring to over 90% in optimized examples. By fundamentally altering the reaction environment, this method effectively mitigates the formation of the problematic double-condensation side products, ensuring a much smoother path to the final API.
Mechanistic Insights into Aqueous Base-Catalyzed Acylation
To fully appreciate the value of this synthesis, one must understand the mechanistic underpinnings that drive its success. The core transformation involves the nucleophilic attack of the secondary amine on Compound I against the carbonyl carbon of dimethylcarbamoyl chloride. In traditional organic base systems, the generation of the free amine and the scavenging of the generated HCl can be sluggish or lead to localized high concentrations of reactive species that promote over-reaction. The introduction of an aqueous inorganic base creates a dynamic interface where the deprotonation of the amine salt (if used) and the neutralization of the acid byproduct occur rapidly and efficiently. This ensures a steady concentration of the nucleophilic amine is available for reaction without promoting excessive reactivity that leads to side products. The use of water-immiscible solvents like dichloromethane or 2-methyltetrahydrofuran further aids in this by creating a distinct phase boundary that helps manage the reaction kinetics.
A critical aspect of this mechanism is the suppression of the double-condensation impurity, a structural analog where two dimethylcarbamoyl groups attach to the molecule. 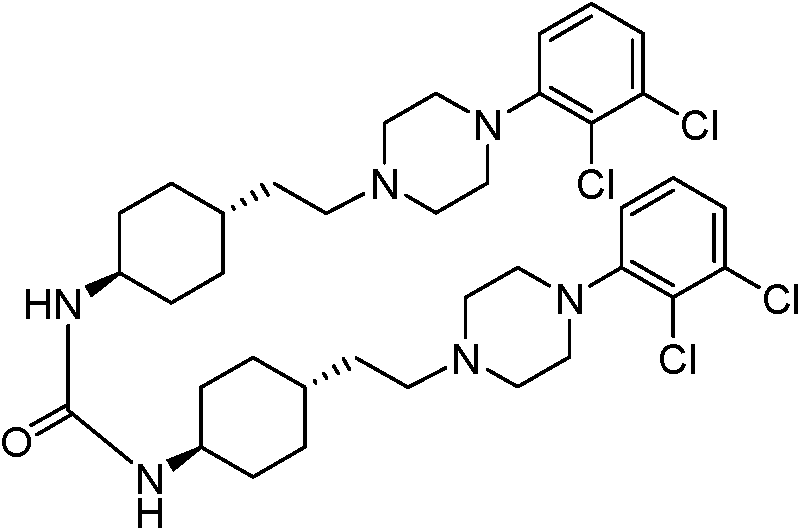 In conventional routes, as the reaction drags on, the probability of this secondary acylation increases, and because this impurity has poor solubility characteristics similar to the product, it is notoriously difficult to purge via crystallization. The new method's ability to drive the reaction to completion quickly minimizes the window of time during which this side reaction can occur. Furthermore, the specific choice of inorganic bases appears to modulate the nucleophilicity of the amine just enough to favor mono-acylation. This precise control over the impurity profile is a hallmark of advanced process chemistry, directly translating to reduced burden on the purification team and higher overall material throughput for the supply chain.
In conventional routes, as the reaction drags on, the probability of this secondary acylation increases, and because this impurity has poor solubility characteristics similar to the product, it is notoriously difficult to purge via crystallization. The new method's ability to drive the reaction to completion quickly minimizes the window of time during which this side reaction can occur. Furthermore, the specific choice of inorganic bases appears to modulate the nucleophilicity of the amine just enough to favor mono-acylation. This precise control over the impurity profile is a hallmark of advanced process chemistry, directly translating to reduced burden on the purification team and higher overall material throughput for the supply chain.
How to Synthesize Cariprazine Efficiently
Implementing this synthesis requires careful attention to the specific parameters outlined in the patent to ensure reproducibility and safety. The process begins with the selection of appropriate starting materials, where Compound I can be introduced either as the free base or as a stable salt such as the dihydrochloride, offering flexibility in raw material sourcing. The reaction is conducted in a biphasic system, typically involving an organic solvent like dichloromethane and an aqueous layer containing the inorganic base at concentrations ranging from 5% to 30% by weight. Temperature control is also paramount, with the reaction proceeding optimally between 15°C and 50°C, allowing for operation under mild conditions that do not require extreme cooling or heating infrastructure.
- Dissolve Compound I (or its salt) in a water-immiscible solvent such as dichloromethane or 2-methyltetrahydrofuran.
- Add an aqueous solution of an inorganic base (e.g., Na2CO3 or NaOH) to the reaction mixture.
- Dropwise add dimethylcarbamoyl chloride while maintaining the temperature between 15°C and 50°C, then stir until completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic advantages that go beyond simple chemical yield. The primary benefit lies in the drastic simplification of the manufacturing workflow, which directly correlates to operational expenditure savings. By eliminating the need for extended reaction times of nearly two days, facilities can significantly increase their batch turnover rate, effectively expanding production capacity without the need for capital investment in new reactors. This efficiency gain is compounded by the simplified workup procedure; the ability to simply separate phases and crystallize the product from the organic layer removes the need for complex chromatographic purifications or multiple recrystallization steps often required to remove stubborn impurities in older methods.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the elimination of inefficient steps and the reduction of waste. Traditional methods often resulted in significant material loss during the purification of the double-condensation impurity, sometimes dropping final yields to as low as 20% after extensive cleaning. In contrast, this new method achieves high crude purity, meaning less starting material is wasted, and fewer solvents are consumed during purification. The use of inexpensive, commodity inorganic bases like sodium carbonate or sodium hydroxide further drives down the cost of goods sold (COGS) compared to expensive organic bases. Additionally, the shorter cycle time reduces utility costs associated with stirring and temperature control, contributing to a leaner and more cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this method ensures greater consistency in delivery schedules. The reliance on readily available, non-hazardous inorganic reagents reduces the risk of supply disruptions that can occur with specialized organic catalysts or sensitive reagents. The process has been successfully demonstrated on a multi-kilogram scale (up to 10kg in the examples), proving its viability for commercial production without the teething problems often associated with scaling up delicate chemical transformations. This scalability assures partners that large volume orders can be fulfilled reliably, reducing the lead time for high-purity pharmaceutical intermediates and stabilizing the supply of this critical medication to the market.
- Scalability and Environmental Compliance: The environmental footprint of pharmaceutical manufacturing is an increasingly critical metric, and this synthesis aligns well with green chemistry principles. The reduction in reaction time and the avoidance of excessive solvent usage for purification directly lower the E-factor (mass of waste per mass of product). The ability to use solvents like 2-methyltetrahydrofuran, which is considered a greener alternative to traditional chlorinated solvents, further enhances the sustainability profile of the process. For global suppliers, adhering to strict environmental regulations is mandatory, and a process that generates less hazardous waste and consumes less energy provides a significant compliance advantage, facilitating smoother regulatory approvals and audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Cariprazine synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear picture of what potential partners can expect. Understanding these details is crucial for making informed decisions about technology transfer and long-term supply agreements.
Q: How does the new synthesis method improve impurity profiles compared to prior art?
A: The novel method utilizing aqueous inorganic bases significantly suppresses the formation of double-condensation impurities, keeping levels below 0.1% compared to up to 0.6% in conventional methods, thereby simplifying purification.
Q: What are the scalability benefits of this Cariprazine synthesis route?
A: The process has been successfully demonstrated on a 10kg scale with high yields (over 85%) and uses common industrial solvents and reagents, making it highly suitable for commercial scale-up.
Q: Why is the reaction time reduced in this patented process?
A: By optimizing the base system and reaction conditions, the acylation proceeds rapidly, completing in approximately 12 to 15 hours, whereas traditional methods often require 36 to 48 hours.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cariprazine Supplier
The technical breakthroughs detailed in patent CN112533908B represent a significant leap forward in the manufacturing of Cariprazine, yet realizing this potential requires a partner with deep expertise in process development and scale-up. NINGBO INNO PHARMCHEM stands ready to leverage this advanced chemistry, bringing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality ensures that every batch meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. We understand that in the pharmaceutical industry, consistency is key, and our team is dedicated to maintaining the highest standards of GMP compliance throughout the manufacturing lifecycle.
We invite you to explore how our optimized synthesis routes can enhance your supply chain resilience and reduce your overall manufacturing costs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical strategy available.
