Advanced Industrial Preparation of Palbociclib Intermediate via Green Catalytic Hydrogenation
Advanced Industrial Preparation of Palbociclib Intermediate via Green Catalytic Hydrogenation
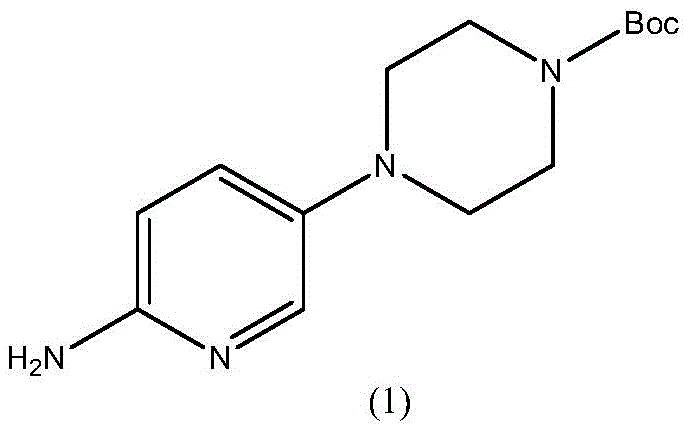
The pharmaceutical industry continuously demands more efficient, cost-effective, and environmentally sustainable routes for synthesizing critical oncology drug intermediates. Patent CN113636973B, published in April 2023, discloses a groundbreaking industrial preparation method for 4-(6-aminopyridin-3-yl)piperazine-1-carboxylic acid tert-butyl ester, a key building block for the CDK4/6 inhibitor Palbociclib. This novel methodology addresses long-standing challenges in impurity control, specifically the formation of difficult-to-remove piperazine dimers and the degradation of the Boc protecting group during reduction. By leveraging acid-catalyzed nucleophilic substitution in a recyclable alcohol-water system and a stabilized catalytic hydrogenation protocol, this process delivers a high-purity product exceeding 99.8% with superior color properties, representing a significant leap forward for reliable pharmaceutical intermediate supplier networks seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
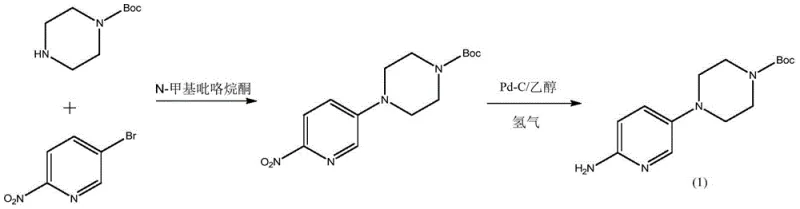
Historically, the synthesis of this critical pyridine-piperazine scaffold has been plagued by economic and technical inefficiencies. Early literature methods, such as those reported by Iameda et al., relied on the direct coupling of 5-bromo-2-nitropyridine with 1-Boc-piperazine. While chemically straightforward, this route suffers from prohibitive costs due to the high price of 1-Boc-piperazine, which itself requires synthesis from piperazine and Boc anhydride with poor atom economy and significant waste generation. Furthermore, alternative routes utilizing 5-chloro-2-nitropyridine and free piperazine often struggle with selectivity. Under alkaline conditions required for the initial substitution, the highly nucleophilic piperazine tends to react at both nitrogen centers, leading to the formation of bis-substituted dimeric impurities (Formula 4 and Formula 5). These lipophilic dimers are notoriously difficult to separate from the desired mono-substituted product using standard crystallization techniques, often carrying through to the final API and compromising safety profiles.
The Novel Approach
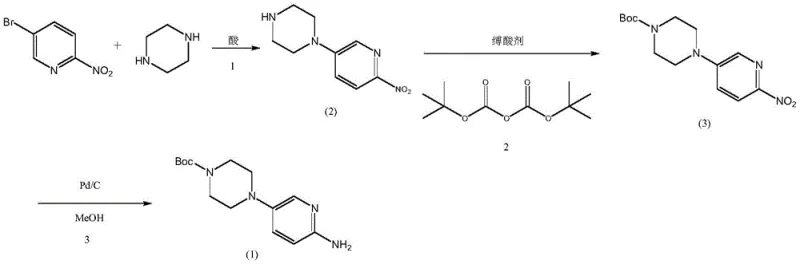
The patented innovation introduces a paradigm shift by reversing the protection strategy and optimizing the reaction medium. Instead of starting with protected piperazine, the process begins with free piperazine and 5-bromo-2-nitropyridine in a mixed solvent of n-butanol and water, catalyzed by hydrobromic acid. This acidic environment ensures that piperazine exists primarily as a mono-salt, effectively masking one nitrogen atom and sterically hindering the formation of the problematic dimeric impurities. Following the substitution, the intermediate is isolated as a wet cake and directly subjected to Boc protection in a biphasic toluene-water system. This telescoped approach eliminates the need for drying steps and allows for the efficient removal of inorganic salts. The final hydrogenation step is meticulously controlled with the addition of a weak base, ensuring the stability of the carbamate moiety while reducing the nitro group, resulting in a robust, scalable, and economically superior manufacturing route suitable for global supply chains.
Mechanistic Insights into Acid-Catalyzed Substitution and Stabilized Hydrogenation
The core chemical innovation lies in the precise manipulation of piperazine's nucleophilicity through pH control. In the initial step, the use of hydrobromic acid creates an equilibrium where the piperazine mono-hydrobromide salt is the dominant species. This protonation significantly reduces the nucleophilicity of one nitrogen atom, directing the substitution reaction exclusively to the unprotonated nitrogen. This mechanistic control is crucial for suppressing the formation of the bis-pyridyl piperazine impurities (Formula 4 and 5), which are generated when free piperazine reacts twice. Even if trace amounts of these dimers form, the subsequent workup leverages their differential solubility; the dimeric impurities are insoluble in the aqueous acidic phase and are removed via filtration before the product is precipitated by basification. This dual mechanism of kinetic control (via salt formation) and thermodynamic separation (via solubility differences) ensures an intermediate purity of over 99.5% prior to protection.
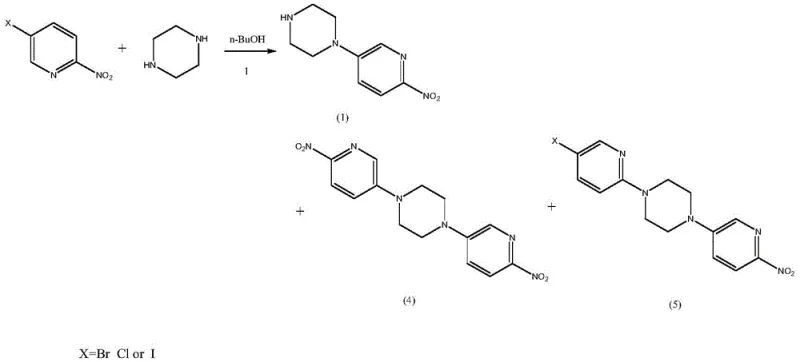
Furthermore, the patent elucidates a critical stability issue during the final catalytic hydrogenation. Trace amounts of bromo-containing impurities (Formula 5) can undergo hydrodebromination over the palladium or platinum catalyst, releasing hydrobromic acid (HBr) in situ. In traditional protocols, this localized acidity catalyzes the hydrolysis of the tert-butoxycarbonyl (Boc) group, leading to deprotected amines and potentially genotoxic urethane derivatives (as shown in Formula 6 degradation pathways). The novel process counters this by introducing a stoichiometric amount of sodium acetate as an acid scavenger. This weak base effectively neutralizes the generated HBr without interfering with the hydrogenation catalyst, thereby preserving the Boc group integrity. This subtle yet powerful modification prevents the formation of complex impurity profiles and ensures the final product meets stringent safety specifications required for oncology therapeutics.
How to Synthesize Tert-Butyl 4-(6-Aminopyridin-3-yl)Piperazine-1-Carboxylate Efficiently
The implementation of this industrial method requires careful attention to solvent ratios and temperature controls to maximize yield and purity. The process is designed to be telescoped where possible, minimizing material handling and exposure to moisture. The following guide outlines the critical operational parameters derived from the patent examples, focusing on the transition from laboratory scale to industrial production. Operators should note the importance of the azeotropic distillation step for solvent recovery, which is key to the process's environmental and economic benefits. For detailed standard operating procedures and safety data sheets, please refer to the technical documentation provided below.
- React 5-bromo-2-nitropyridine with piperazine in an alcohol-water mixed solvent using hydrobromic acid as a catalyst to form the acid salt, followed by azeotropic solvent recovery and alkaline precipitation to isolate high-purity 1-(6-nitropyridin-3-yl)piperazine.
- React the wet intermediate directly with Boc anhydride in a toluene-water biphasic system using sodium carbonate as an acid-binding agent to generate 4-(6-nitropyridin-3-yl)piperazine-1-tert-butyl carboxylate.
- Perform catalytic hydrogenation using platinum carbon in methanol with sodium acetate added to neutralize generated acids, followed by activated carbon decolorization and crystallization to obtain the final high-purity amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic advantages beyond mere technical feasibility. The shift from expensive protected starting materials to commodity chemicals like free piperazine and 5-bromo-2-nitropyridine drastically reduces the raw material cost base. Moreover, the ability to recycle the n-butanol solvent through azeotropic distillation significantly lowers waste disposal costs and reduces the overall solvent consumption per kilogram of product. This aligns perfectly with modern green chemistry initiatives and reduces the environmental footprint of the manufacturing site, facilitating smoother regulatory approvals and community relations.
- Cost Reduction in Manufacturing: The elimination of the pre-formed 1-Boc-piperazine step removes a significant cost driver, as the commercial price of this reagent includes margins for its own synthesis and purification. By performing the Boc protection in situ on the wet intermediate, the process saves energy costs associated with drying and reduces the total number of unit operations. The use of cheaper catalysts like platinum carbon, which can be recovered and recycled, further contributes to a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for API intermediates.
- Enhanced Supply Chain Reliability: The robustness of the purification strategy, which relies on simple filtration and crystallization rather than complex chromatography, ensures consistent batch-to-batch quality. The removal of genotoxic impurities at the intermediate stage mitigates the risk of batch rejection during final API testing. This reliability is critical for maintaining continuous supply to downstream pharmaceutical partners, reducing the lead time for high-purity pharmaceutical intermediates and preventing costly production delays caused by out-of-specification materials.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard glass-lined or stainless steel reactors common in fine chemical manufacturing. The aqueous workups generate wastewater that is primarily inorganic salts, which is easier and cheaper to treat compared to organic-heavy waste streams from other methods. The high yield and purity achieved reduce the need for re-processing, thereby minimizing resource consumption. This scalability ensures that the supply chain can easily ramp up from pilot batches to multi-ton commercial production to meet the growing demand for Palbociclib and related CDK inhibitors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and quality control of this intermediate. These answers are derived directly from the experimental data and claims within the patent specification, providing transparency for potential partners evaluating this technology for their supply chain.
Q: How does the new process prevent the formation of piperazine disubstituted impurities?
A: The process utilizes an acid-catalyzed nucleophilic substitution where piperazine forms a mono-acid salt. This protonation protects one nitrogen atom, effectively preventing the second nucleophilic attack that leads to dimeric impurities (Formula 4 and 5). Additionally, the resulting dimeric impurities have low solubility in the aqueous acidic phase and are removed via filtration before the basification step.
Q: Why is sodium acetate added during the catalytic hydrogenation step?
A: During hydrogenation, trace amounts of bromo-impurities undergo debromination, releasing hydrobromic acid (HBr). This acidity can catalyze the hydrolysis of the sensitive Boc protecting group, leading to genotoxic impurities. Sodium acetate acts as a buffer to neutralize the HBr in situ, preserving the integrity of the Boc group and ensuring product safety.
Q: What are the purity specifications achievable with this industrial method?
A: The patented method achieves a final product purity of greater than or equal to 99.8% (HPLC) with a white to off-white appearance. This is accomplished through a combination of selective precipitation, biphasic washing, and a final activated carbon decolorization and crystallization step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tert-Butyl 4-(6-Aminopyridin-3-yl)Piperazine-1-Carboxylate Supplier
As the global demand for targeted cancer therapies continues to rise, securing a stable and high-quality supply of key intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to support your development and commercial needs with our advanced manufacturing capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-(6-aminopyridin-3-yl)piperazine-1-carboxylic acid tert-butyl ester meets the highest industry standards for oncology drug synthesis.
We invite you to collaborate with us to leverage this innovative synthetic route for your projects. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this optimized process can improve your bottom line. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples to evaluate the superior quality of our intermediates.
